Date de publication :
2025-12-01 Nombre de vues : 159
I. Materials Preparation
1. Cell culture dish
2. 75% ethanol
3. Ophthalmic scissors and forceps (or scalpel)
4. 1-3 mL syringe and needle
5. 70 μm or 200 mesh filter
6. 15 mL or 50 mL centrifuge tube
7. Red blood cell (RBC) lysis buffer
Note: If the cells are to be cultured afterwards, sterilise the materials to ensure aseptic conditions.
II. Harvesting Mouse Tibia and Femur
Euthanise the mouse using cervical dislocation, followed by immersion in 75% ethanol for 5 minutes. Place the mouse on the operating surface, and then cut the thigh at both the hip joint and the ankle, carefully removing the fibrous and muscle tissues from the bone to obtain the tibia and femur. The mouse femur is shown in Figure 1.
Figure 1. Mouse Femur
III. Preparation of Bone Marrow Single-Cell Suspension
1. Place the bones in a petri dish containing PBS or culture medium.
2. Remove the joints at both ends of the tibia and femur to expose the bone marrow cavity.
3. Using a syringe, flush the bone marrow cavity with an appropriate amount of PBS or culture medium to release the bone marrow (repeat the washing until the marrow cavity appears white).
4. Gently pipette or syringe the cell clumps to disperse them.
5. Filter the cells through a 70 μm or 200 mesh filter, and collect the filtrate into a centrifuge tube. Centrifuge at 300-500 g for 5 minutes and discard the supernatant.
6. Lyse red blood cells (optional: determine if RBC lysis is necessary based on subsequent experiments).
7. Resuspend the cells in PBS or culture medium, count the cells, and adjust the concentration to 1×10⁷/mL.
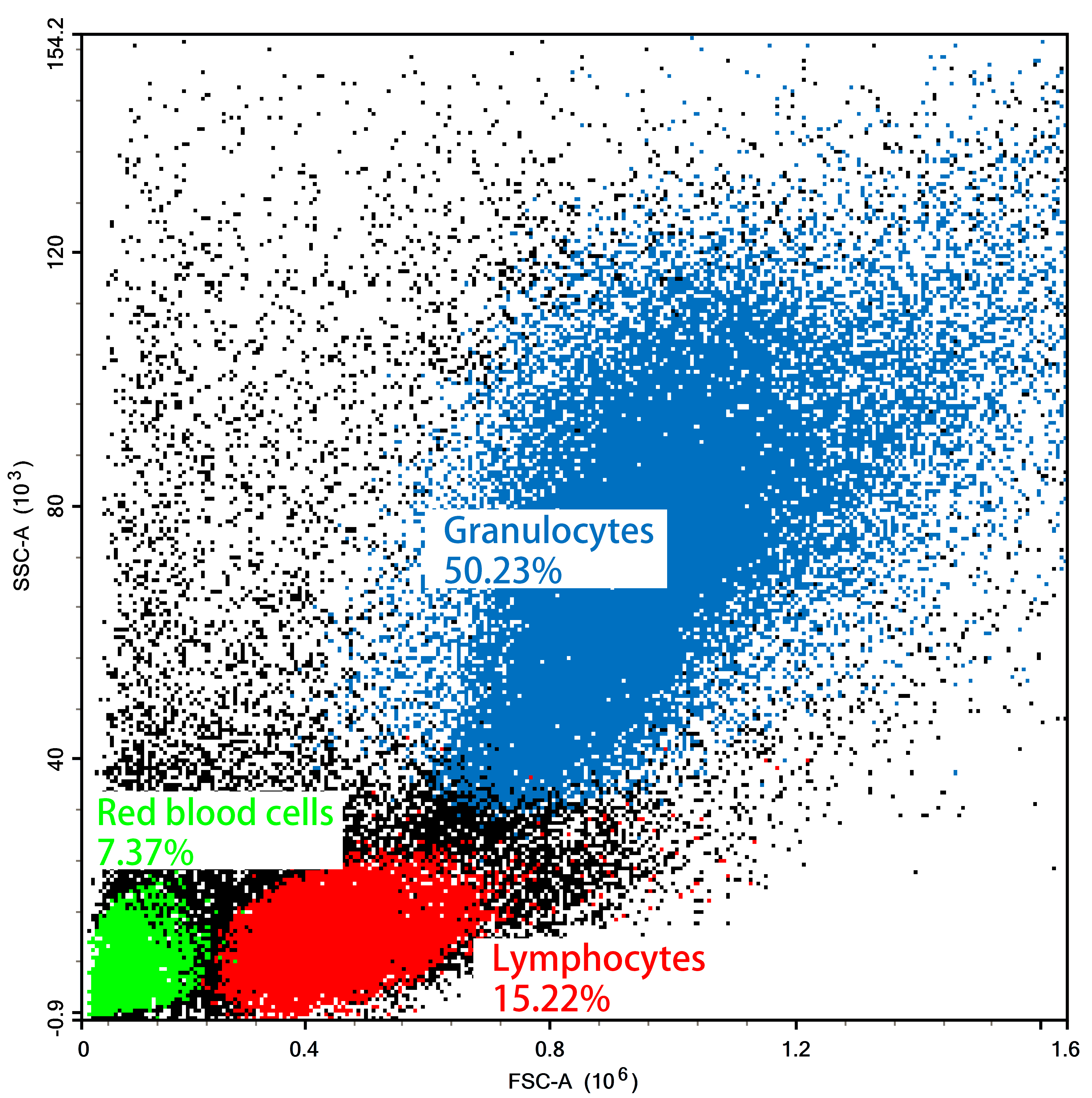
Figure 2. Flow Cytometry FSC/SSC Results of Mouse Bone Marrow Cells
IV. Notes
1. When stripping bone and muscle, ensure that the joints at both ends of the bone remain intact; breaking them may result in cell loss.
2. If MSCs are to be cultured, RBC lysis can be omitted; however, if differentiation of mononuclear cells from the bone marrow into macrophages or dendritic cells is required, RBC lysis is generally necessary.
3. If the bone marrow cells are to be further cultured, maintain aseptic conditions throughout the procedure; for regular flow cytometry experiments, sterile conditions are not essential.
4. Cell counting can be performed using a haemocytometer or an automated cell counter.
5. To maintain cell viability and slow down metabolism, perform the procedure at low temperatures (on ice or in a 4°C environment).
About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский




