When analyzing the CD4/CD8 phenotype of T cells in peripheral blood via flow cytometry, a small, unique cell population is frequently detected outside the clusters of CD4+ and CD8+ T cells. These cells fall within the double-positive quadrant for both markers (R10 in Figure 1), defying long-established immunological conventions.
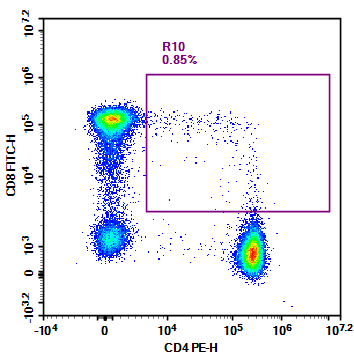
Figure 1. CD4/CD8 T Cell Detection in Human Peripheral Blood
According to the classical immunological theory (Figure 2), this cell population should not exist in the peripheral blood. After mature T cells egress from the thymus, they exclusively exist in one of two single-positive phenotypes: CD4+ T cells that mediate immune regulation, and CD8+ T cells that execute cytotoxic effector functions. The CD4+CD8+ double-positive (DP) phenotype is an undetermined transitional state during thymic T cell development, and cells bearing this phenotype are supposed to be strictly eliminated through thymic selection processes. So why do these cells appear in peripheral blood? Are they simply artifacts from non-specific binding during detection, or do they represent an undiscovered mechanism of the immune system?
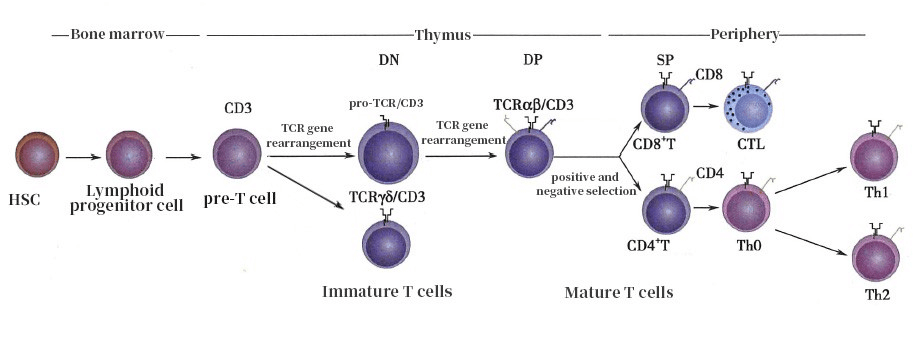
Figure 2. T Cell Development Process
As early as the 1980s, researchers identified that approximately 3% of T cells in the peripheral blood of healthy individuals co-express CD4 and CD8 molecules using two-color flow cytometry—a finding that directly challenged the decades-old canonical theory of T cell development. With the advancement of polychromatic flow cytometry, researchers further clarified that these cells are not a homogeneous population, but are mainly divided into two distinct subsets: the CD4hiCD8low subset, which exhibits a memory phenotype and maintains long-term immunological memory; and the CD4lowCD8hi subset, which is biased toward effector functions and possesses direct cytotoxic activity against abnormal cells.
More notably, the frequency of these cells can change dramatically under specific conditions. In individuals with HIV, the proportion of DP T cells in peripheral blood can rise to over 10%, and they also undergo abnormal expansion in patients with autoimmune diseases. When peripheral blood mononuclear cells (PBMCs) are stimulated in vitro with phytohemagglutinin (PHA), the frequency of DP T cells can even exceed 50%, with the majority retaining the CCR7+CD45RA+ phenotype of naïve T cells. These observations fully demonstrate that DP T cells are by no means an accidental detection artifact, but rather a functionally critical component of the immune system.
