In our previous articles: Single-Stain Controls: The Hidden Driving Force Behind Compensation Matrix Stability and Decoding Basic Terminology of Flow Cytometry: Rapid Comprehension and Practical Application, as well as in widely accepted experimental guidelines, fluorescence compensation has consistently been emphasized as a critical step in multicolor flow cytometry experiments, directly impacting data accuracy.
1. The Essence of Compensation: Deconvolution of Mixed Signals
Upon excitation, fluorochromes emit light over a broad spectrum rather than at a single wavelength. As a result, emitted signals are not confined to their primary detection channels but also spill over into adjacent channels.
For example, in a FITC–PE combination, approximately 15% of the FITC emission may be detected in the PE channel. Consequently, the signal measured in the PE channel represents a composite of true PE fluorescence and FITC spillover.
The purpose of compensation is to mathematically subtract the FITC-derived contribution from the PE channel, thereby restoring the true PE signal. Similarly, spillover from PE into the FITC channel must also be corrected.
In this sense, compensation can be understood as a signal deconvolution process—separating the measured composite signal into the independent contributions of each fluorochrome.
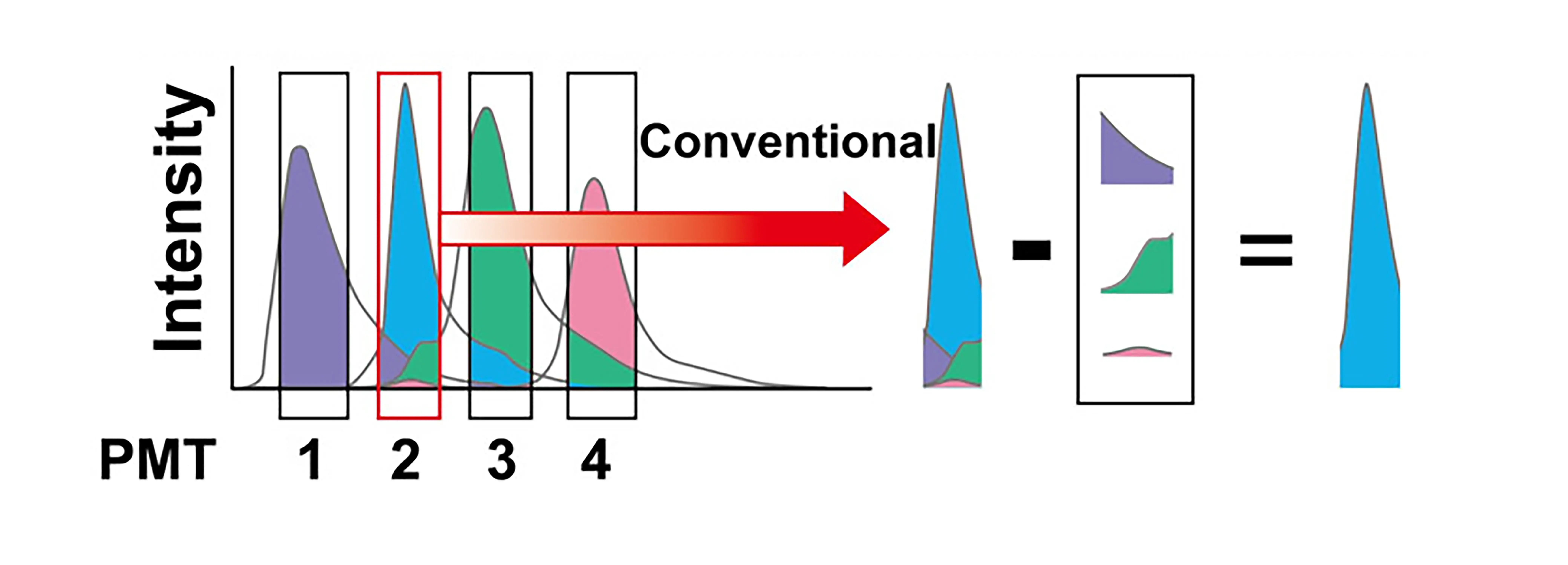
Figure 1. Principle of fluorescence compensation: deconvolution of mixed signals across detection channels
2. Why Compensation is Unavoidable: Intrinsic Properties of Fluorochromes and Instrumentation
Compensation is an inherent requirement in multicolor flow cytometry, arising from both the physical properties of fluorochromes and the signal detection mechanism of flow cytometers.
2.1 Broad Emission Spectra of Fluorochromes
All fluorochromes exhibit continuous, broad emission spectra. There is no such thing as a truly “monochromatic” fluorophore. Therefore, whenever two or more fluorochromes are used simultaneously, spectral overlap is inevitable. This is an intrinsic physical property and is independent of instrument performance or operator technique.
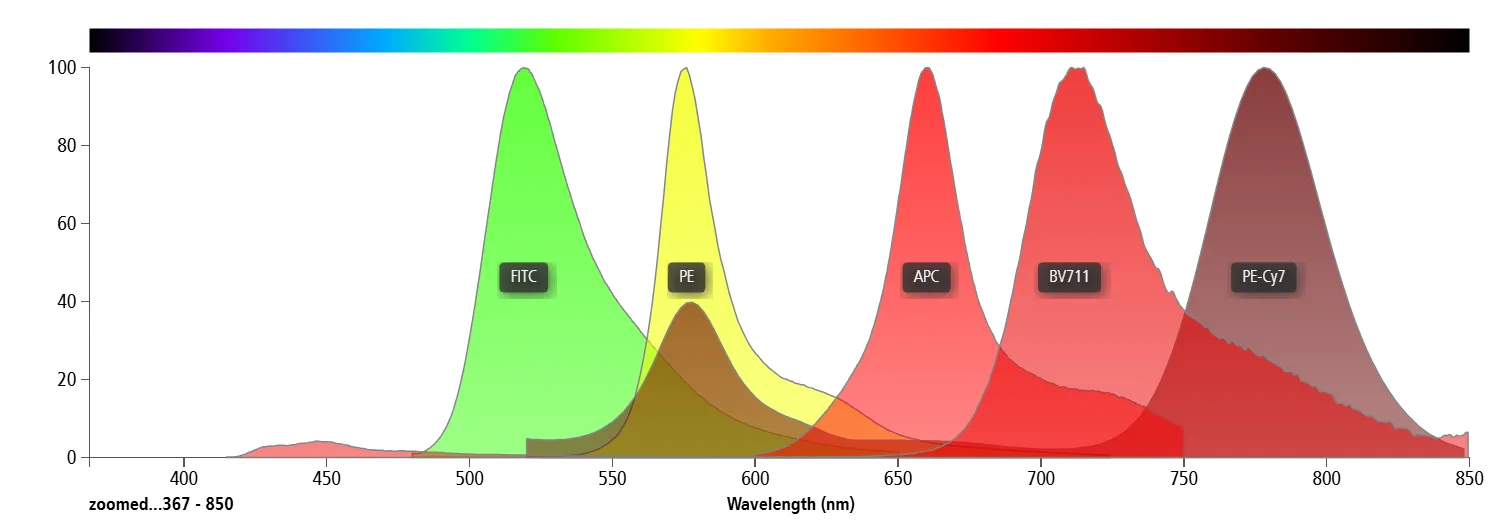
Figure 2. Spectral overlap between fluorochromes
2.2 Signal Acquisition via Bandpass Filters
Flow cytometers use bandpass filters to collect fluorescence signals within defined wavelength ranges rather than at a single wavelength. This design enhances sensitivity, particularly for low-abundance antigens.
Because fluorescence channels collect signals within defined wavelength ranges rather than at a single wavelength, overlapping spectral components are inherently captured and amplified together, thereby exacerbating signal mixing between channels.
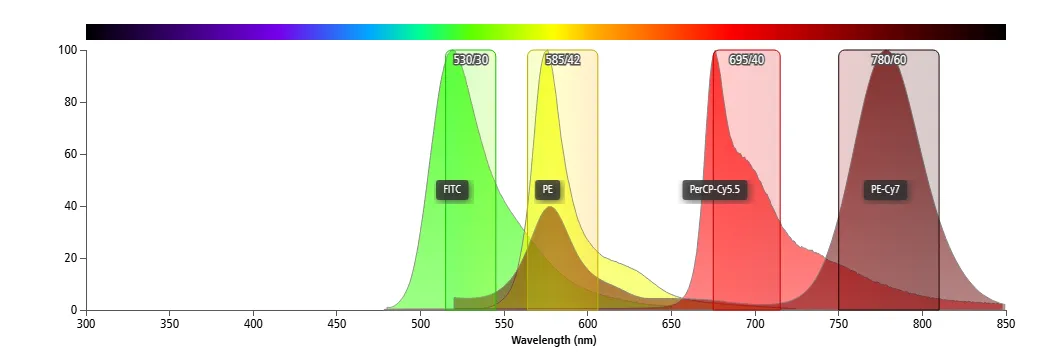
Figure 3. Fluorescence detection using bandpass filters in flow cytometry
Therefore, spectral overlap and signal mixing cannot be eliminated through hardware optimization alone and must be corrected through proper compensation.
3. Key Practices for Accurate Compensation
3.1 Preparation of High-Quality Single-Stained Controls
Single-stained controls serve as the reference standard for compensation calculation. Their quality directly determines compensation accuracy.
(1) Consistent treatment with experimental samples
Single-stained controls must undergo identical processing as the experimental samples, including:
• Fixation and permeabilization steps
• Incubation time before acquisition
• Use of identical staining buffers
(2) Clearly distinguishable positive populations
Accurate compensation relies on a clear separation between negative and positive populations. Weak or poorly resolved signals compromise spillover estimation.
Positive populations should be sufficiently bright and well separated from negatives, though excessively high intensity is not required.
(3) Proper use of compensation beads
When sample availability is limited or antigen expression is low, compensation beads provide an effective alternative.
These beads typically consist of negative and positive populations, with the latter coated with anti-immunoglobulin antibodies (e.g., anti-mouse IgG), enabling strong and uniform signal generation upon binding to fluorochrome-conjugated antibodies.
Key considerations include:
• Matching fluorochromes with target antibodies
• Ensuring robust positive signal
• Strict adherence to manufacturer protocols
Failure to account for differences between beads and cells may introduce compensation bias.
3.2 Voltage Setting Must Precede Compensation
Detector voltage determines signal amplification, while compensation values are calculated based on spillover measured under specific voltage settings.
Any change in voltage after compensation invalidates the compensation matrix.
Recommended workflow:
• Adjust PMT voltages using unstained samples
• Lock voltage settings
• Acquire single-stained controls and fully stained samples
Strict adherence to this sequence is essential for reliable compensation.
3.3 When to Use Automatic vs Manual Compensation
Modern flow cytometry software can generate accurate compensation matrices automatically, provided that single-stained controls are of high quality.
However, manual adjustment may be required in specific scenarios:
• Use of tandem dyes (e.g., PE-Cy7, APC-Cy7), which are prone to degradation and variability
• Weak antigen expression resulting in suboptimal single-stained signals
In high-parameter panels, compensation should always be verified post-calculation, typically using N×N plots to assess both single-stained and fully stained samples.
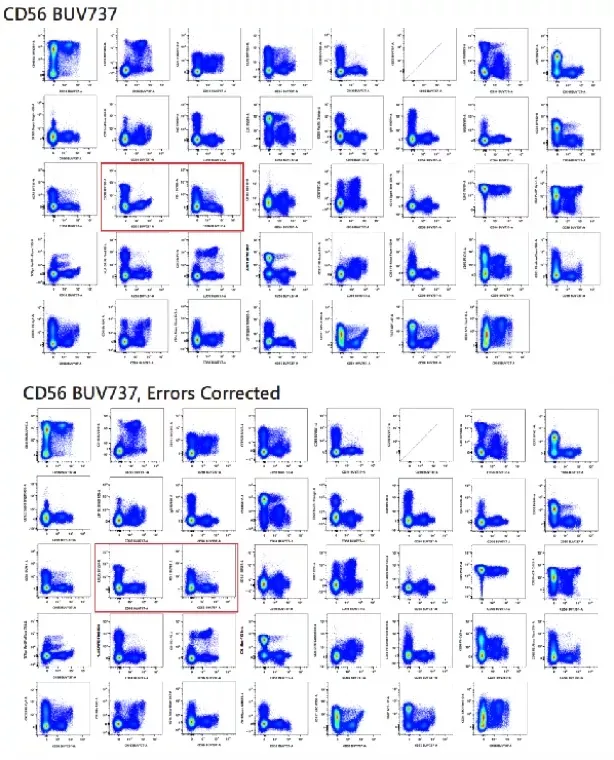
Figure 4. Post-compensation evaluation and manual refinement using N×N plots
4. Common Issues in Compensation
4.1 Reusability of Compensation Matrices: Strict Conditions Apply
Compensation values can only be reused under highly controlled conditions:
• Identical antibody panel (including fluorochromes, clones, and lot numbers)
• Unchanged PMT voltage settings across all channels
• Identical sample processing (fixation, permeabilization, buffers, incubation)
• Same instrument with no hardware changes or optical realignment
Even under these conditions, reuse beyond one month is not recommended due to potential variability from:
• Antibody lot changes
• Instrument drift
• Environmental factors
For experiments involving tandem dyes or ≥10 colors, fresh compensation should be performed for each experiment.
4.2 Compensation vs FMO Controls: Distinct but Complementary Roles
Compensation and FMO (Fluorescence Minus One) controls address fundamentally different issues and should not be considered interchangeable.
• Compensation: Corrects spectral spillover and decouples fluorescence signals across channels
• FMO controls: Define gating boundaries by accounting for signal spread and background distribution
Even with correct compensation, negative populations may appear broadened in multicolor panels due to the propagation of measurement noise from bright signals into other channels.
For low-expression markers, compensation alone is insufficient for accurate gating. FMO controls are essential for defining true positive populations.
4.3 Origin and Interpretation of Negative Values
Compensation involves subtractive calculations. As a result, weakly fluorescent or negative populations may yield negative values after spillover correction. This is a normal statistical outcome.
In logarithmic display, these values are compressed toward the lower axis and do not interfere with downstream analysis or gating.
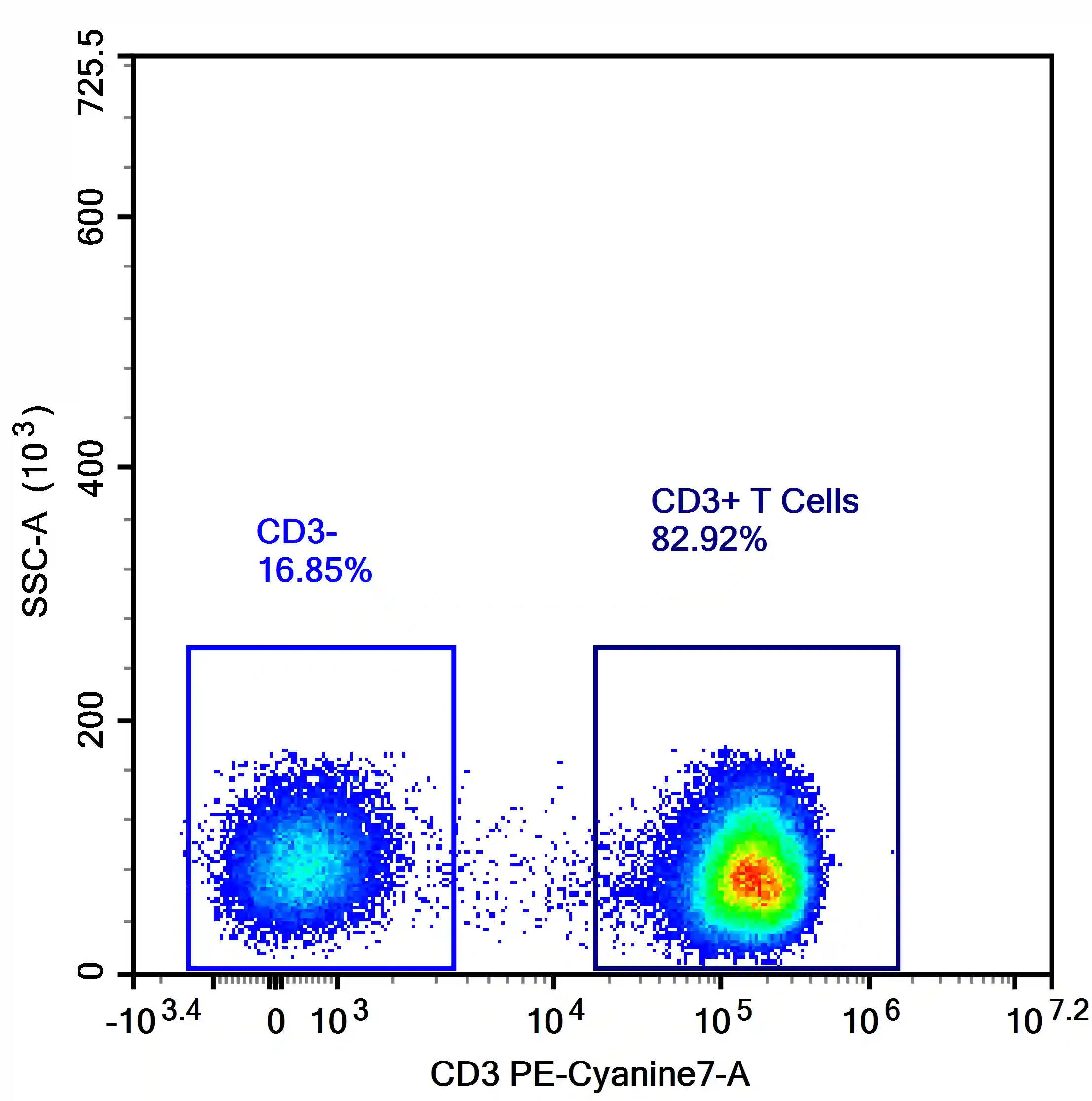
Figure 5. Distribution of negative populations in the negative range after compensation
No correction is required unless abnormal patterns are observed, such as:
• Median of negative population significantly below zero (suggesting overcompensation)
• Excessive spread or increased CV in negative populations
• Positive populations shifting into the negative range
These scenarios indicate inaccurate compensation and require reassessment.
Conclusion
Accurate compensation in multicolor flow cytometry depends not only on understanding the physical principles of fluorescence but also on rigorous control of experimental procedures.
Only through standardized workflows and careful attention to detail at every step can reliable and reproducible data be achieved, providing a solid foundation for downstream analysis and scientific conclusions.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience specializes in the development and production of high-quality life science reagents, and is committed to providing researchers with high-performance, highly reliable Flow Cytometry Antibodies, we offer:
1. Stringently validated flow cytometry antibodies including CD3, CD4, CD8 ensuring high sensitivity and minimal background noise;
2. A comprehensive portfolio of immunology research tools for multiple species, including Human, Mouse, Rat, Dog, Hamster;
3. Expert technical support and panel design consultation to help you accurately resolve complex immune cell populations.
For more information on abinScience flow cytometry antibodies, please visit:
abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский








