Key Takeaways
Safety Signal: The clinical termination of Rocatinlimab underscores the critical correlation between OX40 blockade and compromised immune surveillance.
Inducible Regulation: As an inducible co-stimulatory receptor, OX40 serves as a late-stage molecular switch regulating T-cell survival and effector functions.
Therapeutic Potential: The OX40/OX40L axis remains a high-value target for bidirectional modulation in both Immuno-Oncology and Autoimmune diseases.
Translational Barriers: Future success hinges on resolving the spatiotemporal heterogeneity of OX40 and identifying validated predictive biomarkers.
On March 3, 2026, Kyowa Kirin officially announced the immediate termination of all global clinical trials for Rocatinlimab, an OX40-targeting monoclonal antibody. This followed a February 2026 decision by Amgen, the global co-development partner, to return all developmental rights to Kyowa Kirin citing a strategic reprioritization.
As the first OX40-blocking antibody to advance into multiple Phase III clinical trials, Rocatinlimab’s primary indications included moderate-to-severe atopic dermatitis, prurigo nodularis, and asthma. It was widely regarded by the industry as a leading blockbuster candidate in the immunology pipeline. According to Kyowa Kirin’s announcement, the rationale for termination was rooted in a critical safety signal identified during clinical review: an emerging risk of immunodeficiency-related malignancies. Following one previously reported case of Kaposi's sarcoma, two additional cases (one confirmed, one suspected) were identified. A joint safety assessment concluded that the potential risks to the target patient population outweighed the clinical benefits.
Kaposi's sarcoma is typically associated with compromised immune surveillance. This safety signal transcends a simple adverse drug reaction; it underscores a fundamental challenge in the field: significant knowledge gaps still regarding the basic biological mechanisms and the safety boundaries of OX40-mediated immune regulation.
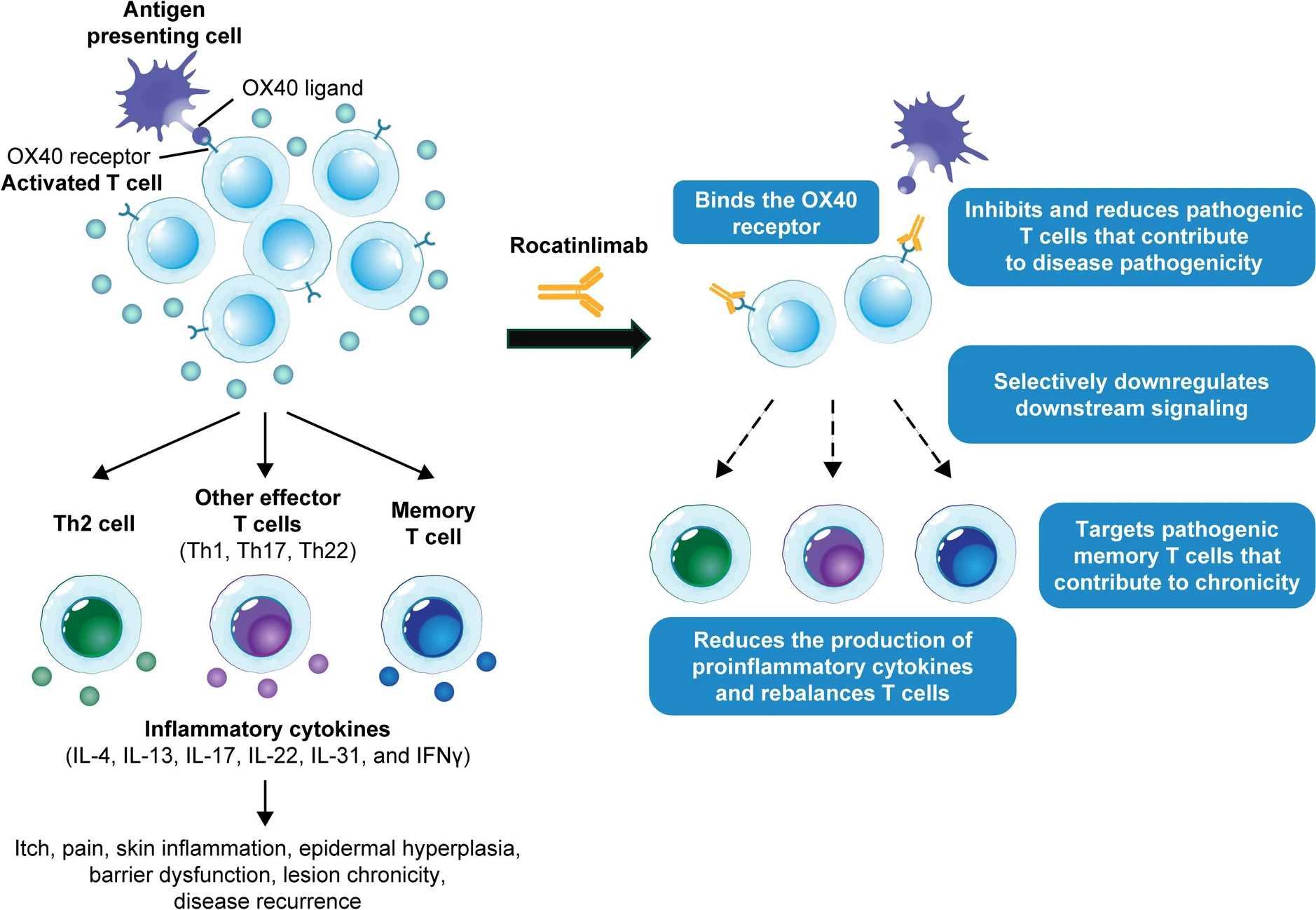
Figure 1. Rocatinlimab mechanism of action
1. Biological Foundation and Translational Value of the OX40 Target
OX40 (CD134), encoded by the TNFRSF4 gene, is a member of the tumor necrosis factor receptor superfamily (TNFRSF). Functioning as a critical late-stage co-stimulatory molecule, OX40 signaling governs the magnitude, duration, and homeostasis of the T-cell immune response.
Human OX40 is a type I transmembrane glycoprotein characterized by an extracellular cysteine-rich domain (CRD) that mediates ligand binding, and an intracellular domain that initiates downstream signaling through the recruitment of TNF receptor-associated factor (TRAF) proteins.
Crucially, OX40 expression is highly inducible and cell-type specific, distinguishing it from the constitutive expression of CD28. OX40 is notably absent on resting T cells; its expression is upregulated only 24–72 hours following initial T-cell activation via TCR and CD28 co-stimulation. OX40 is predominantly expressed on activated CD4+ helper T cells, CD8+ cytotoxic T cells, and regulatory T cells (Tregs). In pathological states, such as the tumor microenvironment or autoimmune lesions, OX40 expression is significantly elevated, correlating with the state of T-cell activation.
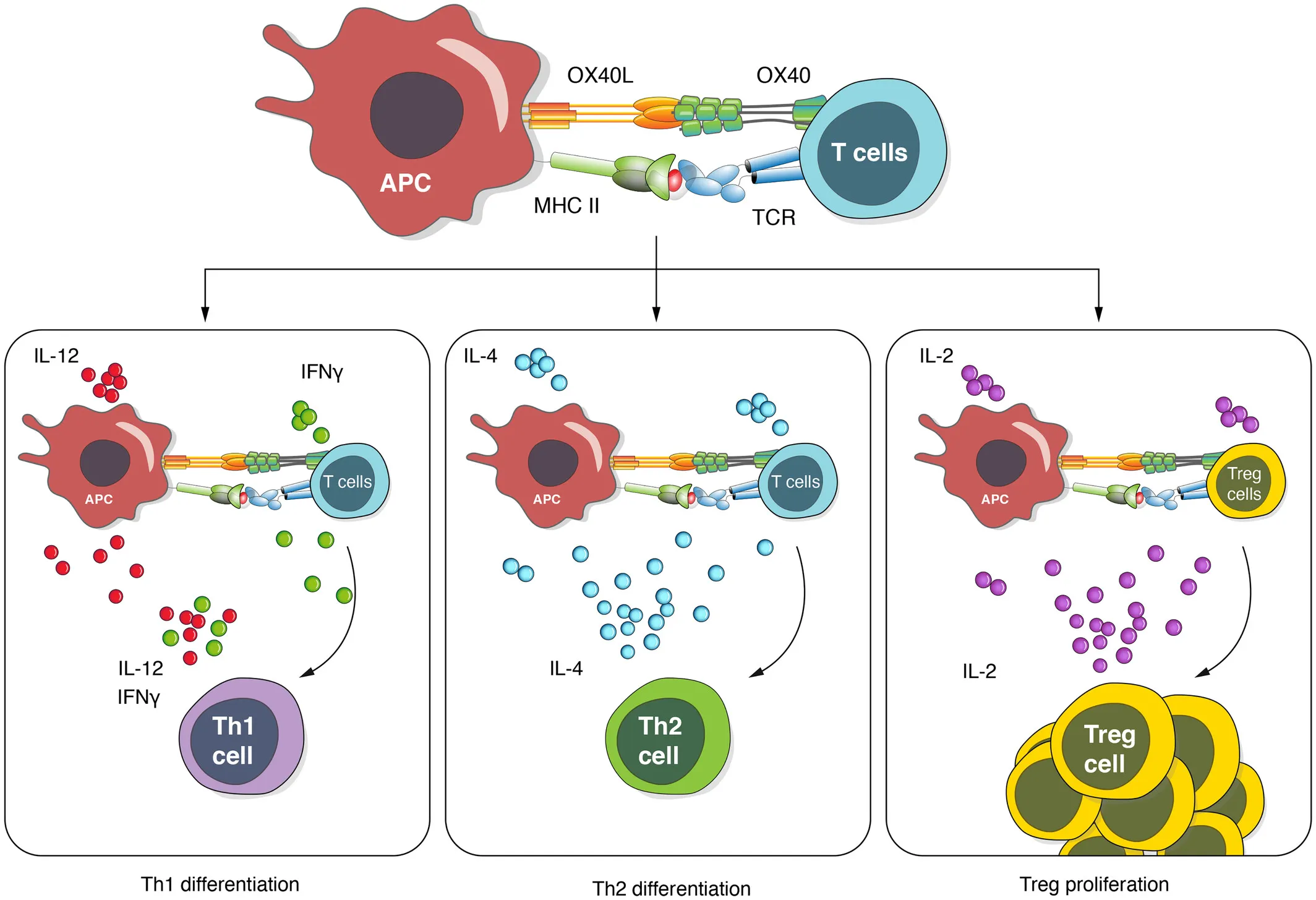
Figure 2. OX40-OX40L Signalling Modulates T Cell Proliferation and Polarization
Upon activation, OX40 signaling maintains prolonged T-cell survival and prevents activation-induced cell death (AICD) by upregulating anti-apoptotic proteins. This mechanism allows for the expansion of the effector T-cell pool and enhances cytokine production. Consequently, OX40 represents a high-value target for bidirectional therapeutic modulation:
• Immuno-Oncology: Agonistic antibodies can augment anti-tumor immunity and reverse T-cell exhaustion, offering synergistic potential with PD-1/PD-L1 inhibitors.
• Autoimmunity: Antagonistic antibodies can suppress pathogenic immune responses by blocking excessive OX40 signaling.
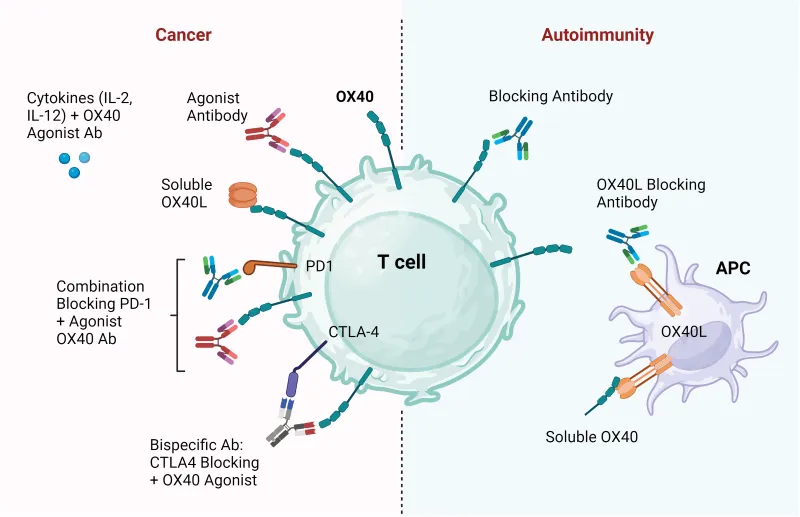
Figure 3. Therapeutic strategies to target OX40OX40L in cancer and autoimmunity.
2. Clinical Setbacks and the Imperative for Mechanistic Research
The clinical termination of Rocatinlimab does not invalidate the biological significance of OX40. Instead, it serves as a critical reminder that successful clinical translation is contingent upon a comprehensive understanding of target biology. The current translational bottlenecks in OX40 development are largely attributed to unresolved mechanistic questions, which the Rocatinlimab data has brought into sharp focus:
Regulation of Immune Homeostasis and Safety Boundaries
OX40 signaling exerts divergent effects on effector T cells versus Treg cells. While activation of effector T cells promotes anti-tumor and anti-infective immunity, the role of OX40 in Tregs is vital for maintaining peripheral tolerance. The field lacks long-term, quantitative data regarding the impact of sustained OX40 blockade on immune homeostasis. Specifically, it remains unclear whether chronic inhibition compromises Treg-mediated surveillance, thereby increasing the risk of latent viral reactivation or tumorigenesis—as exemplified by the Kaposi’s sarcoma cases in the Rocatinlimab trials.
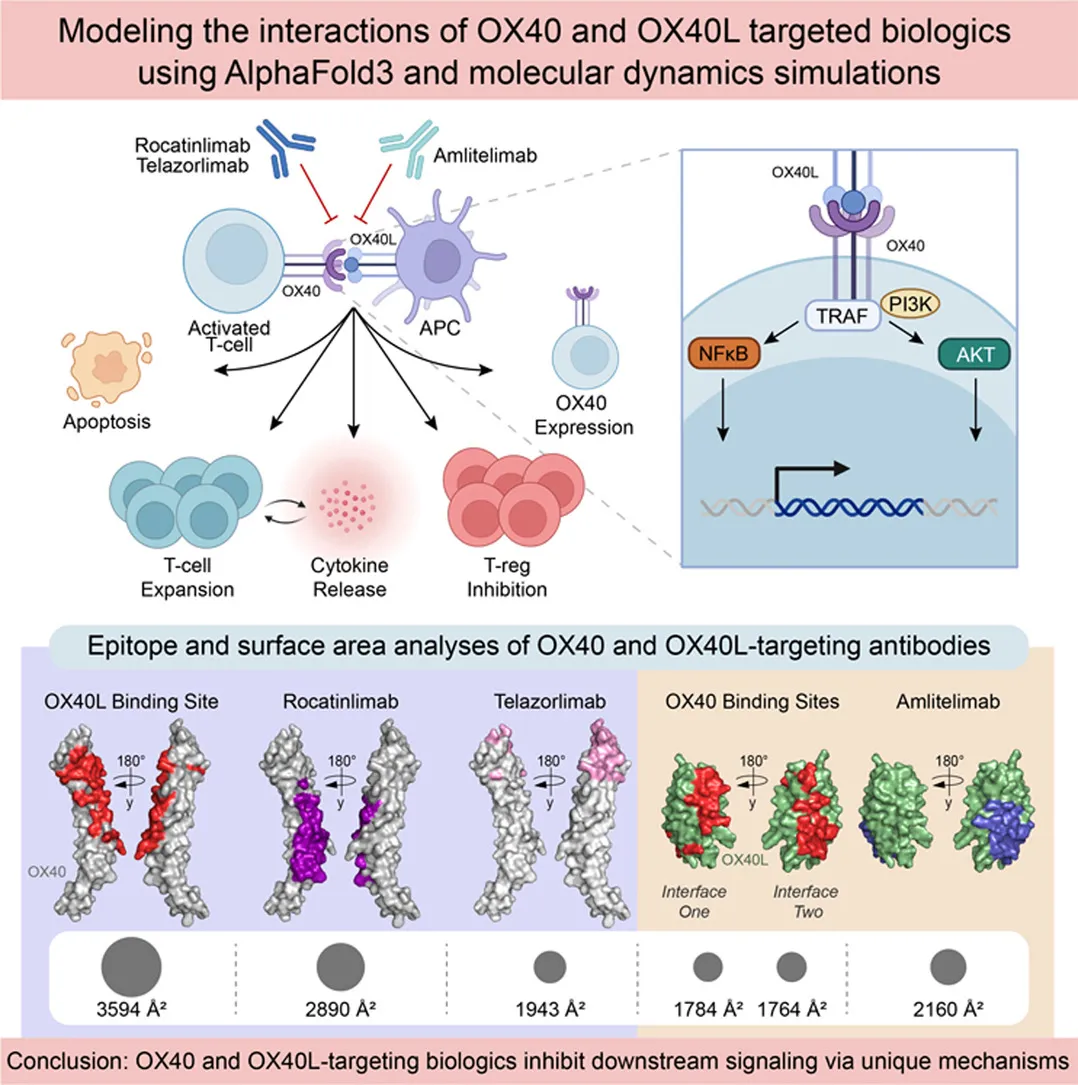
Figure 4. Divergent Effects of OX40 Signaling on Effector T Cells vs. Tregs
Spatiotemporal Heterogeneity in Target Expression
OX40 expression is highly dynamic and context-dependent, exhibiting significant heterogeneity across different disease stages, tissue microenvironments, and patient populations. Current therapeutic strategies often rely on systemic blockade; however, a lack of resolution regarding the precise distribution and function of OX40 across various tissues complicates the ability to achieve targeted regulation without disrupting global immune balance.
Structure-Activity Relationships (SAR) and In Vivo Efficacy
Rocatinlimab is an OX40-blocking antibody with antibody-dependent cellular cytotoxicity (ADCC) activity. Its in vivo efficacy is governed by an intricate interplay of binding epitopes, affinity, and Fc-mediated effector functions. There is currently no established consensus on how these molecular design parameters translate to clinical outcomes, hindering the rational design of next-generation OX40 targeted therapies.
Deficiency in Predictive Biomarker Systems
Precision medicine requires robust patient stratification, yet the OX40 field lacks validated predictive biomarkers. Mechanistically, it is not yet clear which patient subsets are primarily driven by OX40 dysregulation. Furthermore, there is a lack of warning biomarkers to screen for pre-existing immunodeficiencies or to monitor real-time immune function during treatment.
3. Future Directions: A Return to Foundational Research
The Rocatinlimab clinical halt underscores the necessity of returning to basic mechanistic research to bridge existing translational gaps. Future developments in the OX40 landscape will prioritize:
• High-Resolution Mapping: Utilizing single-cell sequencing, spatial transcriptomics, and mass cytometry (CyTOF) to elucidate the expression profiles and functional heterogeneity of OX40 across diverse tissues and disease states.
• Rational Molecular Design: Leveraging structural biology (e.g., cryo-EM) to decipher the SAR of antibody-target interactions.
• Homeostatic Quantification: Investigating the regulatory role of OX40 in immune surveillance to define safe therapeutic windows and dosing regimens.
• Biomarker Discovery: Integrating multi-omics data to identify biomarkers for efficacy prediction and risk mitigation.
As a master regulator of the immune response, the translational value of OX40 remains significant. The termination of Rocatinlimab marks a pivot point toward more rigorous, mechanistically-driven development. Unlocking the therapeutic potential of OX40 will require addressing these core scientific questions through high-quality foundational research.
4. abinScience Products for OX40 Research
To help researchers address these specific knowledge gaps, abinScience offers a comprehensive, high-quality suite of OX40 research tools. Optimized for applications spanning Western Blot (WB), IHC, ELISA, Flow Cytometry, and in vivo assays, our portfolio provides end-to-end support for protein detection, functional analysis, and preclinical validation.