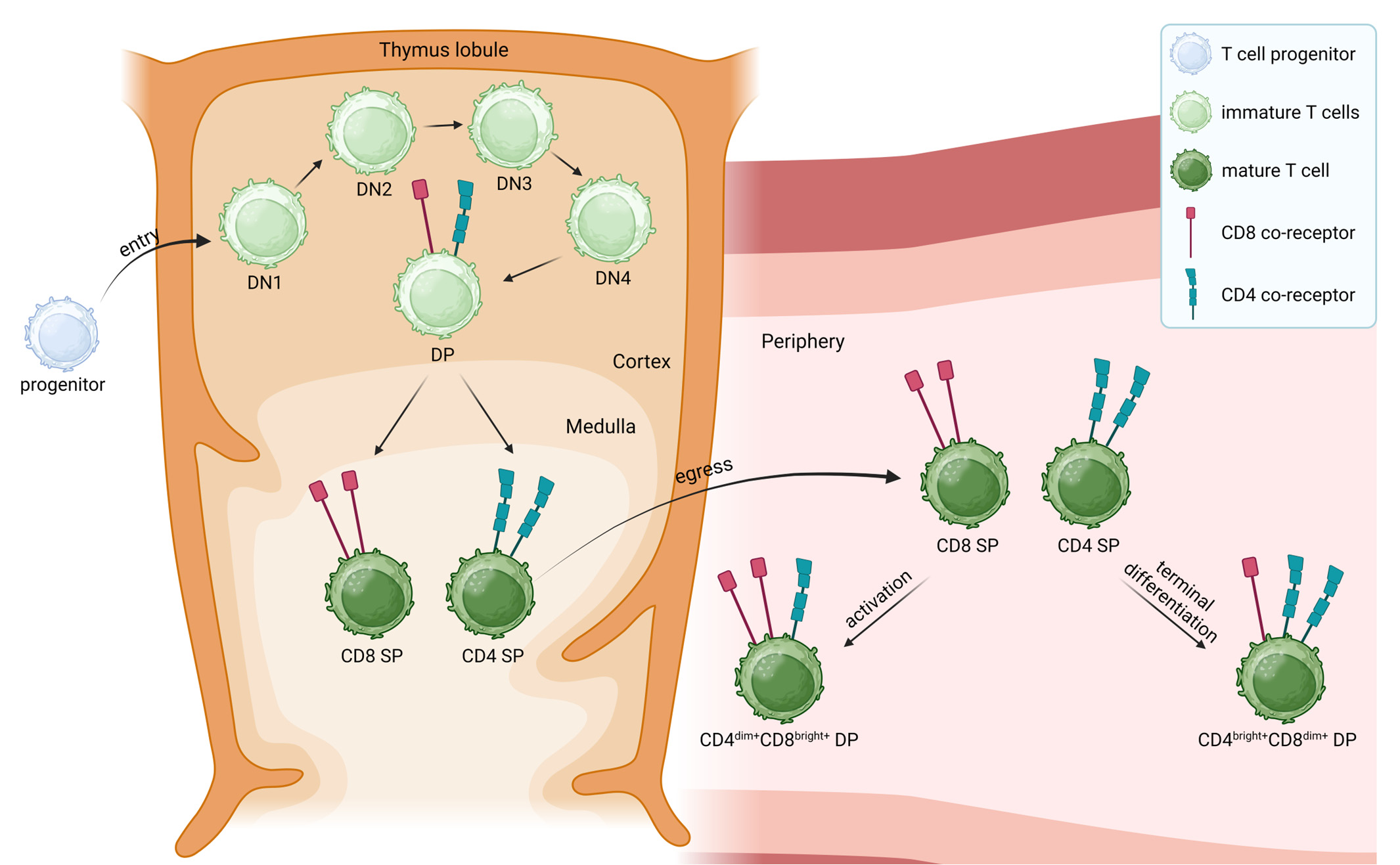
Figure 3. T Cell Development and Potential Origins of Mature CD4hiCD8low and CD4lowCD8hi Double-Positive T Cells
2.1 Thymic Origin: Residual Immature Thymocytes
The thymus is the central organ for T cell development, and the DP stage is an obligate step in T cell maturation. At this stage, thymocytes co-express CD4 and CD8 molecules, awaiting positive selection based on their interaction with self-MHC molecules. Under physiological conditions, thymocytes that fail selection undergo apoptosis, while those that pass selection differentiate into SP T cells before egressing to the periphery. However, a small number of incompletely mature DP thymocytes can prematurely migrate to the peripheral blood. These cells typically express immature markers such as CD1a, and are found at higher frequencies in children and individuals with active thymic function.
2.2 Peripheral Conversion: Phenotypic Plasticity of Mature T Cells
This is the predominant source of DP T cells in peripheral blood, centered on the phenotypic plasticity of mature T cells. Under specific stimulatory signals, the previously silenced CD4 or CD8 co-receptor genes can be reactivated in mature SP T cells, endowing them with a DP phenotype.
This conversion has been validated both in vitro and in vivo. In in vitro experiments, stimulation of PBMCs with PHA rapidly induces the generation of a large number of DP T cells, most of which retain the CCR7+CD45RA+ phenotype of naïve T cells. In vivo, this process is triggered by antigenic stimulation such as persistent viral infection and chronic inflammation under pathological conditions. For example, the frequency of DP T cells in the peripheral blood of HIV-infected individuals can exceed 10%, and these cells often highly express exhaustion markers such as PD-1 and CTLA-4, displaying a phenotype of hyperactivation yet functional impairment.
Among the confirmed origins of peripheral DP T cells, peripheral conversion of mature T cells is widely accepted as the major contributing pathway. Its core mechanism lies in the phenotypic reprogramming of mature CD4+ or CD8+ SP T cells: under sustained antigenic stimulation from chronic viral infection, persistent inflammation, or other pathological contexts, transcription of the silenced CD8 or CD4 gene is reinitiated, resulting in concurrent expression of both co-receptors.
2.3 Cell-Cell Interaction: Trogocytosis-Mediated Phenotype Acquisition
Another potential mechanism underlying the detection of DP T cells is trogocytosis, an active process by which cells acquire membrane proteins and cytoplasmic fragments from other cells via direct cell-to-cell contact. When CD4+ and CD8+ T cells form tight interactions through an immunological synapse, they can mutually acquire membrane fragments from one another: CD4+ T cells may obtain CD8 molecules, and CD8+ T cells may acquire CD4 molecules, thus displaying a double-positive phenotype in flow cytometric detection. This phenomenon does not represent genuine phenotypic conversion of the cells, but rather a unique manifestation of crosstalk and coordination between immune cells.
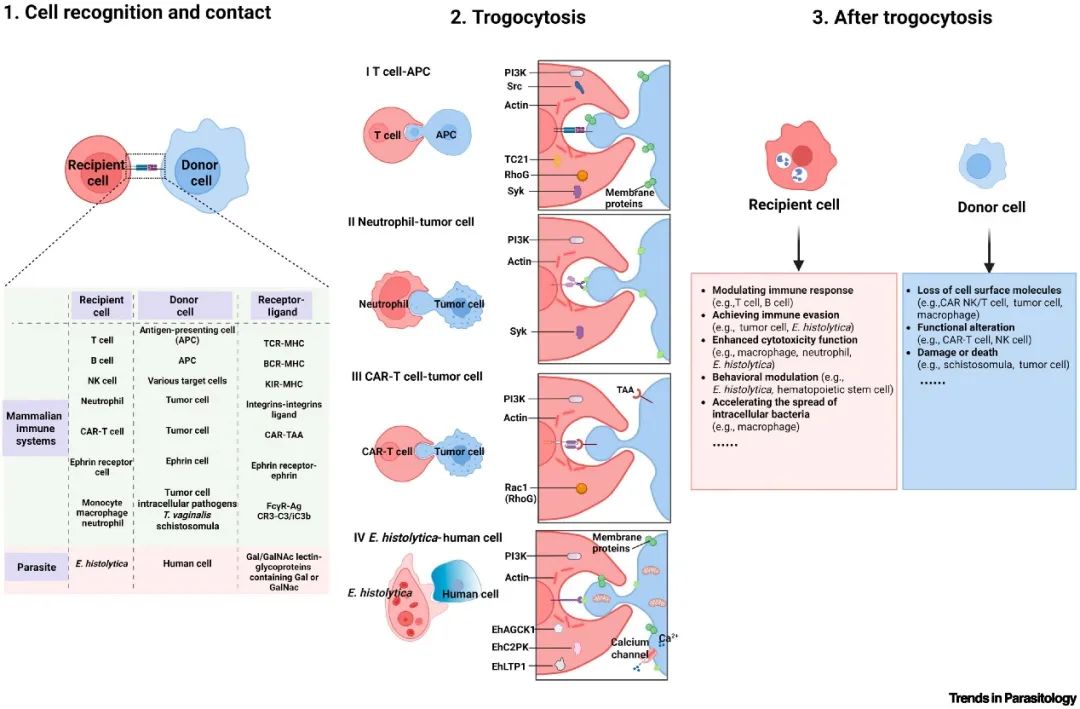
Figure 4. Molecular Mechanism Underlying Trogocytosis
Although DP T cells are considered an "exception" in classical immune theory, a growing body of research indicates that they are not a functionally redundant or purely aberrant population. Instead, they play unique and non-redundant roles in immune regulation and effector functions:
Dual Functional Potential of Helper and Cytotoxic Activity:
CD4+CD8+ DP T cells break the functional dichotomy between CD4+ helper and CD8+ cytotoxic T cells. They can not only recognize antigens via CD4 molecules and secrete cytokines to orchestrate immune responses, but also trigger the perforin/granzyme pathway through CD8 molecules to directly kill abnormal cells, exerting a synergistic regulatory-cytotoxic effect in contexts such as the tumor microenvironment.
uperiority in Immunological Memory and Rapid Recall Response
Particularly within the CD4lowCD8hi subset, cells universally express memory markers including CCR7 and CD45RO, and can form a memory reservoir in lymph nodes. Upon re-exposure to cognate antigens, their activation speed is 3-fold faster than that of conventional memory T cells, enabling rapid differentiation into effector cells to sustain immune protection.
An Immune Warning Marker for Disease Progression
In diseases such as HIV infection and multiple sclerosis (MS), quantitative and phenotypic changes of DP T cells are directly correlated with disease progression. The frequency of these cells in peripheral blood can exceed 10% in HIV patients, and their number in the cerebrospinal fluid of MS patients is 4-fold higher than that in healthy individuals. These characteristics make them promising potential biomarkers for disease monitoring and therapeutic efficacy evaluation.
A Novel Intervention Target for Clinical Translation
Based on their unique functional properties, DP T cells have emerged as a promising new direction in immunotherapy. Adoptive transfer of in vitro expanded DP T cells significantly inhibits tumor growth, while targeted depletion of these cells can alleviate inflammation in autoimmune diseases. In vaccine development, inducing the generation of DP T cells can nearly double the duration of immune protection, offering new insights for the prevention and treatment of diseases.
As a strategic venture of AtaGenix (established 2011), abinScience specializes in the development and production of high-quality life science reagents, and is committed to providing researchers with high-performance, highly reliable Flow Cytometry Antibodies, we offer:
1. Stringently validated flow cytometry antibodies including CD3, CD4, CD8 ensuring high sensitivity and minimal background noise;
2. A comprehensive portfolio of immunology research tools for multiple species, including Human, Mouse, Rat, Dog, Hamster;
3. Expert technical support and panel design consultation to help you accurately resolve complex immune cell populations.
For more information on abinScience flow cytometry antibodies, please visit:
abinScience Flow Cytometry Antibodies
[1] Blue ML, Daley JF, Levine H, Schlossman SF. Coexpression of T4 and T8 on peripheral blood T cells demonstrated by two-color fluorescence flow cytometry. J Immunol. 1985 Apr;134(4):2281-6.
[2] Kochanowicz AM, Osuch S, Berak H, Kumorek A, Caraballo Cortés K. Double Positive CD4+CD8+ (DP) T-Cells Display Distinct Exhaustion Phenotype in Chronic Hepatitis C. Cells. 2023 May 22;12(10):1446.
[3] Sullivan YB, Landay AL, Zack JA, Kitchen SG, Al-Harthi L. Upregulation of CD4 on CD8+ T cells: CD4dimCD8bright T cells constitute an activated phenotype of CD8+ T cells. Immunology. 2001 Jul;103(3):270-80.
[4] Hagen M, Pangrazzi L, Rocamora-Reverte L, Weinberger B. Legend or Truth: Mature CD4+CD8+ Double-Positive T Cells in the Periphery in Health and Disease. Biomedicines. 2023 Oct 5;11(10):2702.
[5] Toberer F, Christopoulos P, Lasitschka F, Enk A, Haenssle HA, Cerroni L. Double-positive CD8/CD4 primary cutaneous acral T-cell lymphoma. J Cutan Pathol. 2019 Mar;46(3):231-233.
[6] Parrot T, Oger R, Allard M, Desfrançois J, Raingeard de la Blétière D, Coutolleau A, Preisser L, Khammari A, Dréno B, Delneste Y, Guardiola P, Fradin D, Gervois N. Transcriptomic features of tumour-infiltrating CD4lowCD8high double positive αβ T cells in melanoma. Sci Rep. 2020 Apr 3;10(1):5900.
[7] Hess NJ, Turicek DP, Riendeau J, McIlwain SJ, Contreras Guzman E, Nadiminti K, Hudson A, Callander NS, Skala MC, Gumperz JE, Hematti P, Capitini CM. Inflammatory CD4/CD8 double-positive human T cells arise from reactive CD8 T cells and are sufficient to mediate GVHD pathology. Sci Adv. 2023 Mar 24;9(12):eadf0567.
[8] Choi YJ, Park HJ, Park HJ, Jung KC, Lee JI. CD4hiCD8low Double-Positive T Cells Are Associated with Graft Rejection in a Nonhuman Primate Model of Islet Transplantation. J Immunol Res. 2018 Jul 10;2018:3861079.
[9] Shen J, Li X, Hide G, Lun ZR, Wu Z. Trogocytosis: revealing new insights into parasite-host interactions. Trends Parasitol. 2025 Jul;41(7):572-590.
+86-027-65523339
중국 우한시 심둔사로 666번지 C동, 우한, 430206

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский