In 2025, multiple foodborne outbreaks caused by Listeria monocytogenes (Lm) were reported across the United States, once again placing this pathogen under intense public health scrutiny. In February 2026, the U.S. Food and Drug Administration (FDA) issued a Class I recall for approximately 56,000 pounds of frozen blueberries produced by an Oregon-based company due to potential Lm contamination. The implicated bulk industrial products had been distributed to Michigan, Oregon, Washington, Wisconsin, and parts of Canada.
As the only major foodborne intracellular pathogen capable of sustained proliferation at 4°C under standard refrigeration, L. monocytogenes remains a primary biological hazard in cold-chain food systems worldwide. Beyond food safety, it is also a canonical model organism in microbiology and infection immunology for dissecting intracellular parasitism and host–pathogen interactions.
I. Pathobiology, Transmission, and Epidemiological Features of Listeria monocytogenes
L. monocytogenes is a Gram-positive, facultative anaerobic rod and the most clinically relevant zoonotic species within the genus Listeria. It is non-spore-forming, lacks a capsule, and exhibits temperature-dependent motility. At 20–25°C, it produces peritrichous flagella and displays active motility; at 37°C, flagellar synthesis is downregulated, reflecting adaptation to the host environment.
A defining physiological characteristic of Lm is its psychrotolerance. It can grow across a broad temperature range (0–45°C) and pH spectrum (4.5–9.0), and it proliferates—albeit slowly—under typical refrigeration conditions (4°C). Additionally, it forms robust biofilms on food-processing equipment surfaces, conferring tolerance to disinfectants and environmental stressors. These features underpin its persistence in cold-chain facilities and its recurrent association with ready-to-eat and frozen products.
Lm is classified into 13 serotypes based on O and H antigens. Among them, serotype 1/2a predominates in both food isolates and clinical cases and serves as a principal laboratory strain background for mechanistic studies. Over 90% of invasive human listeriosis cases worldwide are attributed to serotypes 1/2a and 4b.
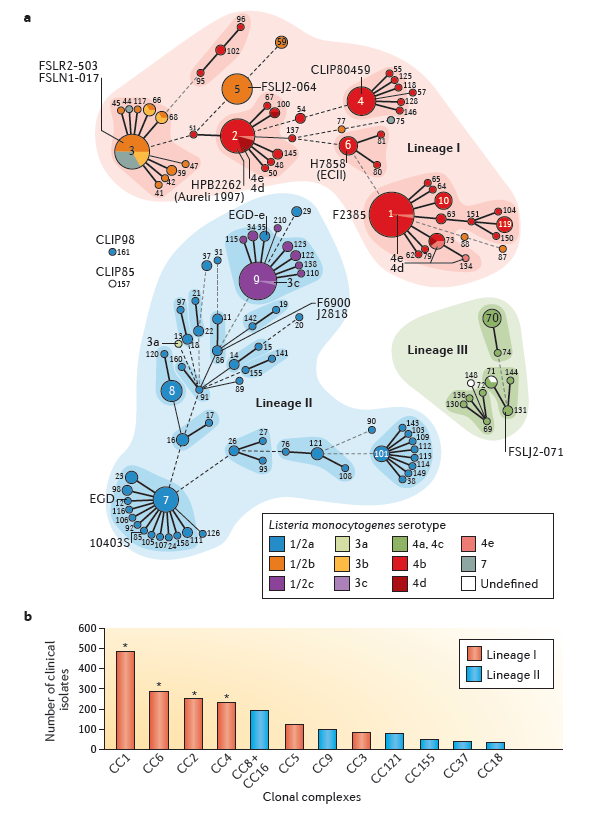
Figure 1: Listeria phylogeny
The bacterial cell wall contains a thick peptidoglycan layer anchoring multiple virulence-associated proteins. Notably, IAP/p60, a peptidoglycan hydrolase, is highly conserved and stably expressed among pathogenic Listeria species. It contributes to cell wall remodeling and bacterial morphology and is closely associated with virulence phenotypes. Importantly, IAP serves as a discriminatory marker between pathogenic and non-pathogenic Listeria species.
Lm is widely distributed in soil, water, and the gastrointestinal tracts of animals. Contamination may occur at any point along the agricultural-to-distribution continuum. High-risk categories include frozen produce and ready-to-eat dairy products. Clinically, listeriosis disproportionately affects pregnant women, the elderly, and immunocompromised individuals. Invasive infections in these populations carry case-fatality rates of 20–30%, highlighting the clinical severity of systemic disease.
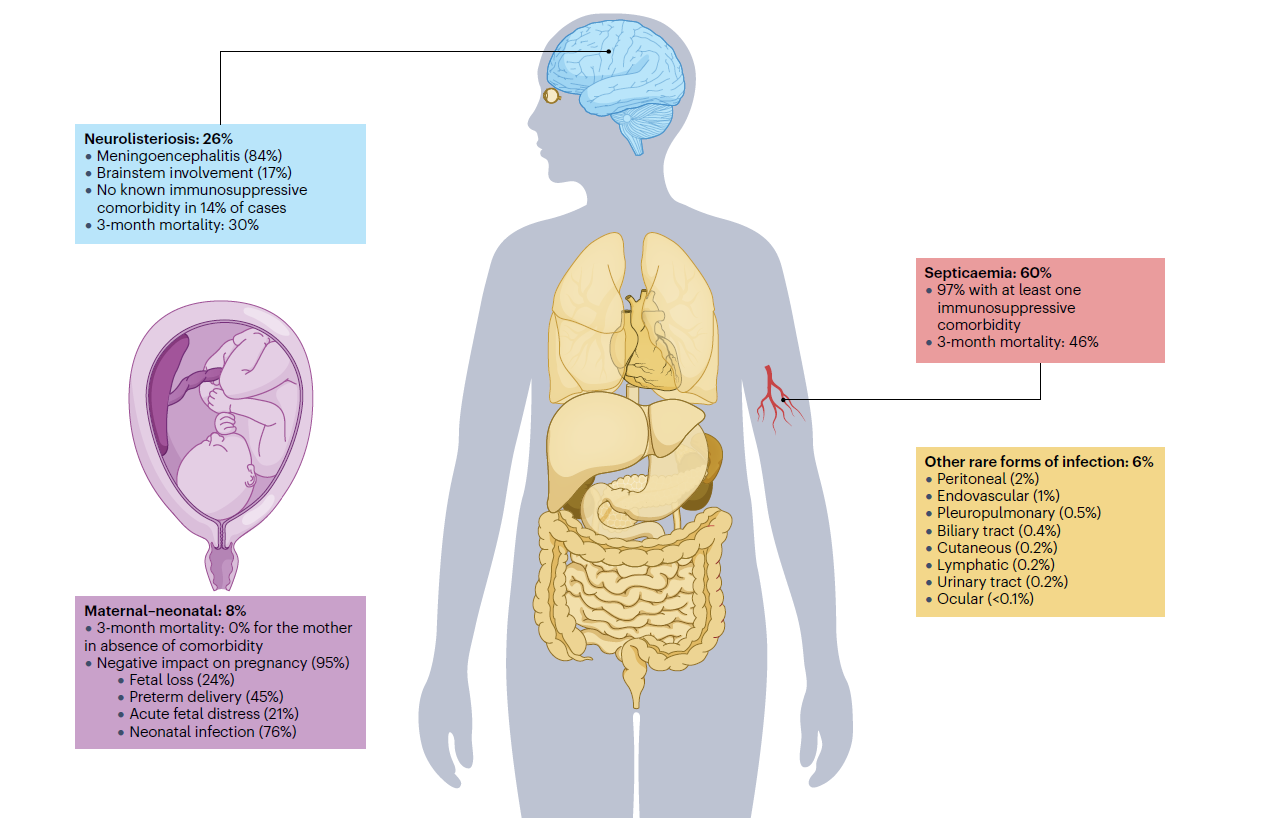
Figure 2: Clinical presentation and features of listeriosis
II. Molecular Mechanisms Underlying Listeria monocytogenes Virulence
The high pathogenicity of Lm derives from its well-orchestrated facultative intracellular lifecycle. Through coordinated expression of virulence factors, the bacterium executes host cell adhesion and invasion, phagosomal escape, cytosolic replication, and cell-to-cell spread, while evading humoral immune clearance. These processes enable translocation across intestinal, blood–brain, and placental barriers, culminating in invasive disease.
Most core virulence determinants are encoded within Listeria Pathogenicity Island 1 (LIPI-1) and are tightly regulated by the transcriptional activator PrfA, forming a hierarchically controlled virulence network.
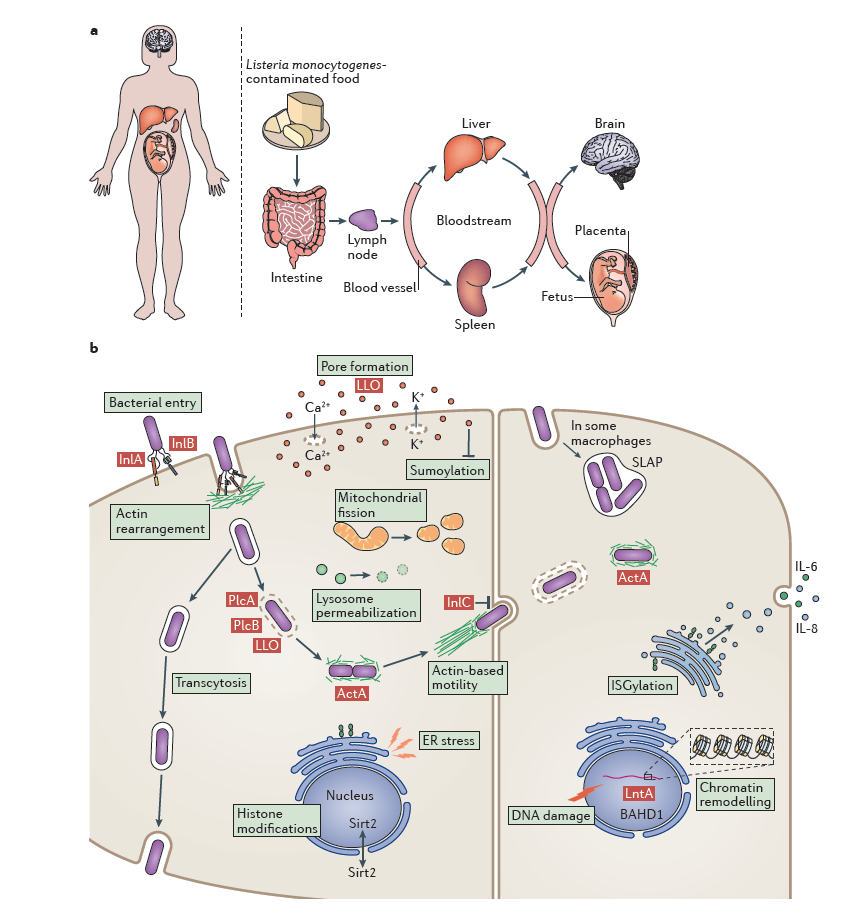
Figure 3: Overview of Listeria monocytogenes infection
1. Host Cell Adhesion and Invasion
Entry into non-phagocytic cells is mediated primarily by the internalin family proteins InlA and InlB.
InlA binds specifically to host E-cadherin via its leucine-rich repeat domain, triggering bacterial adhesion and internalization. This interaction is central to intestinal epithelial invasion and barrier translocation.
InlB interacts with the hepatocyte growth factor receptor (Met), activating the PI3K–Akt signaling pathway and inducing cytoskeletal rearrangements that facilitate bacterial uptake. InlB also contributes to traversal of the blood–brain and placental barriers.
The synergistic function of InlA and InlB establishes efficient colonization of diverse host cell types, including hepatocytes, trophoblasts, and brain microvascular endothelial cells.
2. Phagosomal Escape
Following internalization, Lm resides transiently within a phagosome and must rapidly escape into the cytosol to avoid lysosomal degradation. This step is mediated by listeriolysin O (LLO), encoded by hly, together with phospholipases PlcA and PlcB.
LLO is a cholesterol-dependent cytolysin activated in the acidic phagosomal environment, where it forms pores in the membrane. PlcA and PlcB hydrolyze membrane phospholipids, synergistically enhancing membrane disruption. Deletion of hly abolishes intracellular survival and virulence, underscoring LLO as a central virulence determinant.
3. Cytosolic Replication and Cell-to-Cell Spread
Once in the cytosol, Lm replicates using host-derived nutrients. The ActA protein, encoded by actA, localizes to one bacterial pole and recruits the host Arp2/3 complex, initiating actin polymerization. This results in the formation of actin comet tails that propel bacteria intracellularly and drive membrane protrusions into adjacent cells.
Neighboring cells internalize these protrusions, enabling direct cell-to-cell spread without extracellular exposure. This strategy effectively circumvents antibody-mediated immunity and is pivotal for persistent infection.
4. Barrier Penetration and Systemic Dissemination
After breaching the intestinal epithelium, Lm disseminates hematogenously to the liver and spleen. In susceptible hosts, it can further penetrate the blood–brain barrier, causing meningitis or encephalitis, or cross the placental barrier, leading to miscarriage, preterm birth, or neonatal sepsis. The coordinated activity of InlA, InlB, LLO, and ActA, combined with host immune status and strain-specific virulence traits, determines clinical severity.
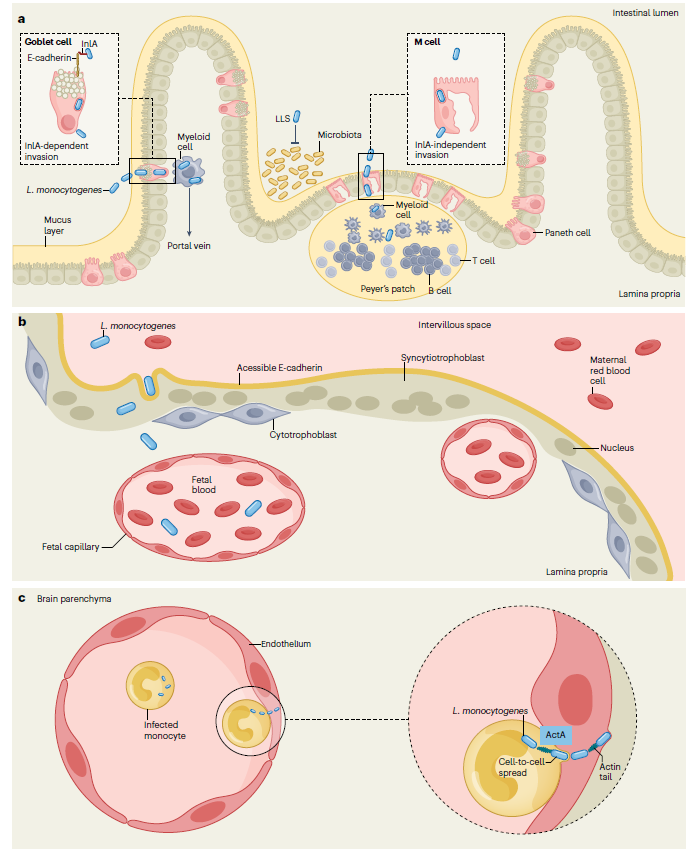
Figure 4: Mechanisms by which L. monocytogenes crosses host barriers
III. Core Research Directions in Listeria monocytogenes
Lm occupies a dual position as both a high-priority food safety pathogen and a foundational model organism in infection biology. Current research spans fundamental mechanistic studies to translational applications.。
1. Detection and Source Tracking Technologies
Rapid and accurate detection underpins both food safety surveillance and clinical diagnostics. Research efforts focus on: Targeting conserved Lm-specific genes and proteins to develop highly specific nucleic acid and immunological assays. Serotyping, whole-genome sequencing (WGS), and multilocus sequence typing (MLST) for phylogenetic analysis and outbreak source attribution. These tools are essential for epidemiological tracing and risk assessment in large-scale food distribution networks.
2. Virulence Regulation and Host–Pathogen Interaction
Key areas include functional dissection of virulence factors, elucidation of the PrfA regulatory network, host pattern recognition mechanisms, and the cellular biology of intracellular parasitism. Insights gained from Lm have broadly informed the conceptual framework for studying intracellular bacterial pathogens.
3. Control Strategies and Vaccine Development
Preventive strategies encompass: Anti-biofilm technologies and novel antimicrobial agents targeting psychrotolerant growth in food-processing environments. Environmental sanitation innovations. Vaccine development targeting major virulence antigens to elicit protective immunity, particularly for high-risk populations.
4. Synthetic Biology and Translational Applications
Lm’s intrinsic capacity for cytosolic antigen delivery and robust cellular immune activation has positioned it as a promising vector platform in cancer immunotherapy. Attenuated strains engineered via targeted virulence gene deletions can be designed to express tumor-associated antigens and immunomodulatory molecules, promoting antigen presentation and potent CD8+ T cell responses. Multiple Lm-based therapeutic vaccines have advanced into clinical evaluation.
About Us
abinScience was founded in France and is focused on the development and production of high-quality life science reagents. Based in the innovative technology hub of Strasbourg, France, our vision is to "Empower Bioscience Discovery."
abinScience Listeria-related proteins and antibodies
| Product Name |
Catalog No. |
| Anti-Listeria monocytogenes serovar 1/2a InlB/Internalin B Antibody (SAA0363) |
JN937013 |
| Anti-Listeria monocytogenes serovar 1/2a IAP Monoclonal Antibody (1A180) |
JN846025 |
| Anti-Listeria monocytogenes serotype 1/2a InlB/Internalin B Nanobody (SAA0997) |
JN937023 |
| Recombinant Listeria monocytogenes IAP/p60 Protein, N-His |
JN846012 |
References
[1] Lecuit M. Listeria monocytogenes, a model in infection biology. Cell Microbiol. 2020 Apr;22(4):e13186. doi: 10.1111/cmi.13186. PMID: 32185900.
[2] Disson O, Moura A, Lecuit M. Making Sense of the Biodiversity and Virulence of Listeria monocytogenes. Trends Microbiol. 2021 Sep;29(9):811-822. doi: 10.1016/j.tim.2021.01.008. Epub 2021 Feb 11. PMID: 33583696.
[3] Meireles D, Pombinho R, Cabanes D. Signals behind Listeria monocytogenes virulence mechanisms. Gut Microbes. 2024 Jan-Dec;16(1):2369564. doi: 10.1080/19490976.2024.2369564. Epub 2024 Jul 9. PMID: 38979800; PMCID: PMC11236296.
[4] Jordan K, McAuliffe O. Listeria monocytogenes in Foods. Adv Food Nutr Res. 2018;86:181-213. doi: 10.1016/bs.afnr.2018.02.006. Epub 2018 Apr 3. PMID: 30077222.
[5] Lopes-Luz L, Mendonça M, Bernardes Fogaça M, Kipnis A, Bhunia AK, Bührer-Sékula S. Listeria monocytogenes: review of pathogenesis and virulence determinants-targeted immunological assays. Crit Rev Microbiol. 2021 Sep;47(5):647-666. doi: 10.1080/1040841X.2021.1911930. Epub 2021 Apr 24. PMID: 33896354.
[6] Radoshevich L, Cossart P. Listeria monocytogenes: towards a complete picture of its physiology and pathogenesis. Nat Rev Microbiol. 2018 Jan;16(1):32-46. doi: 10.1038/nrmicro.2017.126. Epub 2017 Nov 27. PMID: 29176582.
[7] Disson O, Charlier C, Pérot P, Leclercq A, Paz RN, Kathariou S, Tsai YH, Lecuit M. Listeriosis. Nat Rev Dis Primers. 2025 Oct 2;11(1):71. doi: 10.1038/s41572-025-00654-x. PMID: 41038831.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






