Ulcerative colitis (UC) is a chronic inflammatory bowel disease that predominantly affects young and middle-aged adults. Clinically, it is characterized by recurrent abdominal pain, diarrhea, and bloody or mucus-containing stools, leading to a substantial decline in quality of life. Importantly, UC is not merely the result of nonspecific intestinal inflammation. Instead, it reflects a sustained breakdown of immune regulation within the colonic mucosa, in which the immune system misinterprets luminal cues and initiates inappropriate immune responses. This dysregulation ultimately drives a self-perpetuating cycle of inflammation, epithelial barrier damage, and further immune activation.
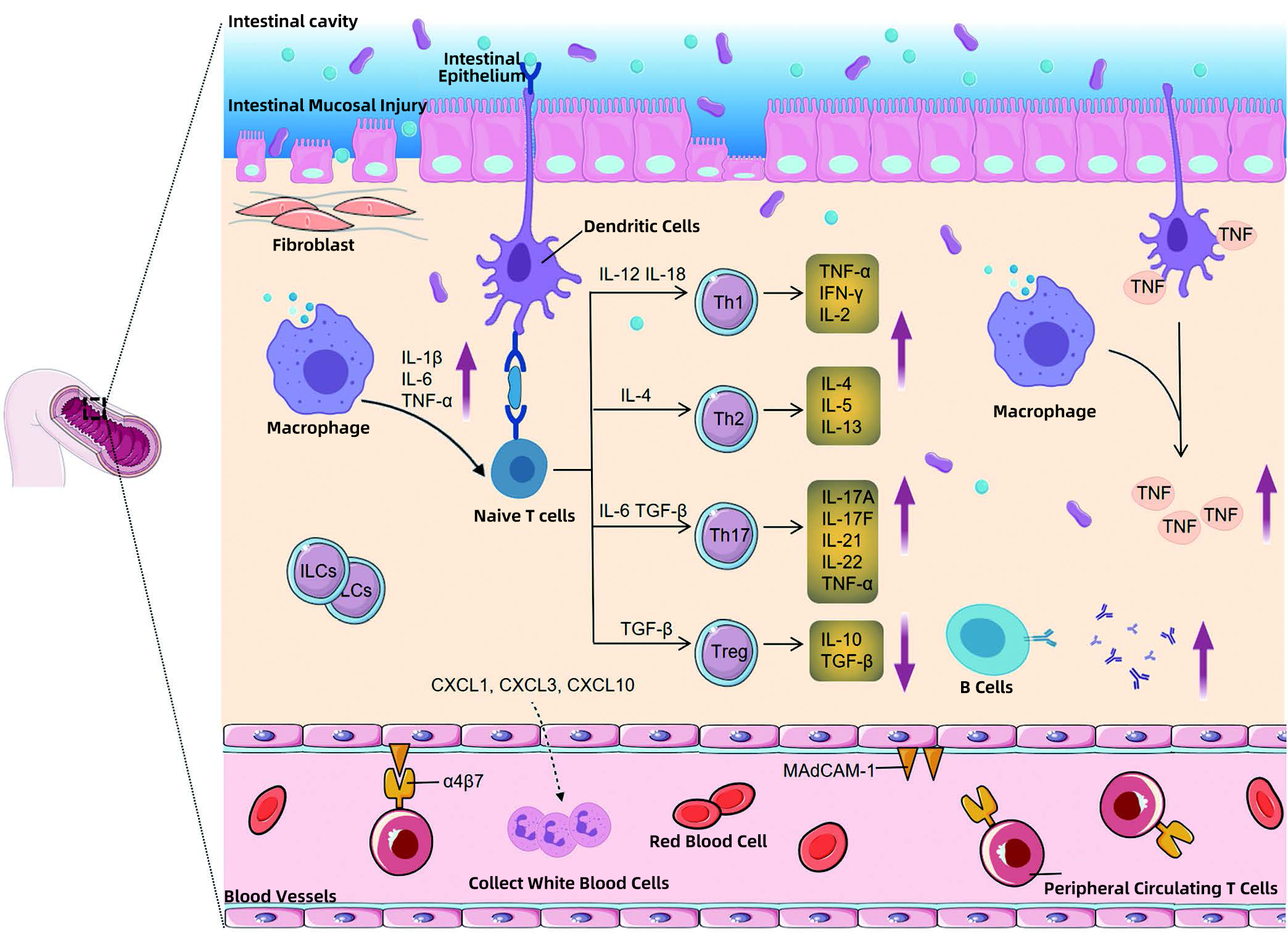
Figure 1. Immunological mechanisms underlying inflammatory bowel disease
1. A Shift in Research Perspective: From Detecting Inflammation to Understanding Its Persistence
Early UC research focused primarily on establishing the presence of inflammation, with elevated levels of classical inflammatory mediators such as TNF and IL-6 serving as key evidence of immune activation. While these findings firmly established the immunological nature of UC, current research has advanced beyond confirming the presence of inflammation to address a more fundamental question: why inflammation is so refractory to resolution.
This shift reflects a growing consensus that UC is not a transient inflammatory condition, but rather the consequence of long-term, dysregulated immune activation within the intestinal mucosa. Instead of merely confirming inflammatory activity, research efforts now aim to explain why subclinical inflammation persists during remission and why disease relapse is so common. This conceptual transition has laid the foundation for deeper mechanistic studies and more refined therapeutic strategies.
2. Three Core Mechanisms Driving Immune Imbalance
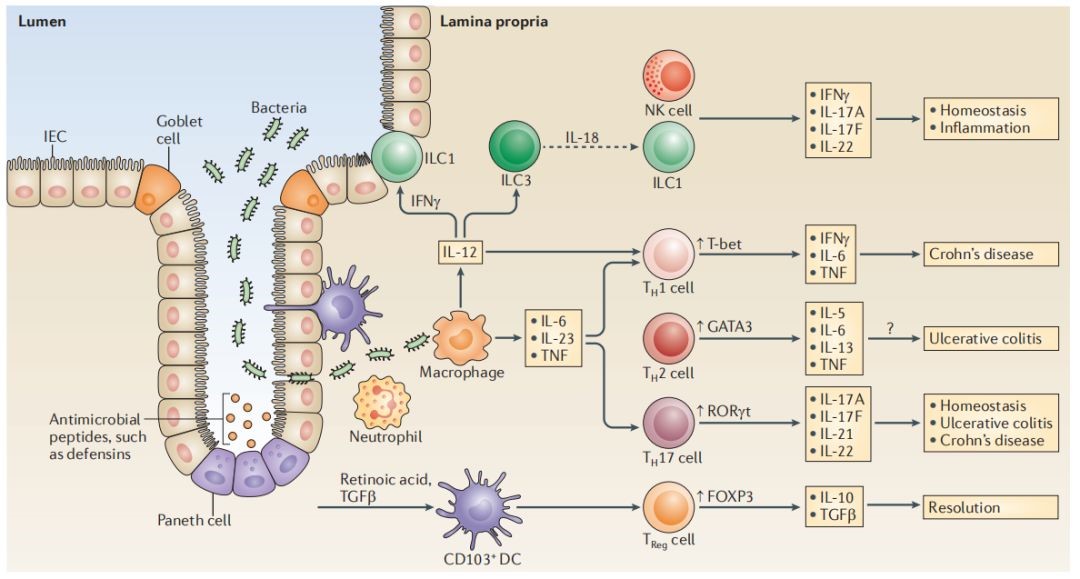
Figure 2. Cytokines and inflammatory pathways in IBD
3.1 The Th17/IL-23 Axis: A Sustained Driver of Chronic Inflammation
The Th17 cell lineage and its upstream regulator IL-23 form a central signaling axis that fuels chronic inflammation in UC. Extensive evidence demonstrates that IL-23 not only supports Th17 cell survival and expansion but also enhances their pro-inflammatory effector functions. This creates a self-reinforcing loop of activation, cytokine secretion, and reactivation, rendering the inflammatory response resistant to spontaneous resolution.
Recognition of this mechanism has reshaped therapeutic thinking, shifting the focus away from broad immunosuppression toward selective inhibition of specific inflammatory circuits. As a result, the Th17/IL-23 axis has emerged as a cornerstone of precision-targeted research in UC.
3.2 The TNF Pathway: An Amplifier of Inflammation and a Source of Heterogeneity
In UC, TNF functions not merely as a pro-inflammatory cytokine but as a central amplifier within the inflammatory cascade. By coordinating the activation of macrophages, T cells, and other immune populations, TNF intensifies and propagates inflammatory signaling. This explains the pronounced clinical efficacy of TNF-targeted interventions in a subset of patients.
At the same time, the variable responses observed across individuals have drawn greater attention to immunophenotypic heterogeneity. Research is now increasingly directed toward identifying dominant inflammatory pathways in distinct patient subgroups, providing critical insights into inter-individual differences in disease mechanisms.
3.3 The Intestinal Epithelial Barrier: From Secondary Factor to Initiating Event
Once considered a downstream consequence of inflammation, disruption of the intestinal epithelial barrier is now recognized as a pivotal contributor to immune imbalance. Barrier dysfunction exposes immune cells to excessive luminal antigens, perpetuating immune activation and fostering a vicious cycle of epithelial barrier impairment and inflammation.
This paradigm shift has elevated mucosal barrier integrity from a background consideration to a primary focus of UC research. Maintaining epithelial homeostasis is now viewed as a fundamental requirement for restoring immune balance and preventing disease recurrence.
3. Evolution of Research Paradigms: From Single Targets to Immune Network Integration
UC research has progressively moved away from the search for a single, decisive target toward a systems-level understanding of concerted immune pathways. Contemporary studies emphasize dynamic profiling of cytokine networks across disease stages, functional differentiation of immune cell subsets, and the influence of the inflammatory microenvironment on cellular fate decisions.
Correspondingly, research endpoints have expanded from simple measures of inflammation reduction to broader assessments of immune homeostasis restoration. Increasing attention is paid to longitudinal immune states and patient-specific variability, reflecting a more holistic view of disease biology.
Looking ahead, continued integration of immune pathways and temporal disease dynamics is expected to deepen mechanistic insight into UC. These efforts not only advance understanding of UC pathogenesis but also provide a conceptual framework applicable to a wide range of immune-mediated disorders, supporting ongoing progress in precision-targeted research.
4. abinScience Research Tools for Ulcerative Colitis
4.1 Research Biosimilar
| Catalog No. |
Product Name |
| HF879026 |
Research Grade Adalimumab |
| HB769016 |
Research Grade Ustekinumab |
| HS856026 |
Research Grade Secukinumab |
| HV466036 |
Research Grade Guselkumab |
| HB996036 |
Research Grade Tafasitamab |
View More Ulcerative Colitis Research Biosimilar
4.2 Antibodies
| Catalog No. |
Product Name |
| HF879010 |
InVivoMAb Anti-Human TNFa/TNF-alpha (Iv0050) |
| HB996407 |
Anti-Human CD19 Antibody (FMC63) |
| HT205107 |
Anti-Human CD284/TLR4 Antibody (SAA0095) |
| HS856014 |
Anti-IL17A Polyclonal Antibody |
| HV466013 |
Anti-Human IL23A/IL-23p19 Nanobody (SAA1160) |
| HF879023 |
Anti-Human TNFa/TNF-alpha Nanobody (SAA1146) |
| HS856010 |
InVivoMAb Anti-Human IL17A (Iv0029) |
| HV466010 |
InVivoMAb Anti-Human IL23A (Iv0034) |
View More Ulcerative Colitis Antibodies
4.3 Assay Kits
View More Ulcerative Colitis Assay Kits
abinScience focuses on core immunological targets implicated in ulcerative colitis and offers a rigorously validated portfolio of research tools. This comprehensive toolkit includes cytokine-specific antibodies, ELISA kits for biologic drug monitoring, and research-grade biosimilars, supporting studies across the Th17/IL-23 axis, TNF signaling, and mucosal barrier–related mechanisms. Together, these tools provide reliable, end-to-end support for UC mechanism exploration, target validation, and translational research.
References
[1] Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017 Apr 29;389(10080):1756-1770. doi: 10.1016/S0140-6736(16)32126-2. Epub 2016 Dec 1. PMID: 27914657; PMCID: PMC6487890.
[2] Ananthakrishnan AN, Murad MH, Scott FI, Agrawal M, Haydek JP, Limketkai BN, Loftus EV Jr, Singh S. Comparative Efficacy of Advanced Therapies for Management of Moderate-to-Severe Ulcerative Colitis: 2024 American Gastroenterological Association Evidence Synthesis. Gastroenterology. 2024 Dec;167(7):1460-1482. doi: 10.1053/j.gastro.2024.07.046. Epub 2024 Oct 18. PMID: 39425738; PMCID: PMC12148344.
[3] Kuwada T, Shiokawa M, Kodama Y, Ota S, Kakiuchi N, Nannya Y, Yamazaki H, Yoshida H, Nakamura T, Matsumoto S, Muramoto Y, Yamamoto S, Honzawa Y, Kuriyama K, Okamoto K, Hirano T, Okada H, Marui S, Sogabe Y, Morita T, Matsumori T, Mima A, Nishikawa Y, Ueda T, Matsumura K, Uza N, Chiba T, Seno H. Identification of an Anti-Integrin αvβ6 Autoantibody in Patients With Ulcerative Colitis. Gastroenterology. 2021 Jun;160(7):2383-2394.e21. doi: 10.1053/j.gastro.2021.02.019. Epub 2021 Feb 12. PMID: 33582126.
[4] Raine T, Verstockt B, De Cruz P. Immune therapies in ulcerative colitis: are we beyond anti-TNF yet? Lancet Gastroenterol Hepatol. 2020 Sep;5(9):794-796. doi: 10.1016/S2468-1253(20)30210-7. PMID: 32818453.
[5] Ling Fangmei, Li Junrong, Chen Yidong, Xu Mingyang, Zhu Liangru. Application advance of cytokine-derived biological agents in inflammatory bowel disease [J] . Chinese Journal of Inflammatory Bowel Disease, 2021, 05(4) : 342-346. DOI: 10.3760/cma.j.cn101480-20201104-00123.
[6] Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023 Aug 12;402(10401):571-584. doi: 10.1016/S0140-6736(23)00966-2. PMID: 37573077.
[7] Jiang Z, Wang Y, Gong J, Chen X, Hang D, Chen C, Hong X, Zhang J, Qiu K, Liao Y, Li P, Wang H, Yang Z, Qiu T, Zhou Y, Chen Z, Zhou H, Shan X, Zhou N, Liu L, Feng F, Su F, Ma H, Liu Z, He W, Fang L, Xuan J, Gan Z, Gao X, Zhang J, Chen H, Wang F, Zhang X, Zhu M. An Aeromonas variant that produces aerolysin promotes susceptibility to ulcerative colitis. Science. 2025 Nov 20;390(6775):eadz4712. doi: 10.1126/science.adz4712. Epub 2025 Nov 20. PMID: 41264716.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