Recombinant proteins
| Product Name |
Catalog No. |
| Recombinant Red fox CD134/TNFRSF4/OX40 Protein, C-His |
ZW342012 |
| Recombinant Dog CD134/TNFRSF4/OX40 Protein, C-Fc |
CW342012 |
| Recombinant Mouse CD134/TNFRSF4/OX40 Protein, N-His |
MW342012 |
| Recombinant Human CD134/TNFRSF4/OX40 Protein, C-His |
HW342011 |
| Recombinant Mouse CD134/TNFRSF4/OX40 Protein, C-Fc |
MW342011 |
| Recombinant Human CD252/TNFSF4/OX40L Protein, C-His |
HB937021 |
| Recombinant Human CD252/TNFSF4 Protein, N-His |
HB937012 |
| Recombinant Human CD252/TNFSF4 Protein, N-Fc |
HB937011 |
Antibodies
| Product Name |
Catalog No. |
| Research Grade Anti-Human CD134/TNFRSF4/OX40 (INCAGN1949) |
HW342086 |
| Research Grade Anti-Human CD134/TNFRSF4/OX40 (BAT6026) |
HW342096 |
| Research Grade Anti-Human CD134/TNFRSF4/OX40 (BMS 986178) |
HW342106 |
| Research Grade Anti-Human CD134/TNFRSF4/OX40 (INBRX-106) |
HW342126 |
| Research Grade Rocatinlimab |
HW342076 |
| Research Grade Gimistotug |
HW342196 |
| Research Grade Ivuxolimab |
HW342016 |
| Research Grade Pogalizumab |
HW342026 |
| Anti-Human CD134/TNFRSF4/OX40 Antibody (SAA2011) |
HW342013 |
| Anti-Human CD134/TNFRSF4/OX40 Antibody (3C8) |
HW342307 |
| Anti-Human CD134/TNFRSF4/OX40 Monoclonal Antibody (1A641) |
HW342035 |
| Anti-Mouse CD134/TNFRSF4/OX40 Antibody (OX-86) |
MW342107 |
| InVivoMAb Anti-Mouse PD-L1 & OX40 Bispecific Antibody (Iv0244) |
MW342010 |
| InVivoMAb Anti-Mouse CTLA4 & OX40 Bispecific Antibody |
MB651040 |
References
[1] Fu Y, Lin Q, Zhang Z, Zhang L. Therapeutic strategies for the costimulatory molecule OX40 in T-cell-mediated immunity. Acta Pharm Sin B. 2020 Mar;10(3):414-433. doi: 10.1016/j.apsb.2019.08.010. Epub 2019 Sep 3. PMID: 32140389; PMCID: PMC7049610.
[2] Croft M. Control of immunity by the TNFR-related molecule OX40 (CD134). Annu Rev Immunol. 2010;28:57-78. doi: 10.1146/annurev-immunol-030409-101243. PMID: 20307208; PMCID: PMC2882161.
[3] Willoughby J, Griffiths J, Tews I, Cragg MS. OX40: Structure and function - What questions remain? Mol Immunol. 2017 Mar;83:13-22. doi: 10.1016/j.molimm.2017.01.006. Epub 2017 Jan 13. PMID: 28092803.
[4] Luo Y, Li J, Li L, Qin B, Zhou R, Tang Y. OX40 signaling in cancer immunotherapy: mechanisms of action, translational applications, and therapeutic perspectives. Front Immunol. 2026 Feb 9;17:1724756. doi: 10.3389/fimmu.2026.1724756. PMID: 41737211; PMCID: PMC12926375.
[5] Gupta AK, Steriopoulos J, Economopoulos V, Mirmirani P, Piguet V. OX40/OX40L modulation: A target for regulating T cells in cutaneous inflammatory disorders. J Eur Acad Dermatol Venereol. 2026 Apr 11. doi: 10.1111/jdv.70443. Epub ahead of print. PMID: 41964298.
[6] Nolden K, Shi Y, Batista VS, Bunick CG. Molecular differentiation of OX40- and OX40L-targeted biologics using AlphaFold3 and molecular dynamics simulations. J Invest Dermatol. 2026 Feb 4:S0022-202X(26)00073-4. doi: 10.1016/j.jid.2026.01.024. Epub ahead of print. PMID: 41651107; PMCID: PMC13047499.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






