Key Takeaways
• Biosimilars are entering a new predictable growth phase
• Autoimmune diseases remain a dominant biologics R&D area
• Several next-generation biologics targeting BAFF, OX40 and IL-36 pathways are approaching key milestones
• Reliable research reagents remain essential for biologics discovery and validati
With ongoing breakthroughs in biotechnology, innovative biologics—including monoclonal antibodies, fusion proteins, and gene therapies are profoundly reshaping the treatment landscape. At the same time, biosimilars—biotherapeutics that are highly similar to approved reference biotherapeutics in key quality attributes including amino acid sequence, higher-order structure, biological activity, safety, and efficacy—have become an indispensable component of the global biopharmaceutical ecosystem.
1. Biopharma Industry Upgrading: Expansion of the Biosimilar Market
Globally, biologics continue to account for an increasing share of new drug approvals. Over the past several years, the proportion of biologics among FDA/CDER-approved drugs has steadily risen, a trend projected to continue over the next decade. Industry analyses suggest that by 2030, biologics and gene therapies could represent over 60% of newly approved therapeutics.
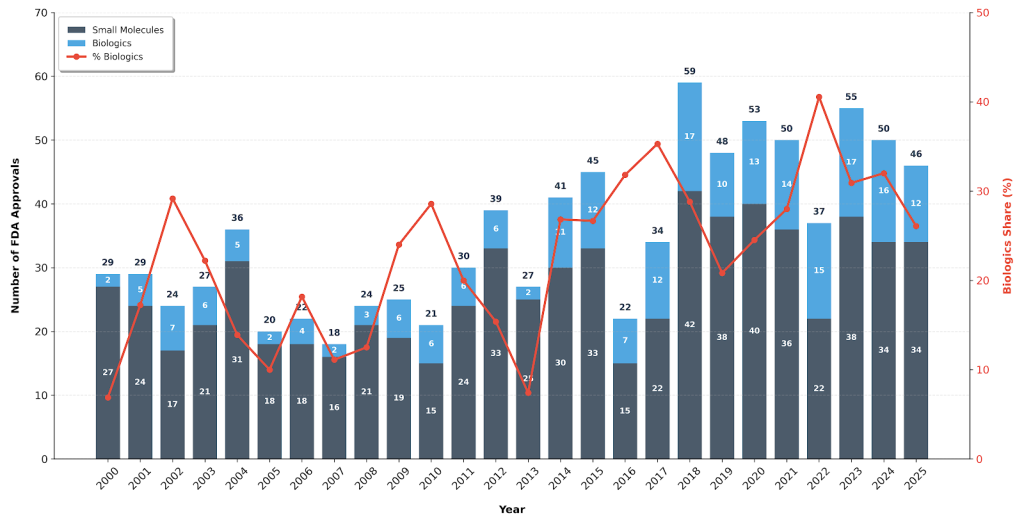
Figure 1. The number of drugs approved in the U.S. by CDER by molecule type and the percentage shared by biologics (2000-2025).
Alongside market expansion, numerous blockbuster monoclonal antibodies and fusion proteins are approaching patent cliffs. This not only breaks the monopoly of reference drugs, but also accelerates the R&D and commercialization of biosimilars. Large-scale biosimilar development significantly cuts the clinical cost of biologics, improves patient access to advanced therapies, and provides a feasible entry point for pharmaceutical companies into biologics R&D. It further drives the upgrade of upstream and downstream production processes, quality control systems, and the entire industry ecosystem.
2. 2026 Biologics R&D Trends: Immunology and Autoimmune Diseases at the Forefront
Recent industry forecasts indicate that immunomodulation will dominate next-generation biologics development in 2026. Over half of the upcoming approvals target autoimmune and inflammatory disorders such as Sjögren’s syndrome, atopic dermatitis, generalized pustular psoriasis (GPP), and IgA nephropathy. These chronic progressive diseases have well-defined immune dysregulation mechanisms, presenting high-value targets for biologics development.
Current research focuses on four key immune regulatory pathways that act on adaptive immune system nodes, enabling precise disease intervention and representing prime targets for future biosimilar development:
BAFF/BAFF-R pathway: Regulates B cell survival and differentiation, pivotal in multiple autoimmune diseases
OX40 co-stimulatory pathway: Involved in T cell activation and immune memory formation
IL-36 inflammatory pathway: Key in psoriasis and other inflammatory skin disorders
BAFF/APRIL dual pathway: Governs antibody production and B cell maturation
As these innovative biologics progress toward clinical application, corresponding biosimilar development will be a central focus over the next decade.
3. Key Autoimmune Biologics to Watch in 2026
Multiple biologics targeting key immune regulatory pathways are approaching important clinical milestones. These candidates focus on B cell regulation, T cell co-stimulation, and inflammatory cytokine signaling—mechanisms central to many autoimmune diseases.
The table below summarizes several biologics drawing significant attention in 2026 clinical pipelines.
| Drug |
Target |
Mechanism |
Key Indications |
| Ianalumab |
BAFF-R |
Direct BAFF-R blockade with ADCC-mediated B cell depletion |
Sjögren’s syndrome, ITP |
| Rocatinlimab (AMG-451) |
OX40 |
Inhibits T cell co-stimulatory signaling |
Atopic dermatitis |
| Imsidolimab |
IL-36R |
Blocks IL-36 inflammatory signaling |
Generalized pustular psoriasis (GPP) |
| Atacicept |
BAFF + APRIL |
Dual inhibition of B cell survival pathways |
IgA nephropathy and other antibody-driven diseases |
3.1 Ianalumab: BAFF-R Targeted B Cell Modulation
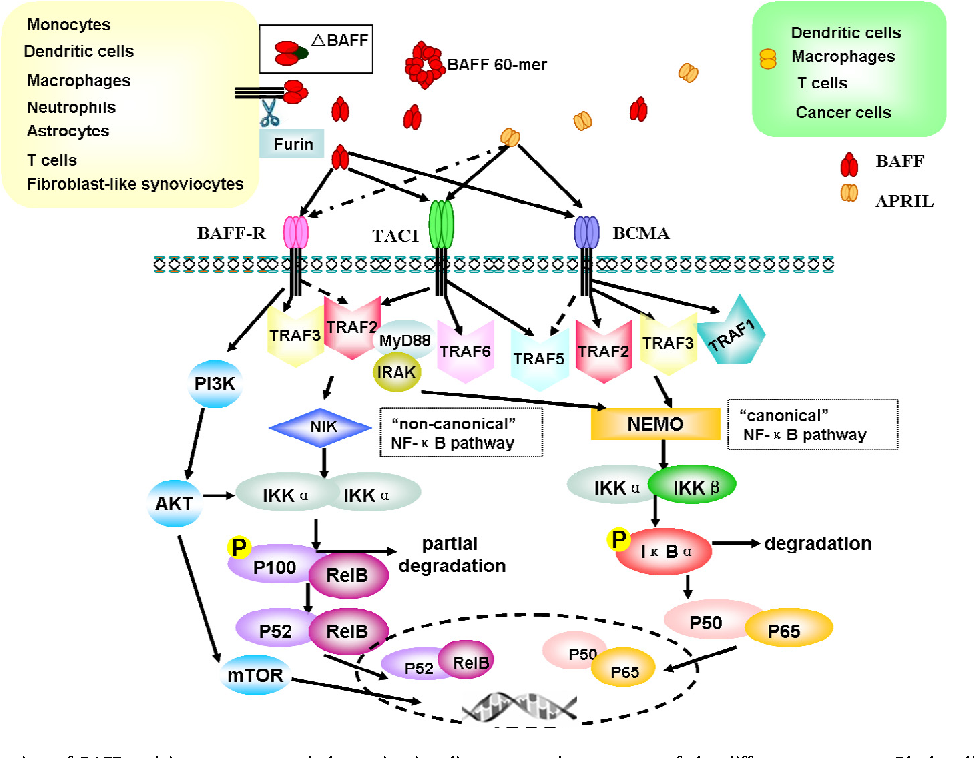
Figure 2. Interaction and expression of BAFF.(DOI: 10.1016/j.cyto.2015.07.014)
Developed by Novartis, Ianalumab targets the BAFF receptor (BAFF-R), a key regulator of B cell survival and differentiation. Unlike therapies that only block the BAFF ligand, it directly inhibits BAFF-R while inducing ADCC-mediated depletion of pathogenic B cells. It met primary endpoints in Phase III trials for Sjögren’s syndrome, with additional studies showing benefits in immune thrombocytopenia (ITP), positioning it as a potential first-in-class BAFF-R-targeted therapy.
3.2 Rocatinlimab: T Cell Immunomodulation via OX40
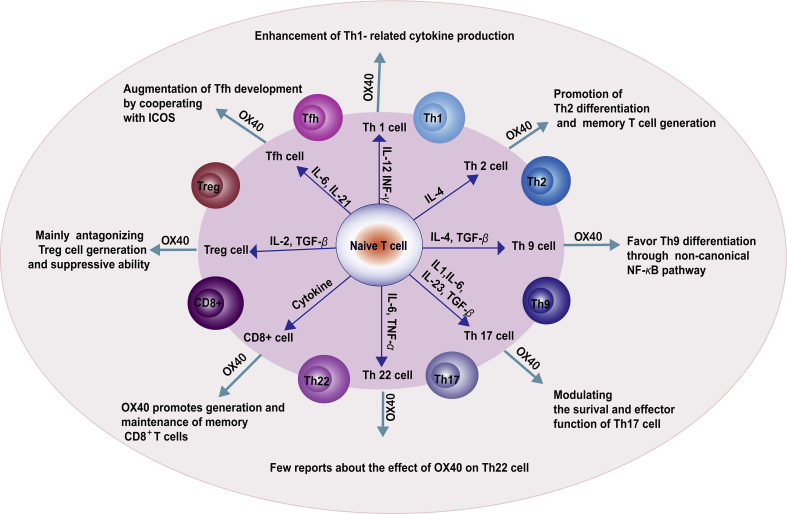
Figure 3. The summary of the impact of the OX40–OX40L interaction on T cell subsets.(Doi: 10.1016/j.apsb.2019.08.010)
Co-developed by Amgen and Kyowa Kirin, Rocatinlimab (AMG-451) targets the T cell co-stimulatory receptor OX40, which sustains pathogenic T cell responses and immune memory. By blocking OX40 signaling, it modulates upstream immune activation. Its Phase III ROCKET IGNITE trial for atopic dermatitis met primary endpoints, delivering meaningful lesion clearance and long-term remission in some patients.
3.3 Imsidolimab: IL-36 Pathway Inhibition
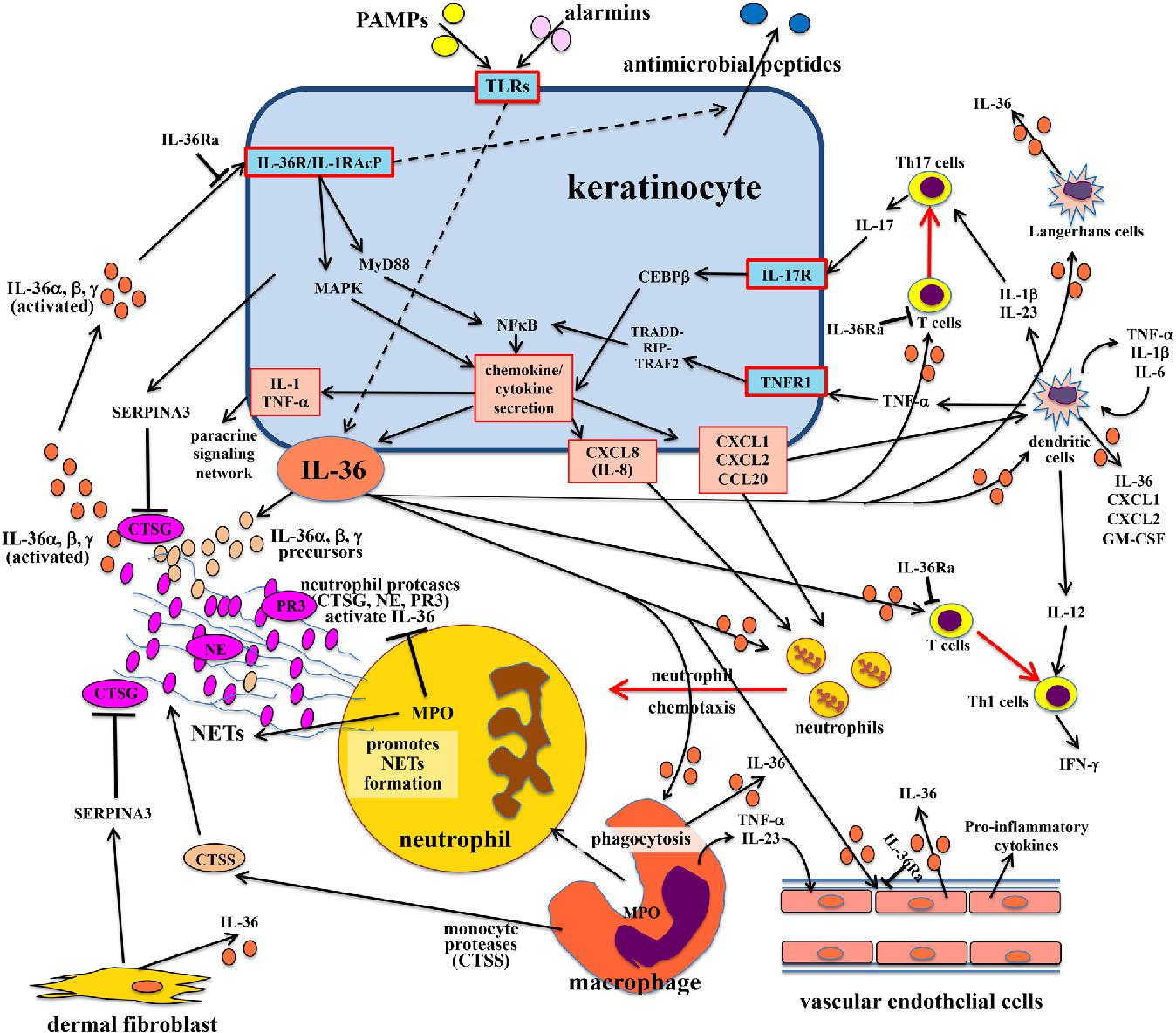
Figure 4. Inflammatory pathways and factors involved in IL-36-associated inflammation in the skin.(DOI: 10.1007/s40259-023-00587-5)
Imsidolimab targets the IL-36 receptor, the critical driver of inflammation in GPP. IL-36 signaling amplifies inflammatory crosstalk between keratinocytes and immune cells, leading to severe neutrophil-mediated skin inflammation. In the Phase III GEMINI-1 study, it achieved rapid and substantial disease control, highlighting its potential as a targeted therapy for this rare and severe condition.
3.4 Atacicept: Dual BAFF/APRIL Targeting
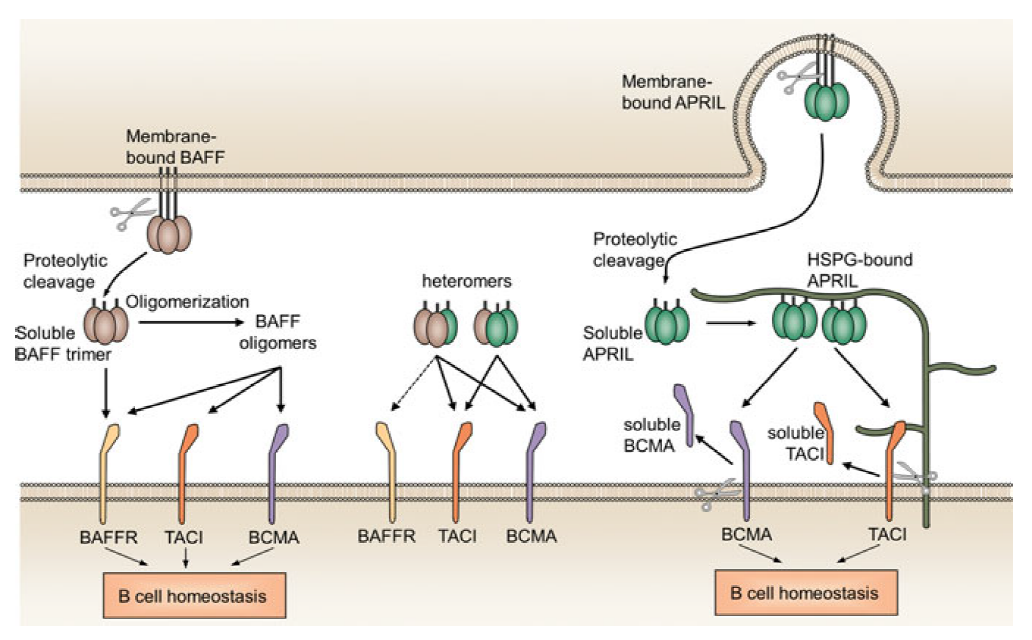
Figure 5. BAFF and APRIL receptor interactions.(DOI: 10.1080/08830185.2016.1276903)
Atacicept is a TACI-Fc fusion protein that simultaneously neutralizes BAFF and APRIL, two key survival factors for B cells and plasma cells. This dual-pathway inhibition enables broader suppression of antibody-mediated immune responses. Clinical studies in IgA nephropathy have shown significant reductions in proteinuria and improved renal biomarkers, demonstrating promising therapeutic potential.
4. The Value of Biosimilars in Industry and Research
With the global patent cliff for blockbuster biologics, biosimilars have become a core growth engine for the pharmaceutical market, delivering value not only in clinical and commercial domains but also as indispensable research tools.
4.1 Industrial and Clinical Value
Improved patient access: Biosimilars reduce treatment costs, increasing availability of high-end biologics and easing financial pressure on healthcare systems and patients.
Technology advancement: Biosimilar R&D demands advanced cell culture, protein purification, and quality control technologies. In turn, this drives technological advancement across the biologics supply chain and fosters the development of local manufacturing capabilities.
Ecosystem development: Biosimilars provide an entry pathway for companies worldwide, cultivating skilled professionals in R&D, manufacturing, and quality assurance, and laying a foundation for the next generation of innovative biologics.
4.2 Research Applications
Biosimilars have become essential research reagents, supporting mechanistic studies, target validation, in vitro and in vivo pharmacology, and bioanalytical method development, providing consistent and reliable materials for biologics innovation.
5. abinScience Research Biosimilars
abinScience offers a comprehensive range of research-grade biosimilars, enabling mechanistic studies, target validation, and preclinical exploration across multiple disease areas.
| Product Name |
Catalog No. |
| Research Grade Tividenofusp Alfa |
HY036116 |
| Research Grade Ianalumab |
HV599016 |
| Research Grade Atacicept |
HF941036 |
| Research Grade Rocatinlimab |
HW342076 |
References
[1] Tuszyner, A. (2026, March 2). The 2026 biologics wave: Which innovations will break through? Contract Pharma. https://www.contractpharma.com/the-2026-biologics-wave-which-innovations-will-break-through/
[2] Wei F, Chang Y, Wei W. The role of BAFF in the progression of rheumatoid arthritis. Cytokine. 2015 Dec;76(2):537-544. doi: 10.1016/j.cyto.2015.07.014. Epub 2015 Jul 18. PMID: 26198030.
[3] Fu Y, Lin Q, Zhang Z, Zhang L. Therapeutic strategies for the costimulatory molecule OX40 in T-cell-mediated immunity. Acta Pharm Sin B. 2020 Mar;10(3):414-433. doi: 10.1016/j.apsb.2019.08.010. Epub 2019 Sep 3. PMID: 32140389; PMCID: PMC7049610.
[4] Fukaura R, Akiyama M. Targeting IL-36 in Inflammatory Skin Diseases. BioDrugs. 2023 May;37(3):279-293. doi: 10.1007/s40259-023-00587-5. Epub 2023 Mar 3. PMID: 36867370.
[5] Samy E, Wax S, Huard B, Hess H, Schneider P. Targeting BAFF and APRIL in systemic lupus erythematosus and other antibody-associated diseases. Int Rev Immunol. 2017 Jan 2;36(1):3-19. doi: 10.1080/08830185.2016.1276903. PMID: 28215100.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский







