Antibody titration is often regarded as a core competency in flow cytometry, with the expectation that each antibody should be evaluated before use. Conversely, some perceive it as labor-intensive and cumbersome, assuming that following the datasheet’s recommended concentration is sufficient.
Although titration is generally recommended, many researchers—particularly those new to flow cytometry—may not fully appreciate its purpose, methodology, or practical significance.
In fact, antibody titration is neither a complex technique nor a procedural formality. It constitutes a fundamental step that ensures optimal signal resolution, enhances data reproducibility, and promotes efficient reagent utilization.
1. What is Flow Cytometry Antibody Titration?
In essence, flow cytometry antibody titration is the process of determining the optimal working concentration of an antibody for a specific experimental system.
The core principle of flow cytometry is using fluorescently labeled antibodies to detect target antigens on cells and distinguishing positive from negative populations based on fluorescence intensity. Antibody concentration directly affects this distinction:
• Too low: Inadequate antibody may fail to occupy all antigen sites, resulting in weak positive signals or undetectable low-expression antigens.
• Too high: Surplus antibody may lead to non-specific binding, increasing background fluorescence and diminishing the separation between positive and negative populations, thereby complicating gating.
The goal of titration is not to maximize signal intensity. Rather, it identifies a balance between specific and non-specific binding, enabling adequate antigen detection while minimizing background staining to achieve clear population resolution.
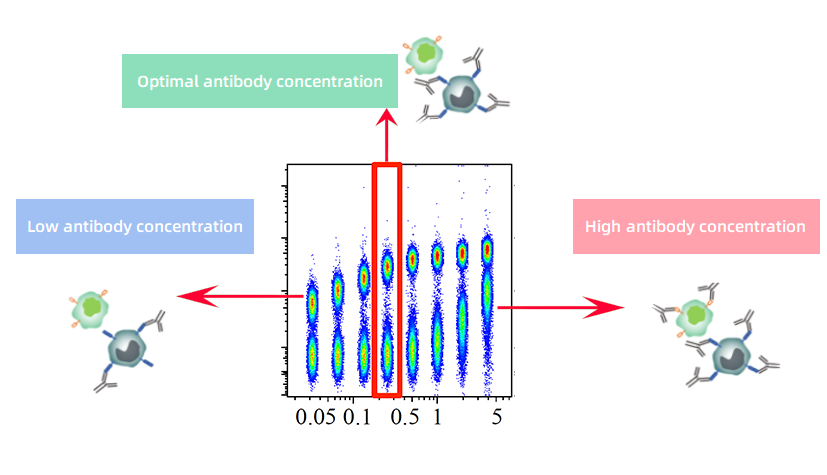
Figure 1. Principle of Flow Cytometry Antibody Titration
2. Workflow and Key Considerations for Antibody Titration
Step 1: Preparation – Set concentration gradients and pre-process samples
• Samples: Prepare at least 5×10⁵ cells per concentration point. Sample type and treatment should match the conditions of the intended experiment.
• Concentration gradient: Begin with the datasheet’s recommended concentration and prepare 5–6 serial dilutions covering suboptimal, optimal, and excess ranges.
• Pre-processing:
Centrifuge antibodies at 16,000–18,000 × g for 5 min at 4 °C to remove aggregates.
Perform Fc receptor blocking for samples with high Fc receptor expression.
For samples with reduced viability, perform live/dead staining first to exclude dead cells.
Step 2: Staining – Maintain consistent conditions
• Standardize staining volume and cell numbers. Add antibodies at designated concentrations and follow the established staining protocol.
• Include unstained and single-stained controls. Use these controls to adjust instrument settings. All titration samples should be acquired under identical instrument settings without further adjustment.
Step 3: Data analysis – Determine the optimal concentration
• Use the Staining Index (SI) to evaluate population separation:
SI=(MFIpositive−MFInegative)/2×SDnegative
This metric provides a more objective assessment of population resolution than simple signal-to-noise ratios.
• Optimal concentration: Choose the lowest concentration that achieves a stable SI plateau without increasing background in the negative population. In multicolor experiments, minimizing background is more critical than maximizing peak signal.
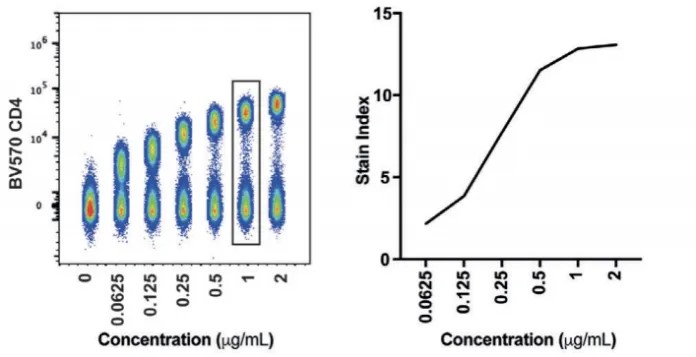
Figure 2. BV750-CD4 Titration Results
Notices:
• Consistency of the experimental system: Sample type, processing, cell status, and incubation conditions must match the intended experiment; otherwise, titration results are not representative.
• Focus on final antibody concentration: Record the working concentration rather than the dilution factor. Adjustments to staining volume should maintain the same final concentration.
• Fixed instrument settings: Acquire all titration samples under the same instrument parameters to ensure objective data analysis.
3. Practical Benefits of Proper Titration
1. Improved population resolution
Optimized antibody concentrations maximize signal separation between negative and positive populations, enabling accurate detection of low-expression antigens. This is particularly important for high-parameter multicolor panels.
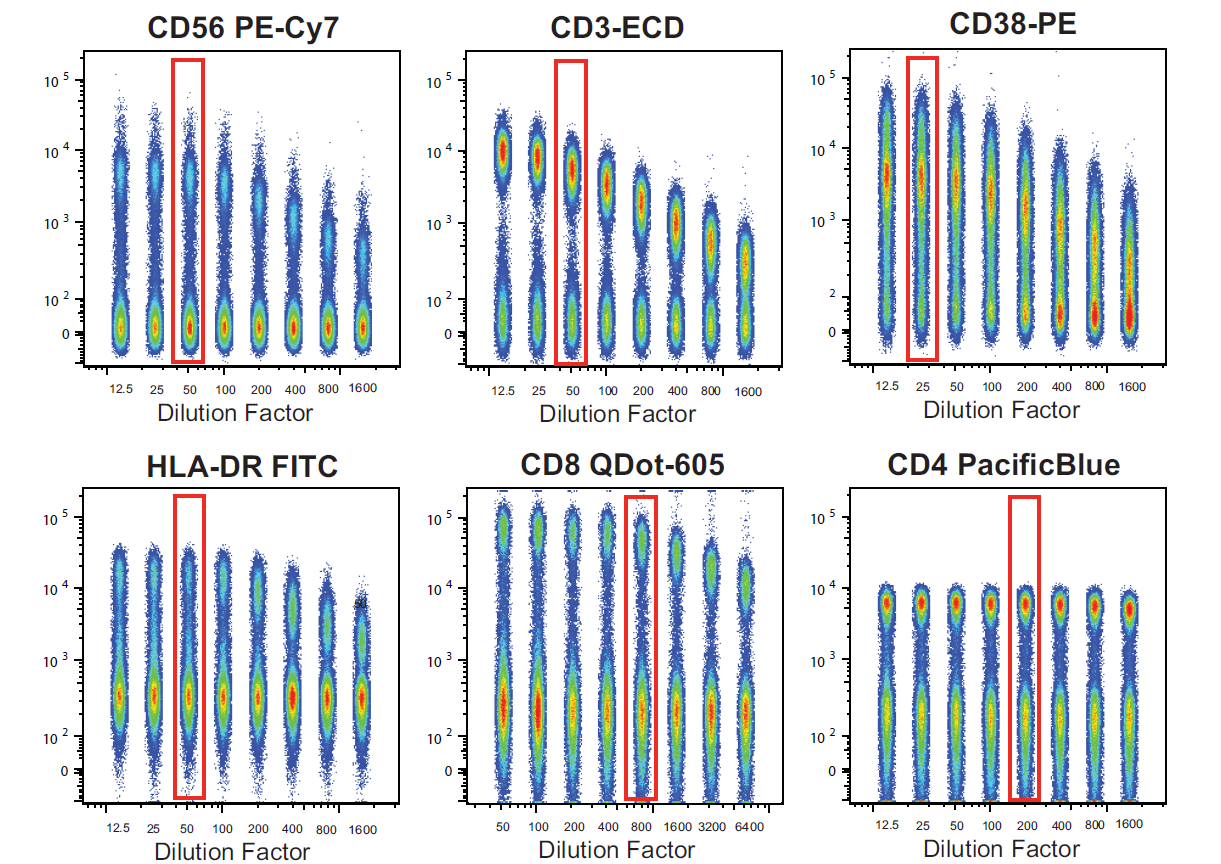
Figure 3. Comparison of Different Antibody Titration Outcomes
2. Reduced background and spillover
Excess antibody can lead to non-specific staining and increased fluorescence spillover, interfering with other detection channels. Titration reduces these artifacts and stabilizes multicolor experiments.
3. Cost efficiency
Titration identifies the minimal effective concentration required, minimizing reagent consumption while maintaining reliable results.
4. Enhanced reproducibility
Using validated titrated concentrations reduces variability between experimental batches and operators, ensuring more stable and reliable data.
4. Common Misconceptions About Antibody Titration
Misconception 1: Datasheet concentration is sufficient
Recommended concentrations are based on standardized test systems. Variations in sample type, processing, staining protocol, cell numbers, and instrument condition affect antibody performance. Therefore, datasheet concentrations serve as a starting point; the optimal concentration must be determined empirically.
Misconception 2: Titration is about achieving maximum fluorescence
Peak signal often reflects antibody excess, which increases background and diminishes population separation. Optimal titration identifies the lowest concentration that maximizes separation while maintaining low background.
Misconception 3: Focus solely on the dilution ratio rather than the final concentration
For example, a 1:200 dilution in a 200 μL system may be effective, but applying the same dilution to 1 mL increases the final concentration fivefold, leading to excessive staining and high background. The key is final concentration, not dilution ratio.
Misconception 4: Focus solely on maximum SI without considering background
Although SI indicates population separation, the point with the highest SI may coincide with significant background, causing interference in multicolor panels. The optimal choice is the first concentration in the SI plateau with minimal background.
Misconception 5: One titration is sufficient for all uses of the same catalog number antibody
Even antibodies from the same lot can vary in protein concentration or fluorophore conjugation efficiency. New lots, different sample types, or altered panel designs require re-titration to maintain reliable results.
Conclusion
Flow cytometry antibody titration determines the optimal concentration that balances specific and non-specific binding for a given experimental system. Proper titration enhances population resolution, reduces background interference, controls reagent consumption, and ensures reproducible and reliable data, forming a critical foundation for high-quality flow cytometry experiments.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience specializes in the development and production of high-quality life science reagents, and is committed to providing researchers with high-performance, highly reliable Flow Cytometry Antibodies, we offer:
1. Stringently validated flow cytometry antibodies including CD3, CD4, CD8 ensuring high sensitivity and minimal background noise;
2. A comprehensive portfolio of immunology research tools for multiple species, including Human, Mouse, Rat, Dog, Hamster;
3. Expert technical support and panel design consultation to help you accurately resolve complex immune cell populations.
For more information on abinScience flow cytometry antibodies, please visit:
abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






