As a pivotal tool for cell analysis, flow cytometry plays an indispensable role in biological research. However, for researchers new to this technology, the terminology and concepts involved can be perplexing. This article highlights common flow cytometry terms to help you quickly grasp core concepts, enhancing the accuracy of experimental operations and data analysis.
I. Instrument Setup
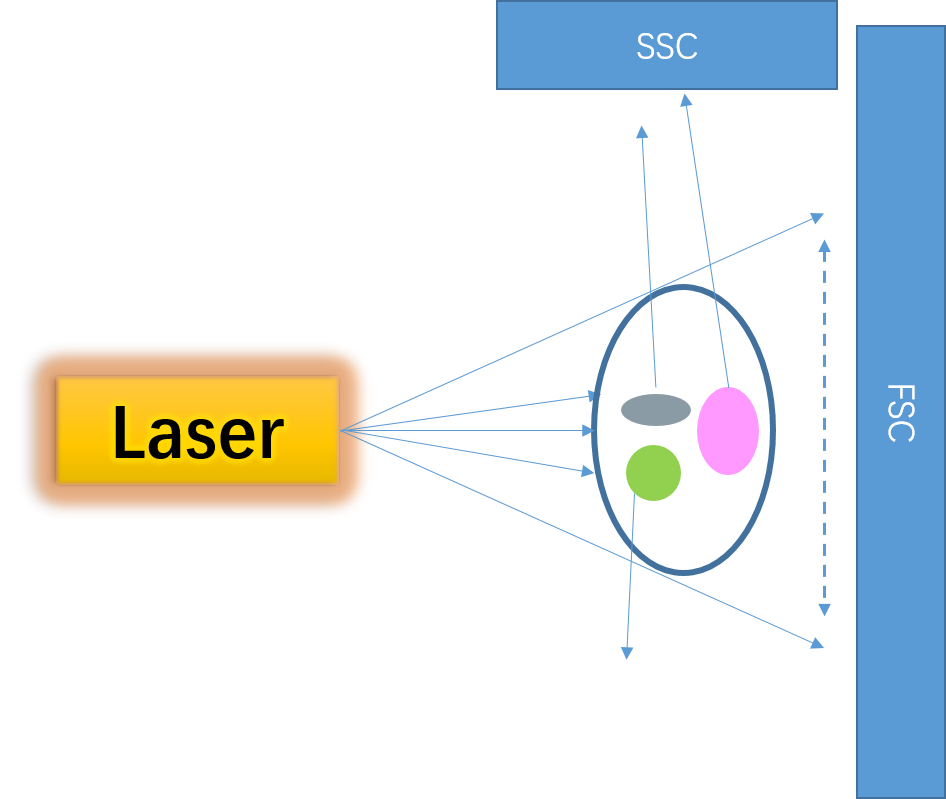
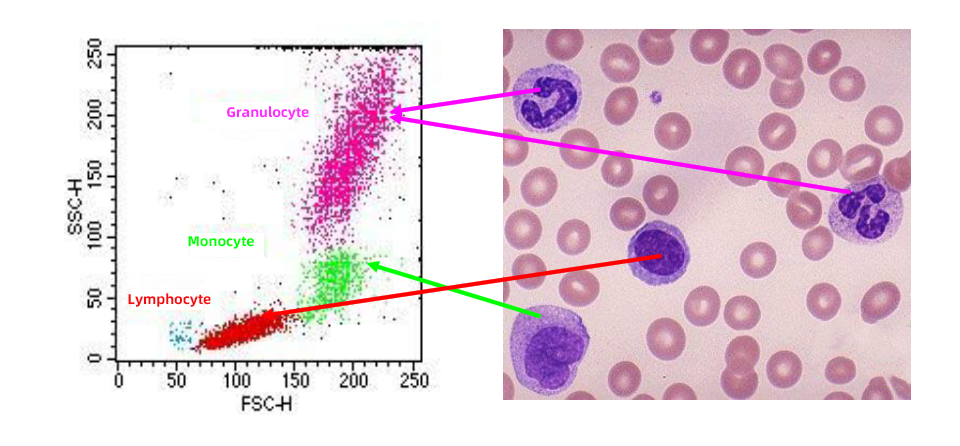
1. FSC/SSC:
The most fundamental parameters detected by flow cytometers, reflecting the inherent physical characteristics of cells.
FSC (Forward Scatter): Indicates cell size, with signal intensity correlating to cell volume.
SSC (Side Scatter): Reflects intracellular complexity (e.g., the complexity of granules, organelles, and nuclear morphology), where signal intensity is proportional to the intricacy of internal cellular structures.
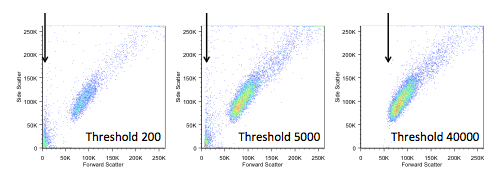
2. Threshold:
The minimum signal intensity standard set for specific detection parameters (e.g., FSC or fluorescence channels). The instrument only collects and analyzes events with signal intensity exceeding this threshold, effectively eliminating background noise and interference from non-target particles to ensure the accuracy and reliability of subsequent analyses.
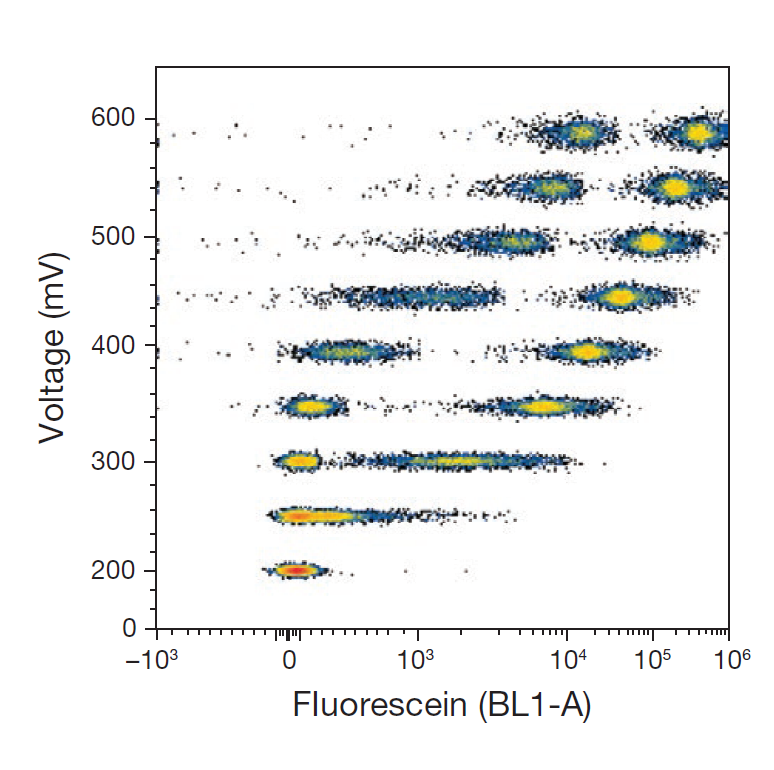
3. Channel Voltage:
A critical parameter regulating the signal intensity of detectors on flow cytometers. It determines the instrument’s sensitivity to fluorescent signals. As illustrated below:
- If the voltage is set too low (e.g., 200 mV), the signal may be excessively weak, making it impossible to distinguish between unlabeled and fluorescently labeled cells.
- If the voltage is set too high (e.g., 600 mV), the signal may exceed the instrument’s detection range, leading to inaccurate measurements (a phenomenon known as "signal clipping" in flow analysis).
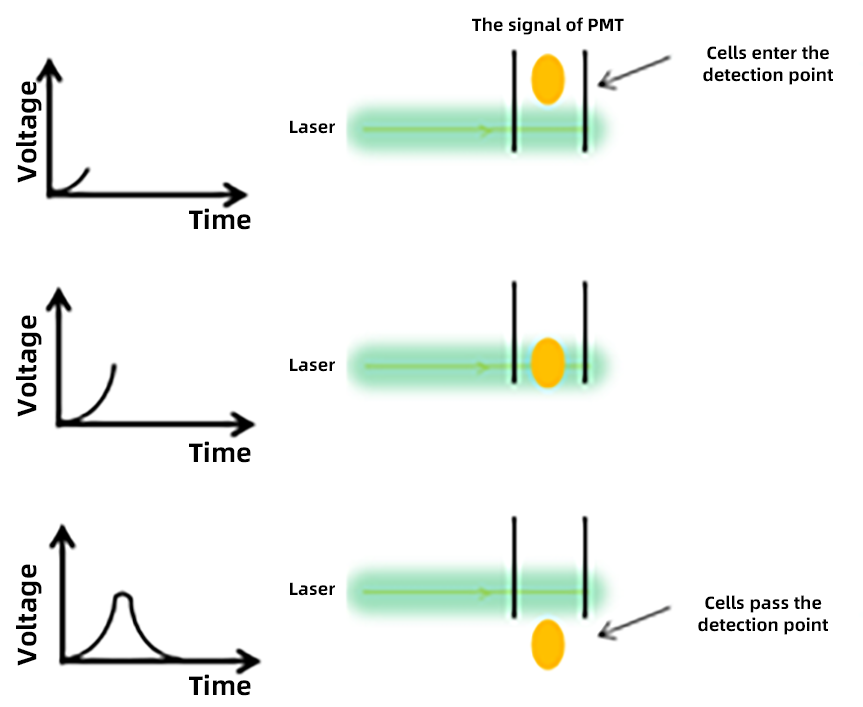
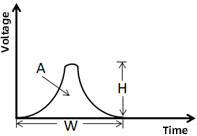
4. A/H/W:
When a cell passes through the laser, the flow cytometer detects an optical signal, which is amplified by a Photomultiplier Tube (PMT) and converted into an electrical pulse. From the cell’s entry into the detection zone to its exit, the electrical pulse rises from 0 to a peak and then returns to 0, recorded as a pulse signal:
✦ H (height): The peak value of the electrical pulse signal.
✦ W (width): The duration of the cell’s passage through the laser detection zone.
✦ A (area): Calculated using the values of W and H, representing the area of the electrical pulse signal.
Applications of A, H, and W Signals
Exclude aggregates: Typically, two of these three signals (e.g., FSC-A/H or FSC-A/W) are used to exclude aggregates during data analysis. Aggregates traverse the detection zone for a longer duration, resulting in larger pulse areas.
II. Data Analysis
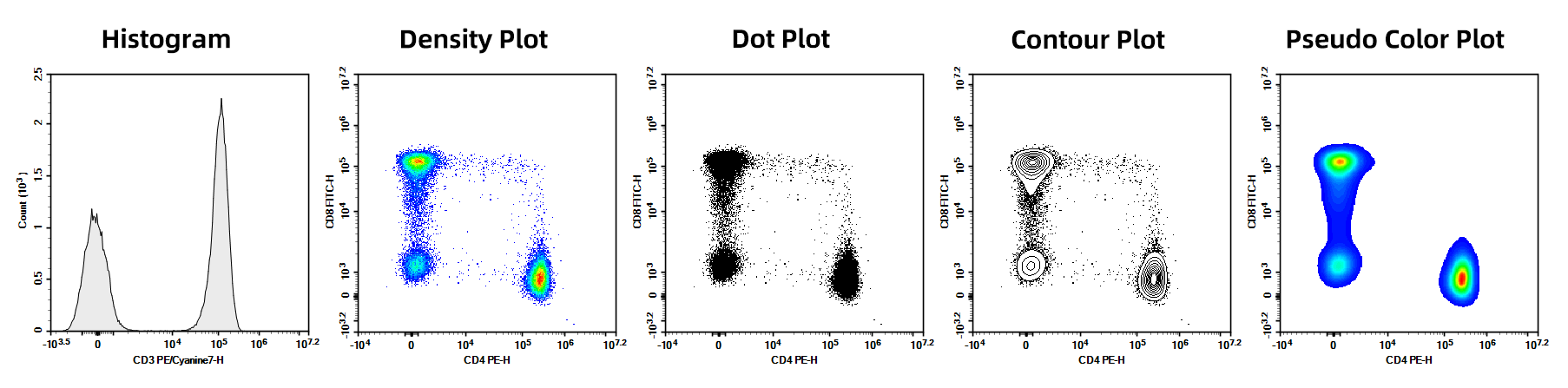
1. Parameter Plots
Different formats for presenting flow cytometry results, including uniparametric plots, biparametric plots, histograms, dot plots, density plots, contour plots, pseudo color plots,etc.
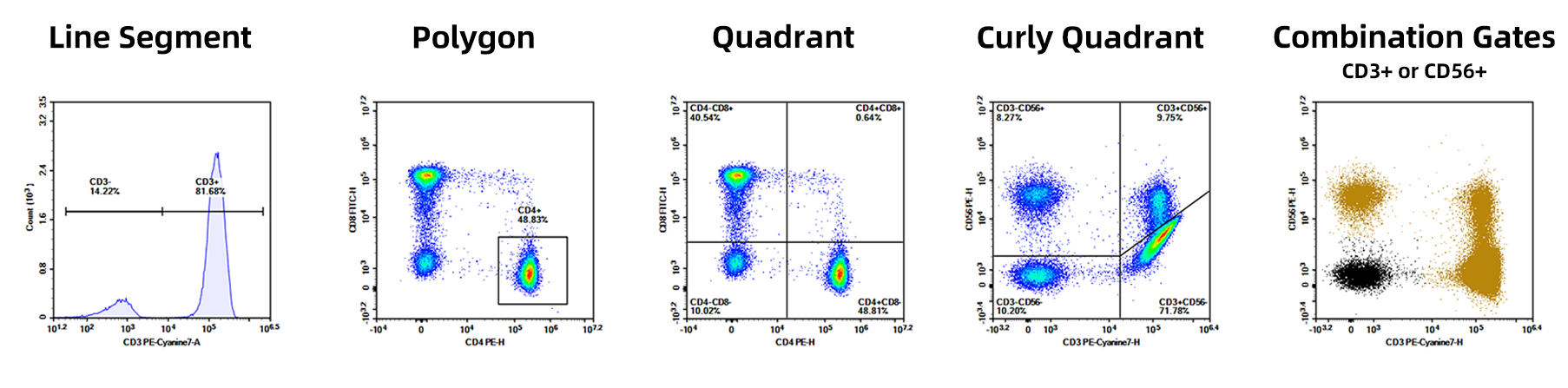
2. Gating
A method for selecting or marking specific cell populations in flow cytometry. It involves the cells of interest from a heterogeneous cell population while excluding irrelevant cells. By adjusting this "gate," researchers can accurately analyze the characteristics of target cells. Common gating types include: Line Segment, Polygon, Quadrant, Curly Quadrant, Combination Gates
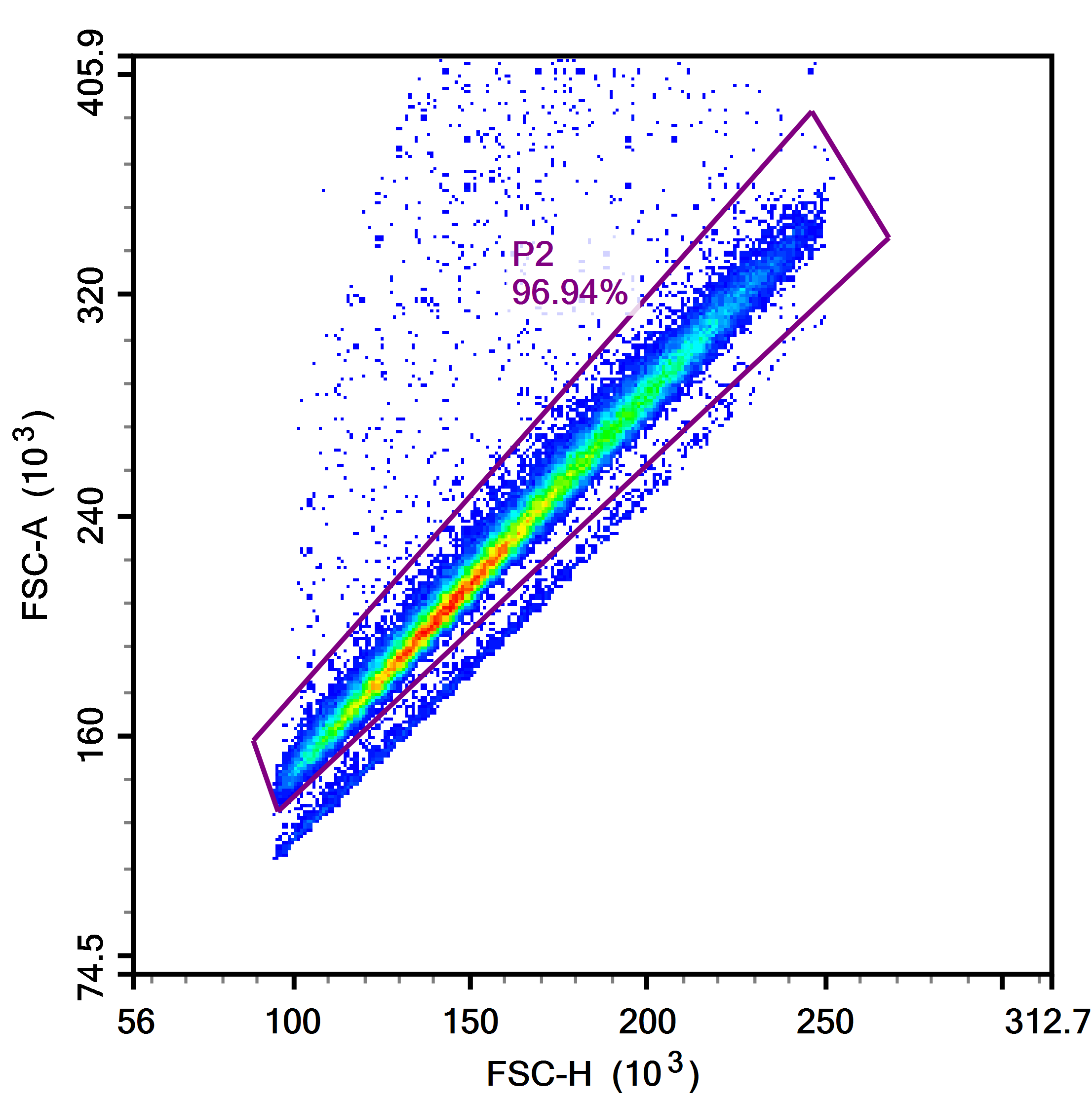
3. Exclude aggregates
Aggregates refer to two or more cells adhering to each other. They can compromise the accuracy of experimental data and thus need to be excluded during analysis. This is generally achieved using two of the A/H/W signals (e.g., FSC-A/H).
4. Compensation
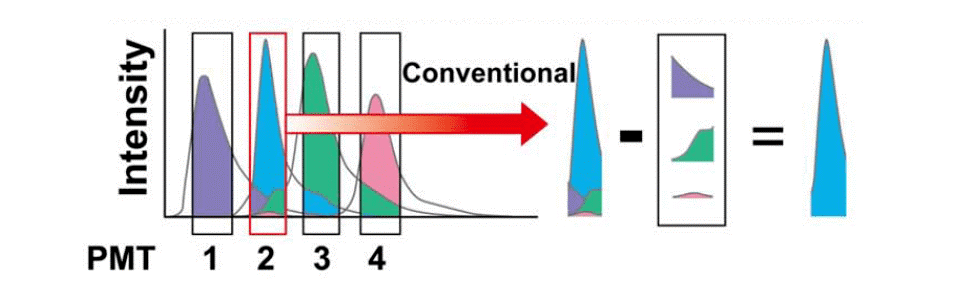
Fluorochromes often have overlapping emission wavelength ranges. Flow cytometers collect signals within specific wavelength bands (e.g., the FITC channel captures signals between 515–535 nm), inevitably collecting signals from multiple fluorochromes. Compensation is the process of correcting for spectral overlap during data analysis, isolating target signals from interfering ones to obtain accurate multicolor flow data.
III. Other Key Terminology
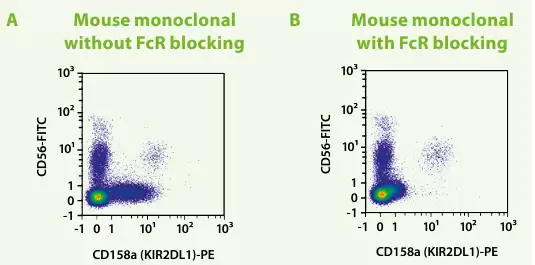
1. Fc Receptor Blocking Reagent
Used to prevent antibodies from binding to Fc receptors on cell surfaces, avoiding non-specific staining. It effectively reduces background staining caused by Fc receptor-mediated interactions, ensuring the accuracy of experimental results.
2. Fixation and Permeabilization
A common cell processing method in flow cytometry, involving preserving cell structure and permeabilizing cell membranes to facilitate staining:
• Fixation: Maintains the integrity of cell surface markers.
• Permeabilization: Renders cell membranes permeable, enabling staining of intracellular antigens.
Note: Two types of fixation/permeabilization reagents are available: mild formulations for cytoplasmic targets (e.g., cytokines) and harsher formulations for nuclear targets (e.g., transcription factors).
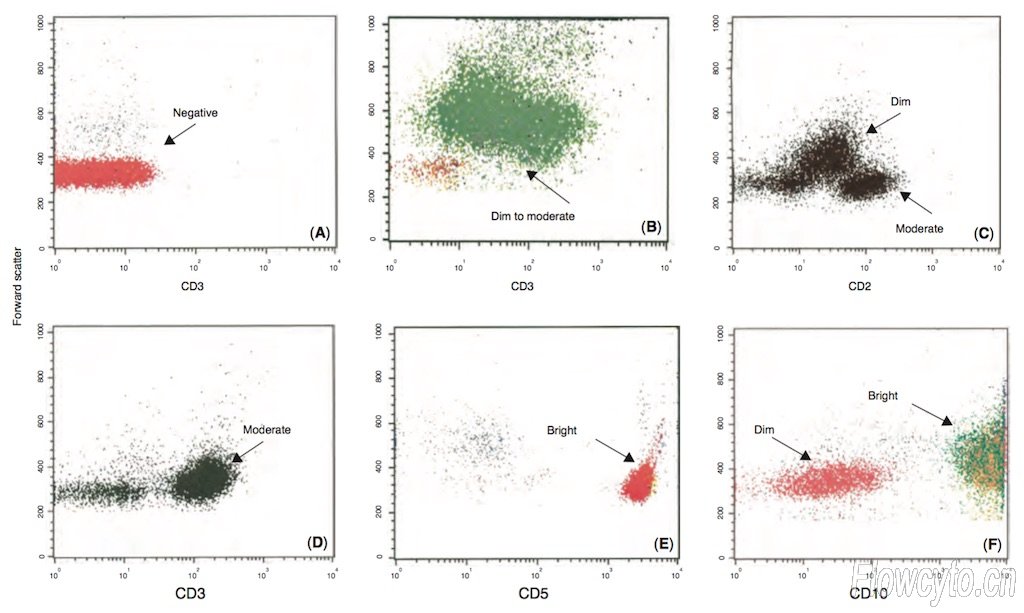
3. Fluorescence Intensity Grading Standards
Terms describing the strength of cellular fluorescent signals, including:
Negative (no signal), dim (weak signal), low (low intensity), moderate (moderate intensity), high (high intensity), bright (strong signal), positive (signal present).
They are also commonly denoted as -/+/++/+++, helping researchers accurately distinguish cell populations with different signal intensities.
4. Lin (Lineage/Lineage Cocktail)
Not a single marker but a panel of markers used to exclude specific cell lineages. For example, when detecting dendritic cells (DCs), Lineage markers (CD3/CD19/CD20/CD14) are used to exclude non-target cells such as T cells and B cells.
5. Autofluorescence/Background/Non-specific Staining
✦ Autofluorescence: Inherent fluorescence emitted by cells themselves (e.g., from NADH, riboflavin, flavin coenzymes, or cross-linking of primary amines in paraformaldehyde).
✦ Background: All non-target fluorescent signals in an experiment.
✦ Non-specific Staining: Signals generated by antibodies or dyes binding to non-target structures on cell surfaces.
Understanding the basic terminology of flow cytometry is the first step toward conducting effective experiments. With a solid grasp of these fundamentals, you will navigate data acquisition, analysis, and result interpretation with greater confidence and precision, ultimately enhancing the quality and reliability of your experiments.
Looking for Reliable Flow Cytometry Antibodies to Support your Experimental Workflows?
abinScience Flow Cytometry Antibodies are subjected to rigorous quality control to ensure consistent fluorescent performance and clear discrimination of cell populations, supporting robust and reproducible flow cytometry analyses.
Learn More about abinScience Flow Cytometry Antibodies
About Us
abinScience was founded in 2023 as a strategic venture of AtaGenix (established 2011), dedicated to delivering premium life science reagents that accelerate discovery.
abinScience flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species(Human, Mouse, Rat, Dog, Hamster, Monkey, etc.)We provide stable and reliable support for scientific research.


 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский