Epstein-Barr Virus: A Latent Pathogenic Threat and Global Health Challenge
Epstein-Barr Virus (EBV), a widely prevalent γ-herpesvirus, causes a broad range of health issues, from infection-related disorders to malignancies, posing a life-threatening risk, especially to immunocompromised individuals. As the first identified human oncogenic virus, EBV is responsible for approximately 358,000 new cancer cases and 209,000 related deaths globally each year, with etiological links to a diverse range of malignancies including nasopharyngeal carcinoma, post-transplant lymphoproliferative disorder (PTLD), and Burkitt’s lymphoma. Notably, PTLD, a fatal EBV-driven lymphoma, has a markedly elevated incidence in patients undergoing immunosuppressive therapy following solid organ or hematopoietic stem cell transplantation, representing one of the most severe post-transplant complications.
Beyond oncology, EBV infection is also strongly associated with a host of immune and inflammatory conditions, including infectious mononucleosis, multiple sclerosis, systemic lupus erythematosus, and rheumatoid arthritis. It can also trigger complications in patients with COVID-19, exacerbating disease severity. Following primary infection, EBV establishes lifelong latent infection in human memory B cells, with the majority of infected individuals remaining asymptomatic carriers for life. However, when immune surveillance is compromised, the latent virus can undergo reactivation, leading to uncontrolled viral replication and the onset of associated pathologies. Most critically, there are currently no globally approved EBV-specific vaccines or therapeutic agents. Clinical management relies on non-specific antiviral drugs and immunomodulatory strategies, which have limited efficacy and fail to specifically block the progression of EBV infection.
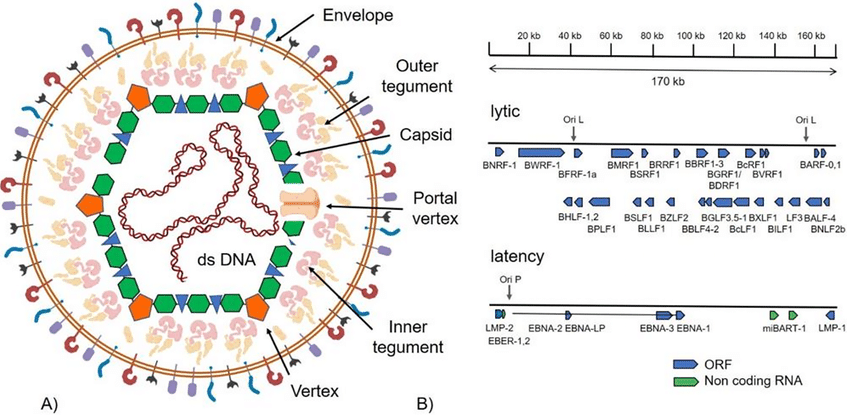
Fig. 1 Structure of the EBV
Scientific Breakthrough: Advances in Fully Human Monoclonal Antibodies Targeting EBV
In a study published in Cell Reports Medicine entitled Transgenic mouse-derived human monoclonal antibodies targeting EBV gp350 and gp42 provide basis for therapeutic development researchers achieved a pivotal technological breakthrough using a transgenic mouse platform to address the critical clinical gap of EBV-specific therapies, delivering a novel paradigm for targeted EBV treatment.
Using ATX-GK transgenic mice as a specialized immunization platform, the research team performed combined immunization with recombinant EBV envelope glycoproteins gp350 and gp42. Coupled with hybridoma technology and high-throughput screening workflows, this approach successfully yielded 2 fully human monoclonal antibody clones targeting gp350 and 8 clones targeting gp42, designated as the ATX-350 and ATX-42 series, respectively. Compared to conventional murine-derived antibodies, these fully human antibodies eliminate the need for post-generation humanization modification, inherently mitigating clinical risks such as the development of anti-drug antibodies (ADAs) and hypersensitivity reactions commonly associated with murine antibodies, thereby significantly enhancing their clinical translatability.
Both structural biology and functional validation assays confirmed that these two classes of fully human antibodies exert potent neutralizing activity by precisely blocking the key pathways of EBV entry into host cells. The ATX-350 series specifically binds to the gp350 protein, potently inhibiting its interaction with CD21/CD35 receptors on the surface of host B cells. Meanwhile, the ATX-42 series targets and binds to the gp42 protein, blocking its engagement with HLA class II molecules. In this manner, the antibodies directly interrupt the EBV infection cascade at two core stages: viral attachment and membrane fusion.
In vitro assays demonstrated that all of these fully human antibodies potently neutralize EBV infection of B cells, with neutralizing activity comparable to that of the established gold-standard antibodies, highlighting robust in vitro antiviral efficacy. The in vivo protective efficacy of the antibodies was validated in a humanized mouse model of EBV challenge. Following a single prophylactic dose, gp42-targeting antibodies conferred complete protection against EBV infection in mice, preventing the development of splenomegaly and viremia, with no EBV DNA detected in the spleens of treated animals. Gp350-targeting antibodies effectively suppressed viremia and also exerted a significant protective effect against splenomegaly.
In parallel, pharmacokinetic studies revealed that the fully human antibodies developed in this study exhibit slow in vivo clearance and an extended half-life, endowing them with long-acting protective biological properties that are highly aligned with the practical requirements of clinical prophylactic intervention. Furthermore, using X-ray crystallography and negative-stain electron microscopy, the research team mapped the precise binding epitopes of the antibodies to gp350 and gp42, and identified broad-spectrum vulnerable sites on the EBV virion. These findings provide a critical structural framework for the rational design and development of next-generation EBV-related vaccines.
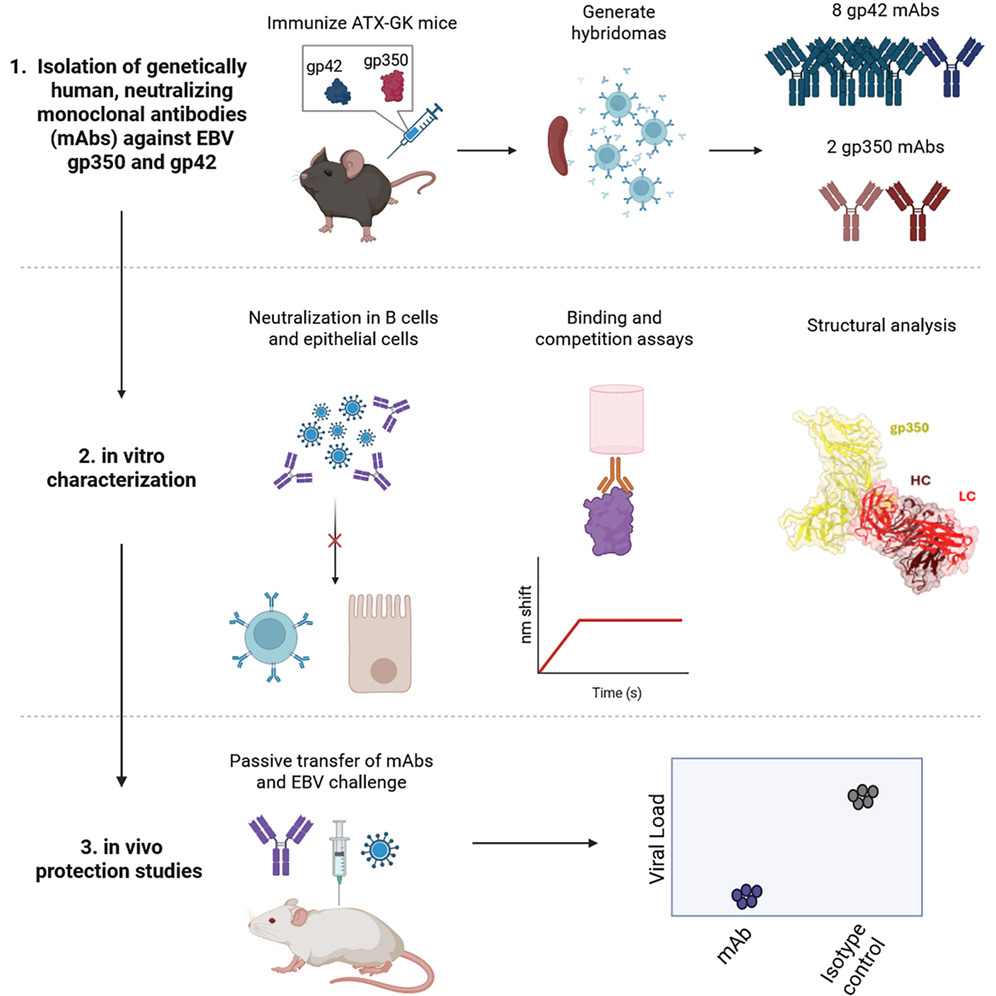
Fig. 2 Graphical abstract
Forward Outlook: The Future Potential of Fully Human Monoclonal Antibodies in EBV Prevention and Treatment
This groundbreaking advance holds dual significance for clinical translation and fundamental scientific research. Clinically, the successfully developed fully human neutralizing monoclonal antibodies fill a long-standing gap in EBV-specific therapeutics, offering an innovative strategy for PTLD prophylaxis in high-risk populations such as transplant recipients. Their fully human nature circumvents the immunogenicity risks associated with conventional antibodies, drastically shortening the clinical translation timeline. Technologically, this work validates the high efficiency of the ATX-GK transgenic mouse platform for the development of fully human EBV antibodies. The established end-to-end technical workflow of "immunization–screening–structural characterization–in vivo validation" provides a directly translatable paradigm for the development of targeted antibodies against other herpesviruses. Scientifically, the identification of vulnerable sites critical for EBV invasion via structural biology analysis is driving the evolution of EBV vaccine development from broad-spectrum immunization to precision immune intervention, providing well-defined targets for the rational design of multi-epitope vaccines.
Future research will advance the translation of these findings along three core directions:
- Accelerate clinical translation by initiating human clinical trials to systematically evaluate the safety and efficacy of the monoclonal antibodies in both healthy and high-risk populations, define the optimal dosing regimen, and advance the regulatory approval of antibody therapeutics for the prevention and treatment of EBV infection.
- Expand therapeutic applications by exploring the therapeutic potential of the antibodies in EBV-associated malignancies such as nasopharyngeal carcinoma, evaluating the synergistic efficacy of monotherapy or combination regimens with chemotherapy and immunotherapy, and broadening their clinical utility.
- Optimize therapeutic strategies by developing multi-epitope combination vaccines based on the vulnerable sites identified by the antibodies, while exploring combination regimens of antibodies targeting distinct epitopes to overcome potential viral escape associated with single-antibody therapy and enhance therapeutic durability.
In addition, further enhancement of antibody affinity and in vivo half-life via antibody engineering, along with the development of convenient administration routes such as subcutaneous injection, will further improve clinical usability and patient compliance, delivering a more comprehensive solution for the precise prevention and treatment of EBV infection-related diseases.
Recommended Research Tools: Latest EBV Products from abinScience
1. Proteins
| Catalog No. |
Product Name |
| VK622011 |
Recombinant EBV/HHV4 BLLF1/MA/GP350 Protein, C-His |
| VK598021 |
Recombinant EBV/HHV4 gH & gL Complex Protein, C-Strep & C-His |
| VK491011 |
Recombinant EBV/HHV4 GP42/Glycoprotein 42 Protein, C-His |
| VK074012 |
Recombinant EBV/HHV4 BMRF1 Protein, N-His |
| VK598012 |
Recombinant EBV/HHV4 gH/Envelope glycoprotein H Protein, N-His |
| VK491012 |
Recombinant EBV/HHV4 gp42 Protein, N-His |
View more EBV-related proteins
2. Antibodies
| Catalog No. |
Product Name |
| VK491013 |
Anti-EBV/HHV-4 GP42/Glycoprotein 42 Antibody (SAA2098) |
| VK622014 |
Anti-EBV/HHV4 BLLF1/MA/GP350 Polyclonal Antibody |
| VK799010 |
InVivoMAb Anti-EBV/HHV4 gB/BALF4 Antibody (Iv0133) |
| VK799020 |
InVivoMAb Anti-EBV/HHV4 gB/BALF4 Antibody (Iv0134) |
| VK598013 |
Anti-EBV/HHV-4 gH/BXLF2 Antibody (CL40) |
| VK598033 |
Anti-EBV/HHV-4 gH & gL Complexes Antibody (769B10) |
| VK799033 |
Anti-EBV/HHV4 gB/BALF4 Antibody (AMMO5) |
View more EBV-related antibodies
abinScience offers a comprehensive portfolio of EBV-focused research products, including high-quality recombinant proteins and fully human antibodies, which are widely used in fundamental EBV research and vaccine development.
References
[1] Zavala-Vega S, Palma-Lara I, Ortega-Soto E, Trejo-Solis C, de Arellano IT, Ucharima-Corona LE, Garcia-Chacón G, Ochoa SA, Xicohtencatl-Cortes J, Cruz-Córdova A, Luna-Pineda VM, Jiménez-Hernández E, Vázquez-Meraz E, Mejía-Aranguré JM, Guzmán-Bucio S, Rembao-Bojorquez D, Sánchez-Gómez C, Salazar-Garcia M, Arellano-Galindo J. Role of Epstein-Barr Virus in Glioblastoma. Crit Rev Oncog. 2019;24(4):307-338. doi: 10.1615/CritRevOncog.2019032655. PMID: 32421988.
[2] Chhan CB, Lang K, Davis AR, Wan YH, Aldridge NT, Kher G, Scharffenberger SC, Hardy SR, Iureniev R, Giltiay NV, Edwards KR, Radtke S, Kiem HP, Pancera M, McGuire AT. Transgenic mouse-derived human monoclonal antibodies targeting EBV gp350 and gp42 provide basis for therapeutic development. Cell Rep Med. 2026 Feb 17;7(2):102618. doi: 10.1016/j.xcrm.2026.102618. PMID: 41707657; PMCID: PMC12923964.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




