Psoriasis is a chronic inflammatory skin disorder mediated by immune dysregulation, with genetic, environmental, and immune factors acting as key drivers. Non-contagious yet marked by persistent, relapsing symptoms, it severely impacts patients’ quality of life, making it a global research priority. Prevalence varies significantly across regions and ethnic groups, ranging from 0.1% to 8%, with a higher incidence in Caucasians (2%–5%). Globally, over 125 million people are affected by this condition.
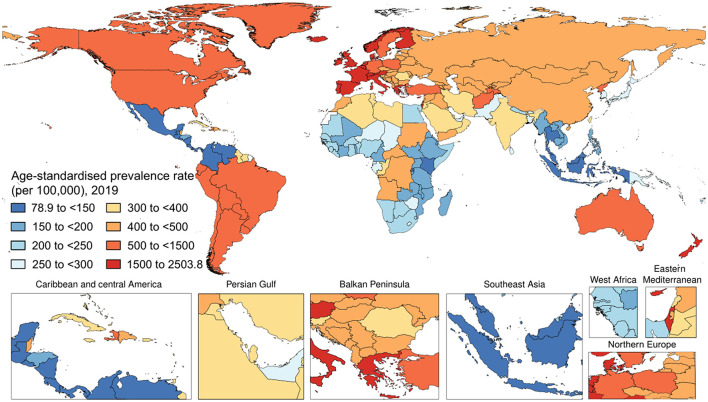
Figure 1. The national age-standardized prevalence rate of psoriasis (per 100,000) in 2019
Core Pathogenesis of Psoriasis: Immune Dysregulation Driving the Inflammatory Loop
The core mechanism of psoriasis lies in the abnormal activation of inflammatory signaling pathways triggered by immune dysregulation, ultimately forming a vicious cycle involving “immune cells—cytokines—keratinocytes.” Specifically, environmental triggers activate dermal dendritic cells and other antigen-presenting cells, stimulating the secretion of pro-inflammatory cytokines such as TNF-α, IL-12, and IL-23. These cytokines then induce the differentiation and proliferation of CD4+ T cells into Th17 cells, which secrete critical effector cytokines including IL-17A, IL-17F, and IL-22.
Among these, IL-17A is the key molecule driving lesion formation. It acts directly on keratinocytes, triggering abnormal proliferation and defective differentiation, while also prompting the release of additional inflammatory mediators and chemokines that recruit neutrophils and other inflammatory cells to lesion sites. Furthermore, IL-17A activates the STAT3 signaling pathway, upregulating keratin 17 expression and further amplifying local inflammation. Non-immune cells—such as fibroblasts and endothelial cells—also participate in shaping the inflammatory microenvironment, interacting with immune cells to sustain the inflammatory amplification cycle and leading to chronic inflammation and tissue remodeling at lesion sites.
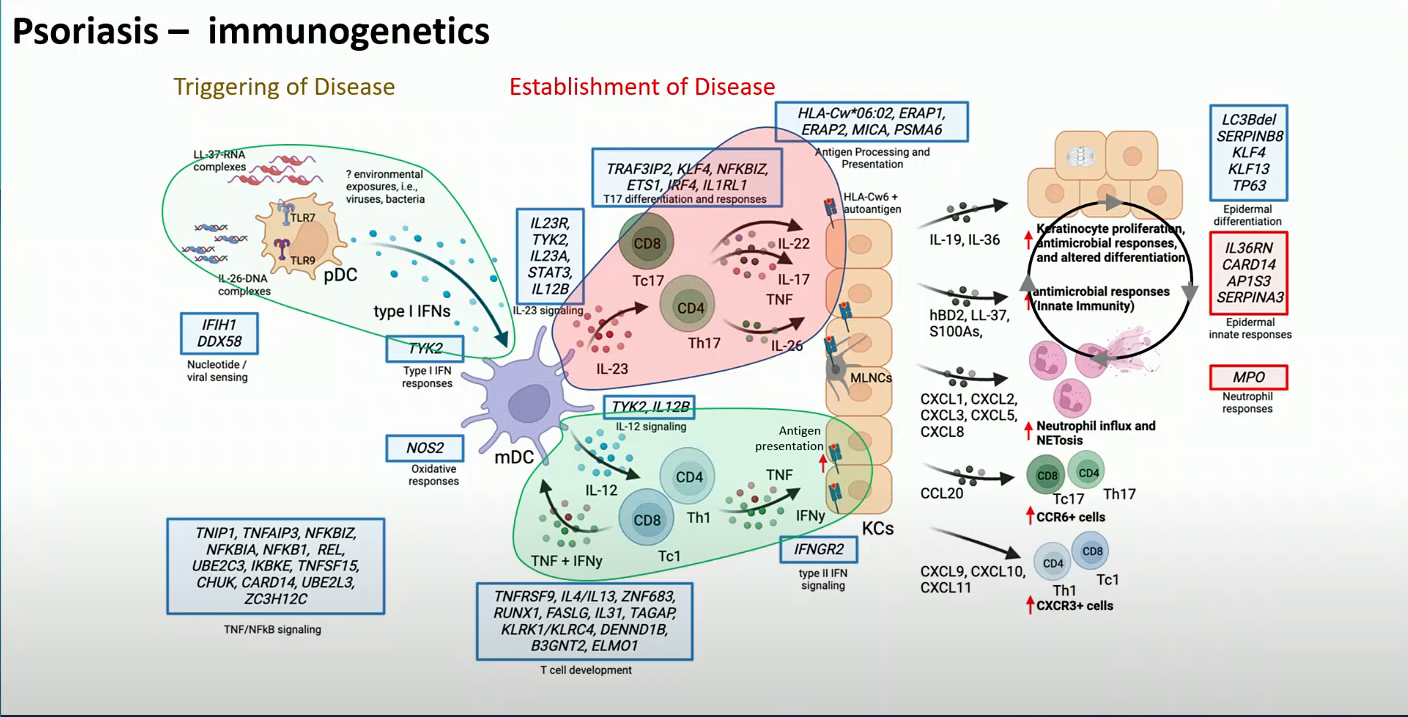
Figure 2: Cytokine-mediated mechanisms of psoriasis
Targeted Therapeutic Key Targets in Psoriasis
Cytokine-Related Core Targets
- TNF-α: Serving as an upstream hub for pro-inflammatory cytokines, TNF-α amplifies inflammatory signals and boosts immune responses. Monoclonal antibodies like adalimumab and infliximab are widely used in clinical practice.
- IL-17: A pivotal effector cytokine in lesion development, IL-17 is targeted by monoclonal antibodies (e.g., secukinumab, ixekizumab) and dual-target IL-17A/F monoclonal antibodies—key therapeutic options for moderate-to-severe psoriasis.
- IL-12/IL-23: These cytokines regulate Th1/Th17 cell differentiation via their shared p40 subunit. Monoclonal antibodies targeting IL-12/IL-23 (e.g., ustekinumab, guselkumab) suppress immune dysregulation at its source.
- IL-22/IL-36: IL-22 collaborates with IL-17 to drive abnormal keratinocyte proliferation and sustain chronic lesion inflammation, with related monoclonal antibodies still in clinical development. IL-36 plays a central role in generalized pustular psoriasis, and IL-36 receptor-targeting monoclonal antibodies (e.g., spesolimab) have gained clinical approval.
JAK Family Targets
- JAK: Mediates downstream signaling for multiple pro-inflammatory cytokines. Small-molecule inhibitors like tofacitinib are clinically approved, with ongoing research focusing on high-selectivity JAK subtype inhibitors to enhance treatment safety.
- JAK2: Plays a vital role in immune responses. JAK2 inhibitors such as ruxolitinib show significant potential for inflammatory regulation.
- JAK3: A key activator of immune cells. The development of highly selective JAK3 inhibitors holds promise for treating immune-related diseases.
- TYK2: Primarily mediates IL-12 and IL-23 signaling. Highly selective TYK2 inhibitors are now available, offering precise anti-inflammatory effects with the convenience of oral administration.
Downstream Transcriptional Regulation Targets
- STAT3: A core transcription factor downstream of pro-inflammatory cytokines like IL-17, directly regulating inflammatory gene expression and abnormal keratinocyte proliferation. Research is focused on developing specific STAT3 inhibitors to block the final amplification of inflammation, with related small-molecule drugs in preclinical stages.
- HIF1α: A downstream target of IL-17A, HIF1α reinforces the inflammatory loop by regulating metabolic reprogramming. Specific HIF1α inhibitors have demonstrated anti-inflammatory effects in vitro, with potential clinical applications.
Regulation of Inflammatory Cell Activation
- PDE4: Enhances inflammatory cell activity by degrading cAMP. PDE4 inhibitors like apremilast increase cAMP levels and broadly suppress pro-inflammatory cytokine secretion, making them a common treatment for mild-to-moderate psoriasis.
- PDE7: Functions similarly to PDE4. Studies indicate that combining PDE7 inhibitors with PDE4 inhibitors enhances anti-inflammatory efficacy while reducing toxicity, though these agents remain in preclinical research.
Multi-Pathway Regulatory Targets
- IRAK4: Located at the core of the IL-1R/TLR pathway, IRAK4 integrates multiple inflammatory signaling cascades. Research is focused on developing inhibitors that simultaneously suppress its dual functions, with these drugs still in preclinical development.
- TAK1: Integrates several signaling pathways, including those mediated by IL-1 and TNF-α. TAK1 inhibitors have shown efficacy in animal models by reducing lesion inflammation, with clinical translation research underway.
abinScience Psoriasis Research Tools: Empowering Precise Scientific Breakthroughs
1. Research Biosimilars
| Target |
Catalog No. |
Product name |
| IL-17A |
HS856026 |
Research Grade Secukinumab |
| IL-17RA |
HV263016 |
Research Grade Brodalumab |
| IL-17A IL-17F |
HS856036 |
Research Grade Bimekizumab |
| IL-23p19 |
HV466036 |
Research Grade Guselkumab |
| TNF-α |
HF879296 |
Research Grade Etanercept |
| IL-12/IL-23 |
HB769016 |
Research Grade Ustekinumab |
| IL-4/IL-13 |
HB732016 |
Research Grade Dupilumab |
View More Psoriasis Research Biosimilars
2. Antibodies
| Catalog No. |
Product name |
| AV466013 |
Anti-Guselkumab Non-Neutralizing Antibody |
| AF879013 |
Anti-Etanercept Non-Neutralizing Antibody |
| HF879010 |
InVivoMAb Anti-Human TNFa/TNF-alpha (Iv0050) |
| HS856010 |
InVivoMAb Anti-Human IL17A (Iv0029) |
| HB829014 |
Anti-JAK1 Polyclonal Antibody |
| HC515014 |
Anti-Human PDE4C Polyclonal Antibody |
| HT250014 |
Anti-Human TRAF3IP2 Polyclonal Antibody |
| HV466010 |
InVivoMAb Anti-Human IL23A (Iv0034) |
| HV466013 |
Anti-Human IL23A/IL-23p19 Nanobody (SAA1160) |
View More Psoriasis Antibodies
3. Assay Kits
View More Psoriasis Assay Kits
abinScience offers rigorously validated, high-quality research tools for psoriasis—including antibodies, ELISA kits, and Research Biosimilars—covering core psoriasis-related targets.. Facilitate the Advancement of Psoriasis Research
References
[1] Damiani G, Bragazzi NL, Karimkhani Aksut C, Wu D, Alicandro G, McGonagle D, Guo C, Dellavalle R, Grada A, Wong P, La Vecchia C, Tam LS, Cooper KD, Naghavi M. The Global, Regional, and National Burden of Psoriasis: Results and Insights From the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021 Dec 16;8:743180. doi: 10.3389/fmed.2021.743180. PMID: 34977058; PMCID: PMC8716585.
[2] Li D, Fan S, Song J, Zhao H, Guo L, Li P, Xu X. Global Psoriasis Burden 1990-2021: Evolving Patterns and Socio-Demographic Correlates in the Global Burden of Disease 2021 Update. Healthcare (Basel). 2025 Sep 26;13(19):2437. doi: 10.3390/healthcare13192437. PMID: 41095525; PMCID: PMC12524270.
[3] Greb JE, Goldminz AM, Elder JT, Lebwohl MG, Gladman DD, Wu JJ, Mehta NN, Finlay AY, Gottlieb AB. Psoriasis. Nat Rev Dis Primers. 2016 Nov 24;2:16082. doi: 10.1038/nrdp.2016.82. PMID: 27883001.
[4] Yang Y, Zhou X, Wang W, Dai H. Glycobiology of psoriasis: A review. J Autoimmun. 2025 Feb;151:103361. doi: 10.1016/j.jaut.2025.103361. Epub 2025 Jan 13. PMID: 39808852.
[5] Grayson M. Psoriasis. Nature. 2012 Dec 20;492(7429):S49. doi: 10.1038/492S49a. PMID: 23254969.
[6] Crow JM. Psoriasis uncovered. Nature. 2012 Dec 20;492(7429):S50-1. doi: 10.1038/492S50a. PMID: 23254970.
[7] Boehncke WH, Schön MP. Psoriasis. Lancet. 2015 Sep 5;386(9997):983-94. doi: 10.1016/S0140-6736(14)61909-7. Epub 2015 May 27. PMID: 26025581.
[8] Griffiths CE, Barker JN. Pathogenesis and clinical features of psoriasis. Lancet. 2007 Jul 21;370(9583):263-271. doi: 10.1016/S0140-6736(07)61128-3. PMID: 17658397.
[9] Ji C, Wang H, Bao C, Zhang L, Ruan S, Zhang J, Gong T, Cheng B. Challenge of Nail Psoriasis: An Update Review. Clin Rev Allergy Immunol. 2021 Dec;61(3):377-402. doi: 10.1007/s12016-021-08896-9. Epub 2021 Sep 3. PMID: 34478047.
[10] Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020 Apr;182(4):840-848. doi: 10.1111/bjd.18245. Epub 2019 Oct 15. PMID: 31225638; PMCID: PMC7187293.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




