In flow cytometry experiments, elevated background fluorescence caused by non-specific staining is one of the most common factors compromising data quality. Excessive background not only blurs the boundary between positive and negative populations and reduces the signal-to-noise ratio, but can also lead to misinterpretation of experimental results, compelling researchers to conduct repeated troubleshooting and optimisation.
In practice, high background fluorescence is rarely driven by a single factor. Instead, it typically arises from the combined effects of experimental design, sample characteristics and subtle procedural details. Effective resolution therefore requires more than ad hoc parameter adjustment. A clear understanding of the underlying mechanisms, identification of often-overlooked sources of interference, and the application of targeted diagnostic and optimisation strategies are essential to establish a robust and reproducible workflow.
1. Common Operational Pitfalls Leading to Non-specific Staining
Many high-background issues originate from seemingly reasonable “standard practices”. Accurately recognising these pitfalls is the first step in efficient troubleshooting, helping to avoid blind optimisation and enabling rapid identification of the root cause.
Pitfall 1: Assuming Fc blocking is universally effective
Interpretation
The primary function of Fc blockers is to prevent interactions between the Fc region of antibodies and Fc receptors expressed on the cell surface. However, Fc blocking is not universally effective against all forms of non-specific staining. In myeloid cell samples, significant background signals are often observed even after thorough Fc receptor blockade.
Mechanistic considerations
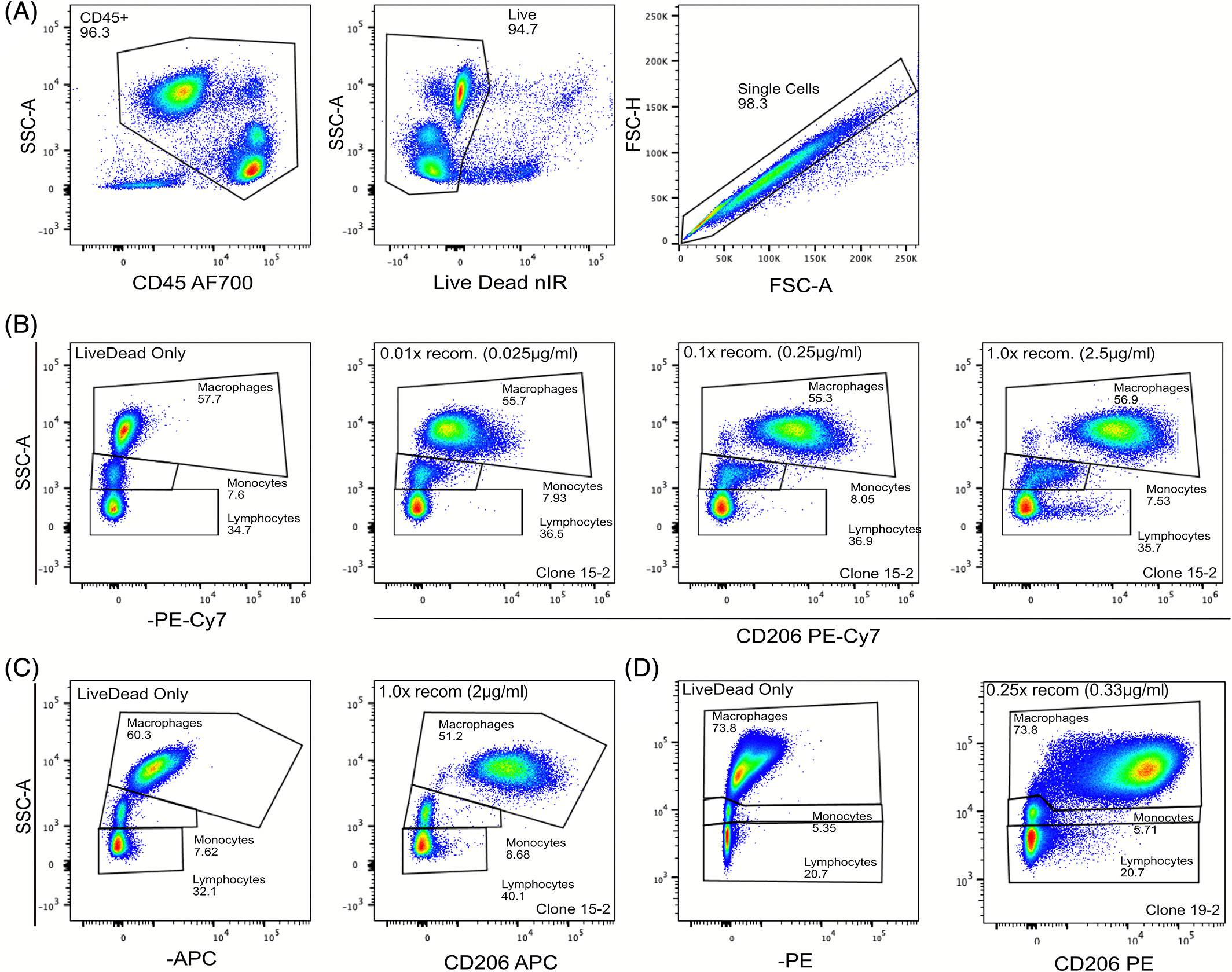
Cyanine-based fluorochromes (such as Cy5 and tandem dyes) frequently exhibit Fc-independent background staining in monocytes, macrophages and dendritic cells. Importantly, this elevated background persists even when Fc receptors are fully blocked, indicating that it is not driven by classical Fc-mediated binding.
The exact molecular mechanism remains incompletely understood. Some studies have suggested potential interactions between cyanine fluorophores and highly expressed Fcγ receptors (e.g. CD64) or other membrane-associated structures. However, broader experimental evidence indicates that this phenomenon is strongly dye-dependent. Under identical antibody clones and cellular conditions, replacing cyanine dyes with non-cyanine fluorophores (such as APC or Alexa Fluor 647) often results in a marked reduction in background.
This observation suggests that the effect is more likely attributable to the physicochemical properties of cyanine fluorophores themselves—such as increased hydrophobicity—leading to non-specific interactions with myeloid cell membranes, rather than binding mediated by a single receptor.
Optimisation strategies
- For myeloid cell panels, avoid cyanine-based dyes (e.g. Cy5, PE-Cy5, PE-Cy7) whenever feasible
- When their use is unavoidable, supplement staining buffers with 1–2% BSA or serum proteins to competitively reduce non-specific dye–membrane interactions
- In selected systems, low concentrations of non-ionic surfactants may be evaluated to reduce hydrophobic adsorption, with careful assessment of membrane integrity and antibody binding
- Dedicated blocking reagents for cyanine dyes, such as True-Stain Monocyte Blocker (BioLegend) or phosphorothioate oligonucleotide-based blockers (e.g. Oligo-Block, Sigma-Aldrich), may also be considered
Note: Fc blocking remains necessary, but its limitations should be clearly recognised to avoid overreliance.
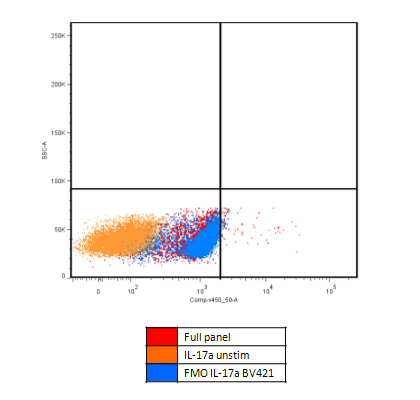
Pitfall 2: Overuse of isotype controls in multicolour panels
Interpretation
Isotype controls are designed to assess Fc-mediated non-specific binding and certain forms of intracellular cross-reactivity. However, over-reliance on isotype controls in multicolour experiments increases experimental complexity and may introduce misleading interpretations when critical parameters are not well matched.
Key issue
Fluorophore-to-protein (F:P) ratios can vary significantly among antibodies from different manufacturers or production batches. For example, if a CD4-PE antibody has an F:P ratio of 2:1 while the corresponding isotype control has an F:P ratio of 4:1, the isotype control may display markedly higher fluorescence intensity even at identical antibody concentrations, leading to the false conclusion that the target antibody exhibits excessive background.
Optimisation strategies
- In multicolour experiments, prioritise fluorescence-minus-one (FMO) controls to assess background distribution under actual staining conditions
- If isotype controls are required, ensure strict matching of manufacturer, fluorophore and antibody concentration to maintain comparability
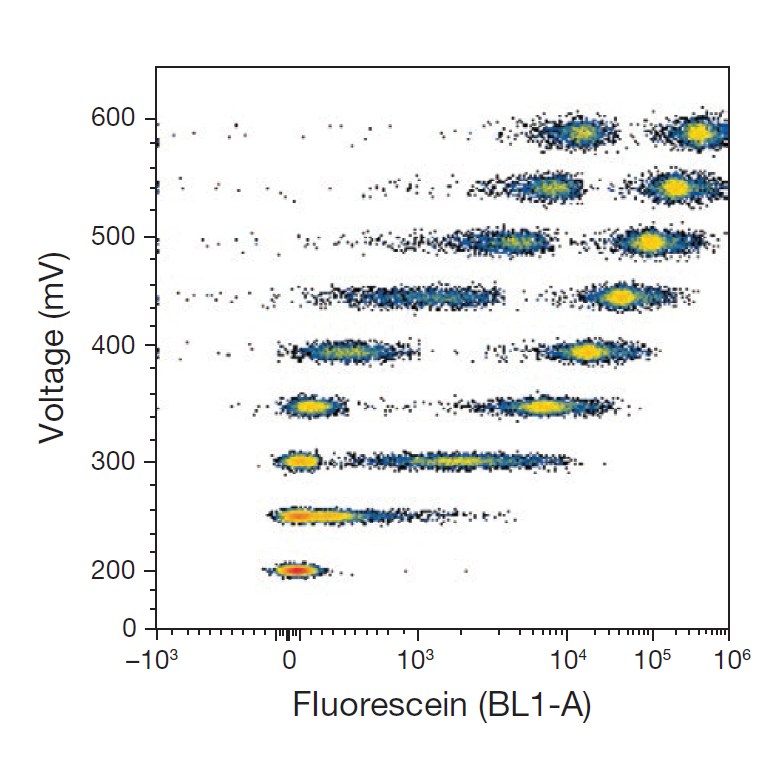
Pitfall 3: Setting detector voltage “as low as possible” to reduce noise
Interpretation
The goal of voltage optimisation is to maximise population resolution while preserving dynamic range—not simply to suppress background signals. Excessively low voltage settings often compress the signal distribution of low-expressing antigens, making the distinction between positive populations and background less clear.
Optimisation strategies
- Use both unstained and positive controls to ensure autofluorescence peaks fall within the linear detection range
- Adjust voltage to achieve clear separation between positive and background peaks, balancing signal-to-noise ratio and dynamic range
2. Frequently Overlooked Sources of Non-specific Staining
When common operational issues have been excluded but background remains elevated, further investigation should focus on context-specific factors that frequently arise under particular experimental conditions.
Hidden factor 1: Metabolic activation–associated baseline fluorescence shifts
In PMA-activated PBMCs or cytokine stimulation assays, simultaneous increases in background across multiple channels are commonly observed. This phenomenon is typically unrelated to antibody or dye quality, but rather reflects changes in cellular metabolic state.
Mechanistic explanation
Metabolic activation increases intracellular levels of endogenous fluorophores such as NADH and flavin cofactors, leading to enhanced autofluorescence. Concurrent increases in membrane permeability further amplify fluorescence leakage, collectively elevating baseline signals.
Targeted optimisation
- Introduce additional wash steps following activation
- Reduce antibody concentrations
- Preferentially select longer-wavelength fluorophores to minimise spectral overlap with endogenous fluorescence
Hidden factor 2: Fixation/permeabilisation-induced “epitope traps” in intracellular staining
Elevated background in intracellular staining is often driven by mismatched fixation and permeabilisation conditions rather than insufficient antibody specificity.
Core mechanisms
- Imbalanced fixation time or concentration resulting in over-crosslinking or under-fixation
- Organic solvent fixation altering epitope conformation
- Inadequate post-fixation washing, leaving residual aldehydes that react non-specifically with antibodies
Precision optimisation
- Select fixation and permeabilisation reagents based on antigen type; cytokine and transcription factor detection require distinct protocols
- Strictly control fixation time (15–30 min) and temperature (20–25°C); for transcription factor assays, lower temperatures (e.g. 4°C) may help reduce antigen degradation
- Introduce protein blocking steps after permeabilisation to reduce intracellular non-specific binding sites
3. A Mechanism-driven Workflow for Rapid High-background Troubleshooting
Shifting from “phenotype observation” to “mechanistic localisation”
After addressing common pitfalls and hidden contributors, background optimisation should adhere to a central principle:
identify the source of background signal before selecting the intervention, rather than relying on empirical parameter adjustment.
Based on the most frequent mechanisms underlying non-specific staining, high-background issues can be categorised into four major sources, enabling a structured, stepwise troubleshooting pathway.
1. Rapid troubleshooting workflow
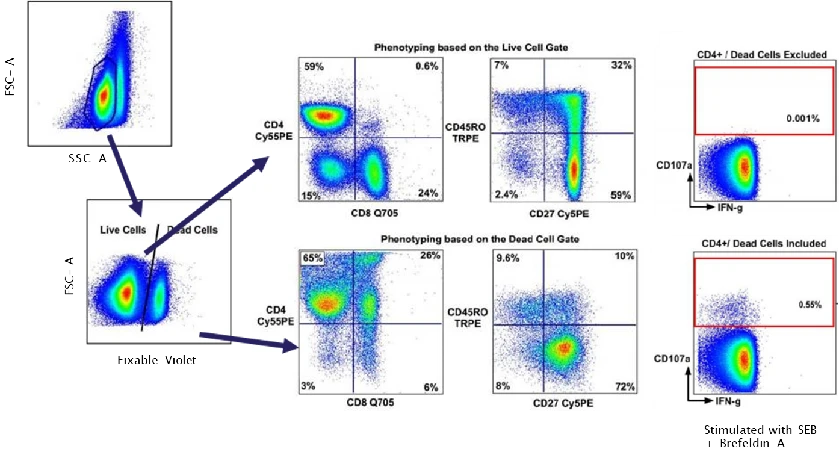
Step 1 | Assess association with dead cells or debris
Examine overlap between background signals and viability dye–positive populations (e.g. DAPI, 7-AAD), and inspect FSC/SSC plots for excessive debris or abnormal populations.
Interpretation:
If background is concentrated in dead cells or debris, it typically reflects non-specific dye or antibody penetration into cells with compromised membrane integrity rather than antigen-specific binding.
Action:
Prioritise sample quality improvement and dead-cell exclusion before adjusting antibody or compensation settings.
Step 2 | Use FMO controls to identify fluorophore-related background
Evaluate FMO controls within the target population rather than across total cells, and determine whether a specific channel shows global baseline elevation.
Interpretation:
Elevated background in FMO controls indicates fluorophore-driven effects (e.g. spectral spillover or dye–cell interactions), rather than antibody specificity issues.
Action:
Focus on fluorophore selection, voltage optimisation and staining conditions.
Step 3 | Perform fluorophore swap validation
Maintain antibody clone, concentration and sample conditions, changing only the fluorophore.
Interpretation:
If background is markedly reduced with non-cyanine dyes (e.g. APC, Alexa Fluor 647), the issue is likely dye–cell non-specific interaction, particularly common in myeloid cells.
Step 4 | Review fixation/permeabilisation workflows (intracellular staining)
Compare surface and intracellular staining results to determine whether background elevation is intracellular-specific.
Interpretation:
If elevated background appears only in intracellular staining and is mirrored in FMO controls, fixation/permeabilisation mismatch is the most likely cause.
2. Scenario-specific optimisation strategies
| Experimental scenario |
Primary background source |
Mechanistic explanation |
Targeted optimisation |
| Multicolour panels (≥6 colours) |
Accumulated spectral spillover |
Overlapping emission amplifies baseline noise |
Optimise fluorophore combinations; titrate antibodies individually before panel assembly |
| Myeloid cell staining |
Autofluorescence and dye-dependent interactions |
Non-specific interactions between cyanine dyes and myeloid membranes |
Avoid Cy5/PE-Cy5 dyes; add 1–2% BSA to staining buffers |
| Activated/stimulated samples |
Enhanced endogenous fluorescence |
Metabolic activation increases autofluorescence and permeability |
Increase washes; reduce antibody concentration; use longer-wavelength dyes |
| Intracellular transcription factor staining |
Fixation/permeabilisation artefacts |
Over-crosslinking and exposure of intracellular binding sites |
Match protocols to antigen type; include post-permeabilisation blocking |
| High dead cell/debris content |
Non-specific antibody penetration |
Loss of membrane integrity causes false positives |
Filter samples; add EDTA; apply combined viability and scatter gating |
Conclusion
Resolving high background in flow cytometry requires a comprehensive logical framework that includes pitfall identification, mechanistic localisation, and targeted optimisation. Elevated background caused by non-specific staining is not a random artefact, but a controllable experimental variable when addressed systematically.
By integrating an understanding of common misconceptions, hidden contributors and scenario-specific strategies, researchers can substantially improve troubleshooting efficiency and optimisation success, ultimately ensuring greater reliability and interpretability of flow cytometry data.
About Us
abinScience was founded in 2023 as a strategic venture of AtaGenix (established 2011), dedicated to delivering premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies


 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский