Flow cytometry is one of the most powerful technologies for quantitative phenotyping and functional analysis of single cells. Among all experimental variables, flow cytometry antibodies are the core reagents that ultimately determine data reliability and reproducibility.
In practice, researchers often invest substantial effort into target selection, multicolor panel design, fluorochrome matching, and instrument optimization, while underestimating one critical variable: the antibody clone.
Extensive laboratory experience and standardization guidance from the International Society for Advancement of Cytometry (ISAC) demonstrate that different clones against the same target can exhibit order-of-magnitude differences in performance within identical experimental systems. Many researchers have encountered the following scenario: the target is correct, the protocol is standardized, instrument settings are appropriate—yet nonspecific signals, poor separation of positive and negative populations, high background, or irreproducible results persist. After excluding operational, sample, and instrument-related factors, the root cause is often a mismatch between the antibody clone and the experimental system.
Understanding clone selection is therefore not a minor technical detail—it is a determinant of experimental validity.
1. The Biological Nature of Antibody Clones
1.1 What a Clone Truly Represents
An antibody clone is not merely a catalog identifier; it represents a unique hybridoma cell line that secretes a monoclonal antibody targeting a single antigenic epitope. Each clone originates from an immortalized B-cell hybridoma and has a unique variable region sequence that defines its binding characteristics.
While the target defines which protein is recognized, the clone determines:
• Which epitope on the protein is bound
• Binding affinity and kinetics
• Sensitivity to fixation, permeabilization, or enzymatic treatment
• Compatibility with specific experimental protocols
Focusing solely on the target while ignoring the clone effectively relinquishes control over antibody performance.
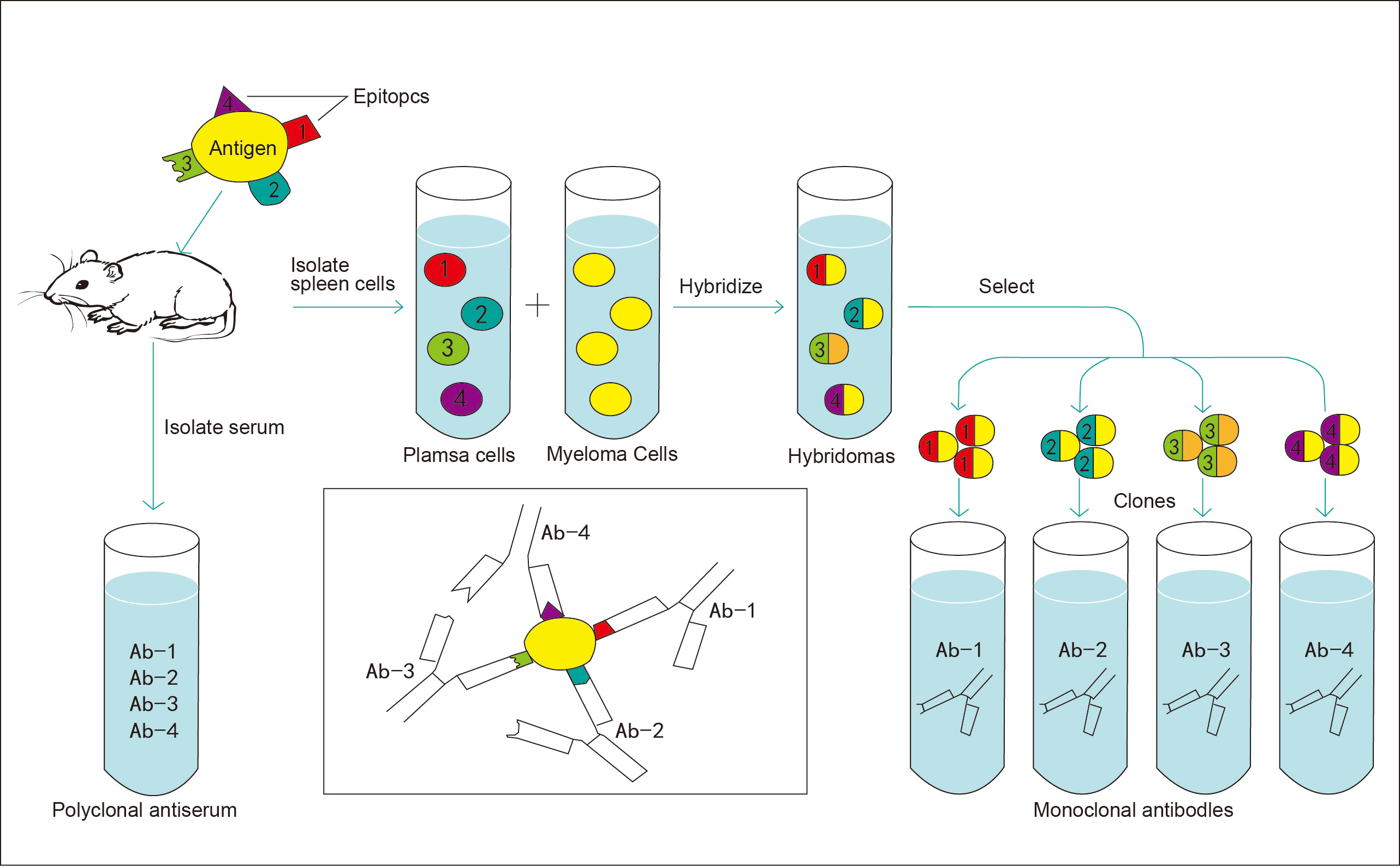
Figure 1. Workflow of monoclonal antibody generation
1.2 Why Clones Against the Same Target Perform Differently
Even when directed against the same antigen, different clones can produce markedly different results due to differences in their variable region sequences. These differences affect several critical parameters:
Epitope Recognition (Fundamental Determinant)
Epitopes may be conformational (dependent on native protein structure) or linear (retained after denaturation). The epitope location (extracellular, intracellular, nuclear) and type directly determine application suitability.
For example, live-cell surface staining requires clones that recognize extracellular conformational epitopes. Clones targeting intracellular domains or denatured epitopes will fail under such conditions.
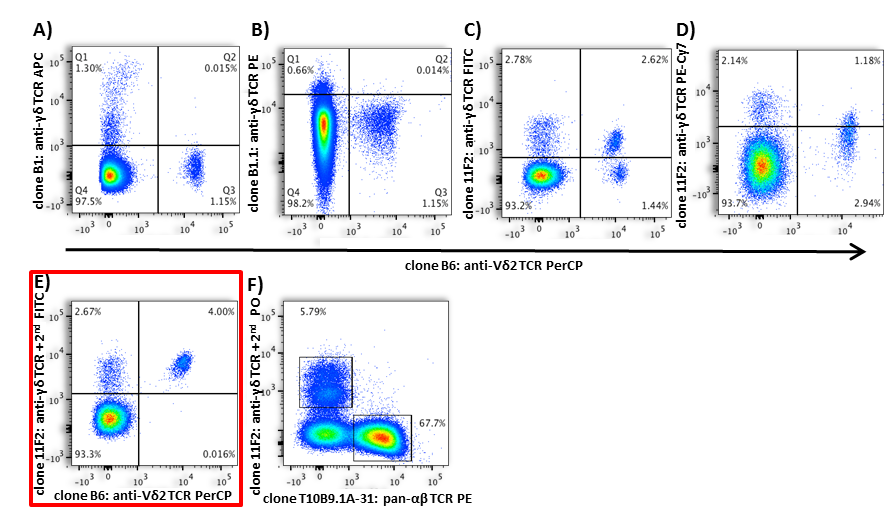
Figure 2. Differential flow cytometric detection of TCR γ/δ and TCR Vδ2 using distinct antibody clones
Affinity and Specificity
Binding affinity directly influences detection sensitivity and is commonly reflected by the staining index (SI). A higher SI typically indicates improved separation between positive and negative populations. Low-abundance targets demand particularly high-affinity clones.
However, high affinity does not guarantee high specificity. Some high-affinity clones exhibit cross-reactivity, which may reduce signal-to-noise ratio rather than improve it.
Compatibility with Sample Processing
Fixation, permeabilization, and enzymatic digestion can alter protein conformation or destroy epitope integrity. Clones recognizing strictly conformational epitopes may completely lose binding activity after formaldehyde fixation or PMA/BFA stimulation protocols.
Clone tolerance to sample processing conditions must therefore be experimentally verified.
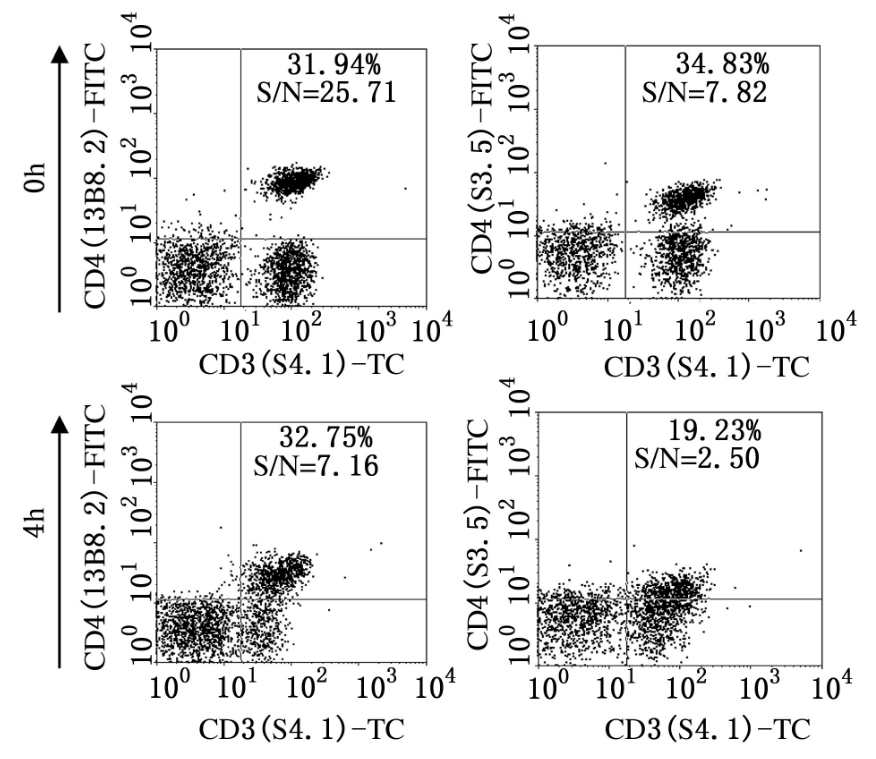
Figure 3. Clone-dependent detection of CD4 following PMA/BFA stimulation for 4 h
Species Cross-Reactivity
Even when homologous proteins share over 95% sequence identity between species, a single amino acid difference within the recognized epitope can abolish binding. Species reactivity must rely on explicit validation data rather than sequence homology assumptions.
Vendor-to-Vendor Variability
A clone defines the hybridoma origin—but final antibody performance is influenced by:
• Purification methods
• Fluorochrome conjugation efficiency
• Buffer formulation
• Lot-to-lot quality control
Thus, the same clone from different suppliers—or even different lots—may demonstrate significant performance variability.
2. Strategic Principles for Clone Selection
For routine and well-established applications (e.g., human PBMC immunophenotyping or basic mouse splenocyte profiling), widely validated “gold-standard” clones are available. In these cases, extensive screening is typically unnecessary.
However, in non-classical systems—such as unconventional fixation/permeabilization protocols, enzymatically digested solid tissues, or low-abundance targets—clone compatibility becomes a decisive factor, particularly when unexplained experimental anomalies arise.
The following principles can guide rational clone selection:
2.1 Define the Experimental Context First
Key parameters that determine clone suitability include:
• Surface vs. intracellular vs. nuclear staining
• Species and tissue source
• Fixation, permeabilization, or digestion protocols
• Target expression level
• Experimental objective (phenotyping vs. functional detection)
The most common cause of clone-related failure is validation mismatch between the supplier’s testing conditions and the actual experimental system.
2.2 Prioritize Validated References to Reduce Trial-and-Error
Whether selecting antibodies initially or troubleshooting unexpected results, choosing clones validated under comparable conditions is the most efficient strategy.
Authoritative references include:
• Industry-recognized gold-standard clones
• Peer-reviewed studies using comparable systems
• The ISAC OMIP (Optimized Multicolor Immunofluorescence Panel) database
In most cases, evaluating two to three candidate clones is sufficient to identify an optimal option.
2.3 Verify Key Technical Specifications
Many experimental failures stem from overlooked specification mismatches. Four critical points must be confirmed:
• Explicit validation for flow cytometry (FC)
• Compatibility with the intended sample preparation method
• Confirmed species reactivity
• Availability of appropriate isotype controls
2.4 Perform Focused Pilot Comparisons When Necessary
For uncommon targets or non-standard systems, parallel pilot experiments under strictly controlled conditions can efficiently identify the most suitable clone. Only a single variable—the clone—should differ between test groups, and a complete control panel should be included.
3. Common Misconceptions in Clone Selection
Misconception 1: Selecting Based Only on Target
The target determines what protein is detected; the clone determines how it is detected. Ignoring the clone frequently results in nonspecific signals, false positives/negatives, and irreproducible data—potentially leading to incorrect scientific conclusions.
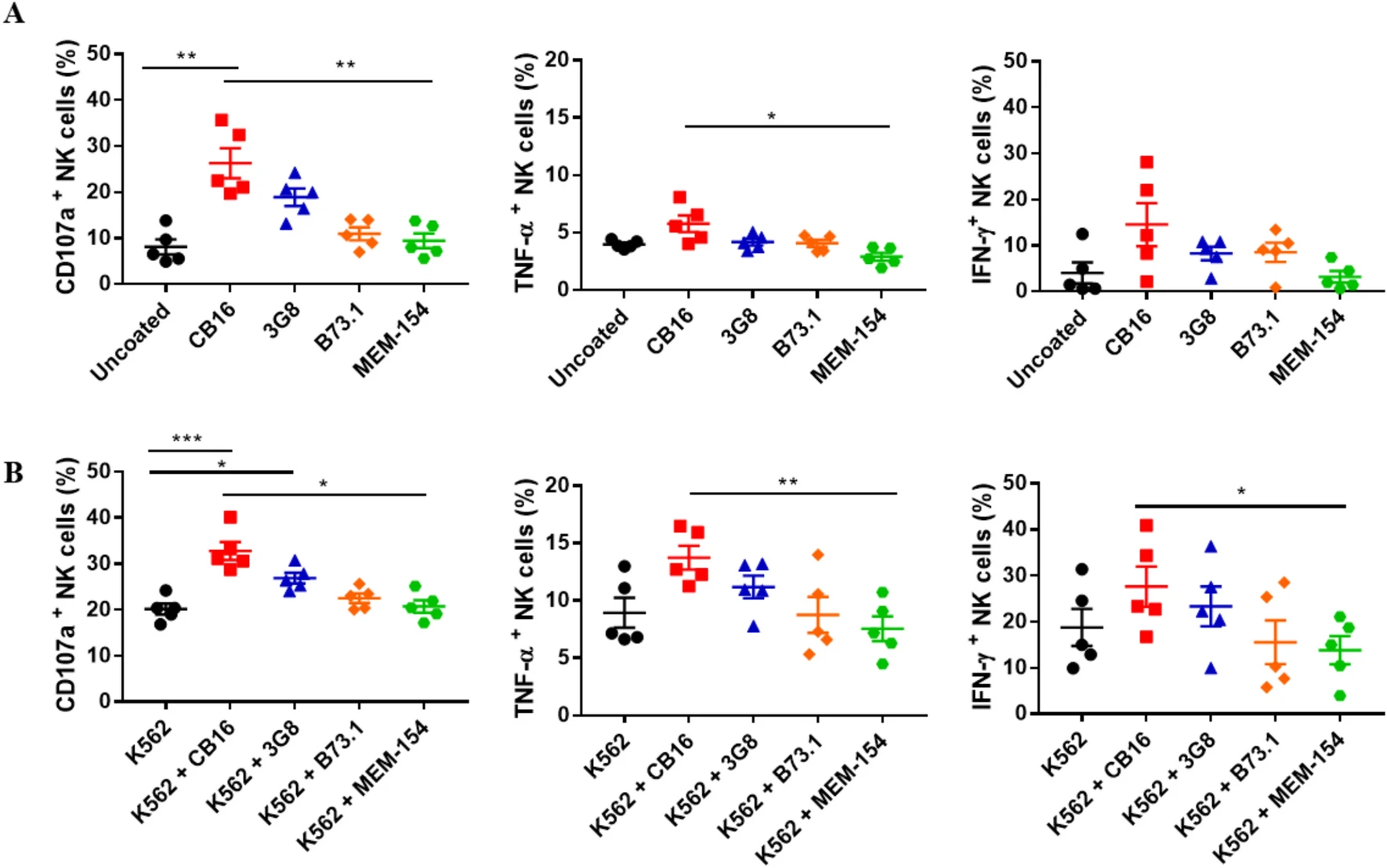
Figure 4. Functional responses of NK cells induced by different anti-CD16 antibody clones
Misconception 2: Blindly Following High-Impact Publications
Clone performance is highly system-dependent. Differences in species, sample processing, or experimental objectives can drastically alter performance. Adopting the clone used in the literature without verifying experimental compatibility often leads to failed replication.
Misconception 3: Assuming Identical Performance Across Vendors or Lots
Even with identical clone names, switching suppliers, fluorochromes, or production lots can significantly alter staining performance. Directly using the original working concentrations without re-optimization may result in increased background or poor reproducibility.
Misconception 4: Using WB/IHC-Validated Clones Directly for Flow Cytometry
Western blotting and immunohistochemistry often detect denatured linear epitopes. Flow cytometry typically requires recognition of native conformational epitopes. Clones validated only for WB or IHC frequently fail in flow applications, leading to undetectable signals or false-negative results
4. Conclusion
Reliable flow cytometry data result from rigorous control of every experimental variable. Among these variables, the antibody clone is one of the most frequently overlooked yet highly impactful determinants of performance.
For routine applications, selecting widely validated gold-standard clones is typically sufficient. However, when unexplained anomalies arise, systematically evaluating clone compatibility often reveals the underlying issue and significantly reduces unnecessary trial-and-error.
A rational, biology-driven approach to clone selection is not merely a purchasing decision—it is a cornerstone of experimental integrity.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience specializes in the development and production of high-quality life science reagents, and is committed to providing researchers with high-performance, highly reliable Flow Cytometry Antibodies, we offer:
1. Stringently validated flow cytometry antibodies including CD3, CD4, CD8 ensuring high sensitivity and minimal background noise;
2. A comprehensive portfolio of immunology research tools for multiple species, including Human, Mouse, Rat, Dog, Hamster;
3. Expert technical support and panel design consultation to help you accurately resolve complex immune cell populations.
For more information on abinScience flow cytometry antibodies, please visit:
abinScience Flow Cytometry Antibodies
References
[1] Bigby M, Wang P, Fierro JF, Sy MS. Phorbol myristate acetate-induced down-modulation of CD4 is dependent on calmodulin and intracellular calcium. J Immunol. 1990 Apr 15;144(8):3111-6. PMID: 2109010.
[2] Wistuba-Hamprecht K, Pawelec G, Derhovanessian E. OMIP-020: phenotypic characterization of human γδ T-cells by multicolor flow cytometry. Cytometry A. 2014 Jun;85(6):522-4. doi: 10.1002/cyto.a.22470. Epub 2014 Apr 22. PMID: 24756989.
[3] Kim J, Phan MT, Hwang I, Park J, Cho D. Comparison of the different anti-CD16 antibody clones in the activation and expansion of peripheral blood NK cells. Sci Rep. 2023 Jun 11;13(1):9493. doi: 10.1038/s41598-023-36200-6. PMID: 37302991; PMCID: PMC10258201.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский







