Sjögren’s syndrome (SS) is a chronic immune-mediated disease with an incompletely elucidated etiology. Extensive basic and translational studies have revealed that its pathogenesis is centered on dysregulated autoimmune responses, primarily manifesting as chronic inflammatory damage to exocrine glands and potential involvement of multiple extrapulmonary organs such as the kidneys, nervous system, and lymphatic system. Consequently, within the current research and disease classification framework, SS is widely recognized as a systemic disease model driven by autoimmune abnormalities and serves as an important model for investigating chronic immune dysregulation.
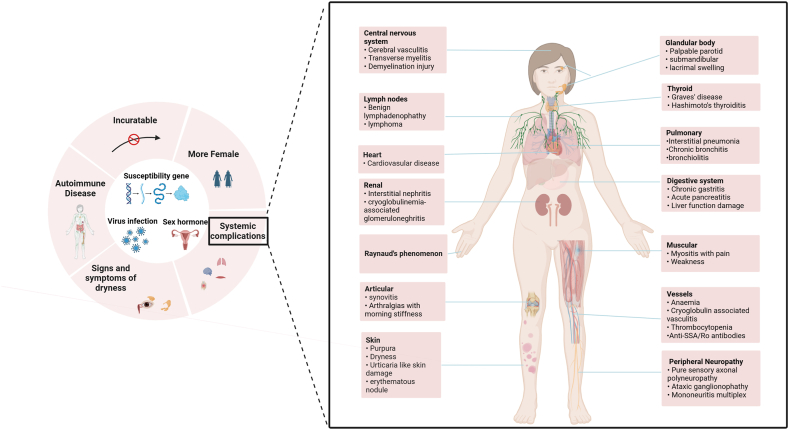
Figure 1. General features of Sjögren's syndrome
I. Core Mechanism of Sjögren’s Syndrome: Initiation and Persistence of Immune Responses
While the initial triggers of SS remain unclear, accumulating evidence supports a multifactorial etiology involving the interplay of genetic susceptibility and environmental factors. Genome-wide association studies (GWAS) have identified multiple immune regulatory susceptibility loci, including HLA-DR, HLA-DQB1, and non-HLA genes such as IRF5, STAT4, and TNFAIP3. These genes are highly enriched in signaling pathways governing interferon (IFN) responses, B cell activation, and inflammation regulation, indicating that the core abnormalities of SS are not random but converge on specific immunological axes.
In the early stages of the disease, glandular epithelial cells are believed to act as critical "initiating nodes" of immune responses by actively sensing nucleic acid–derived signals and amplifying local innate immune activation. Through pattern recognition receptors (PRRs) such as TLR3, TLR7, and TLR9, these cells sense viral or endogenous nucleic acid signals, activating the NF-κB and IRF pathways to induce the secretion of type I IFN and various inflammatory mediators. This rapidly transforms the local glandular microenvironment into a hyperimmune-activated state, laying the foundation for subsequent adaptive immune responses.
Notably, this process is not transient; instead, sustained immune dysregulation perpetuates the inflammatory response, which gradually expands from local to systemic immune abnormalities.
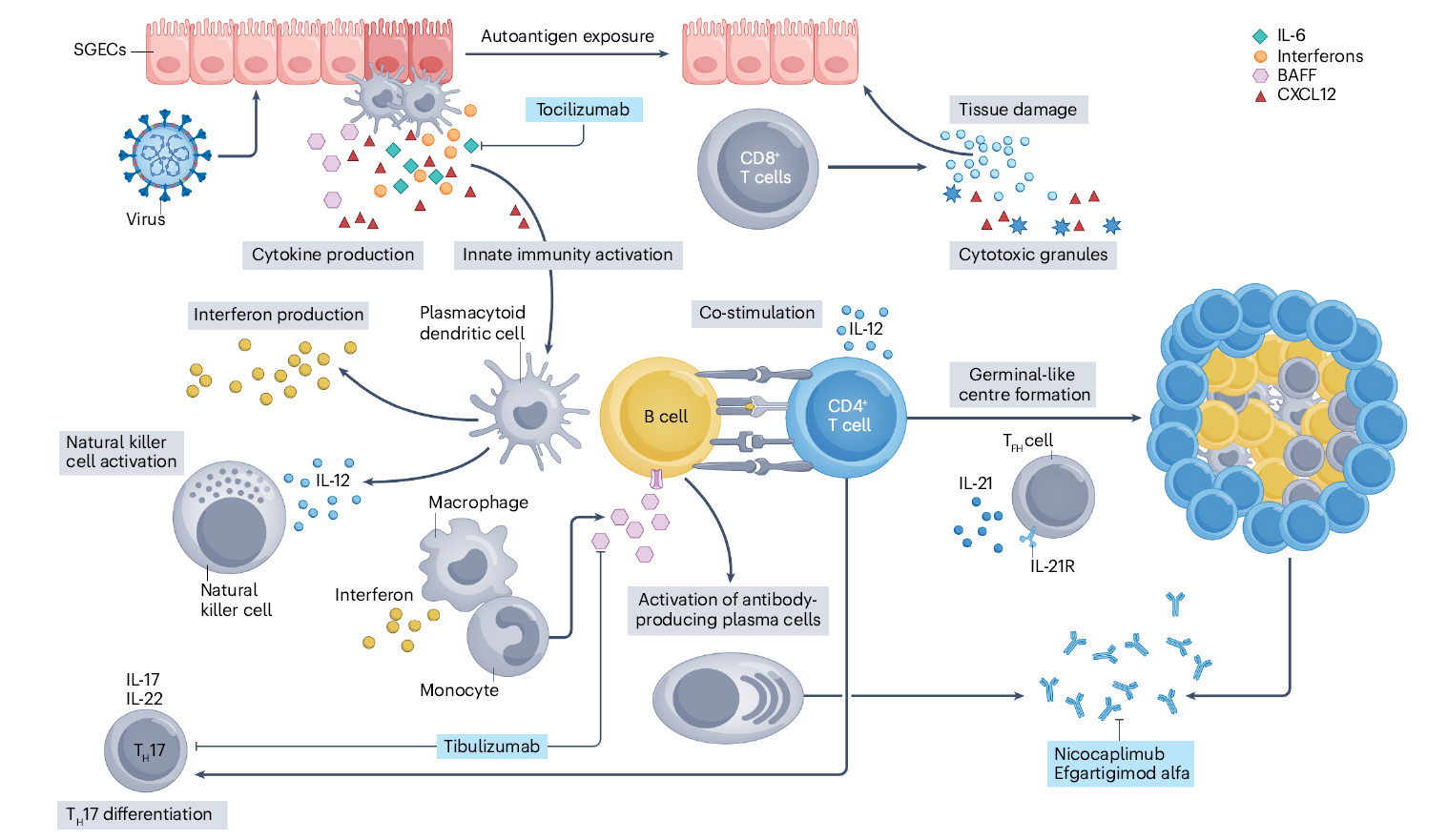
Figure 2. Sjögren syndrome pathogenetic model
II. Type I Interferon–B Cell Axis: The Central Immunological Axis in Sjögren’s Syndrome Research
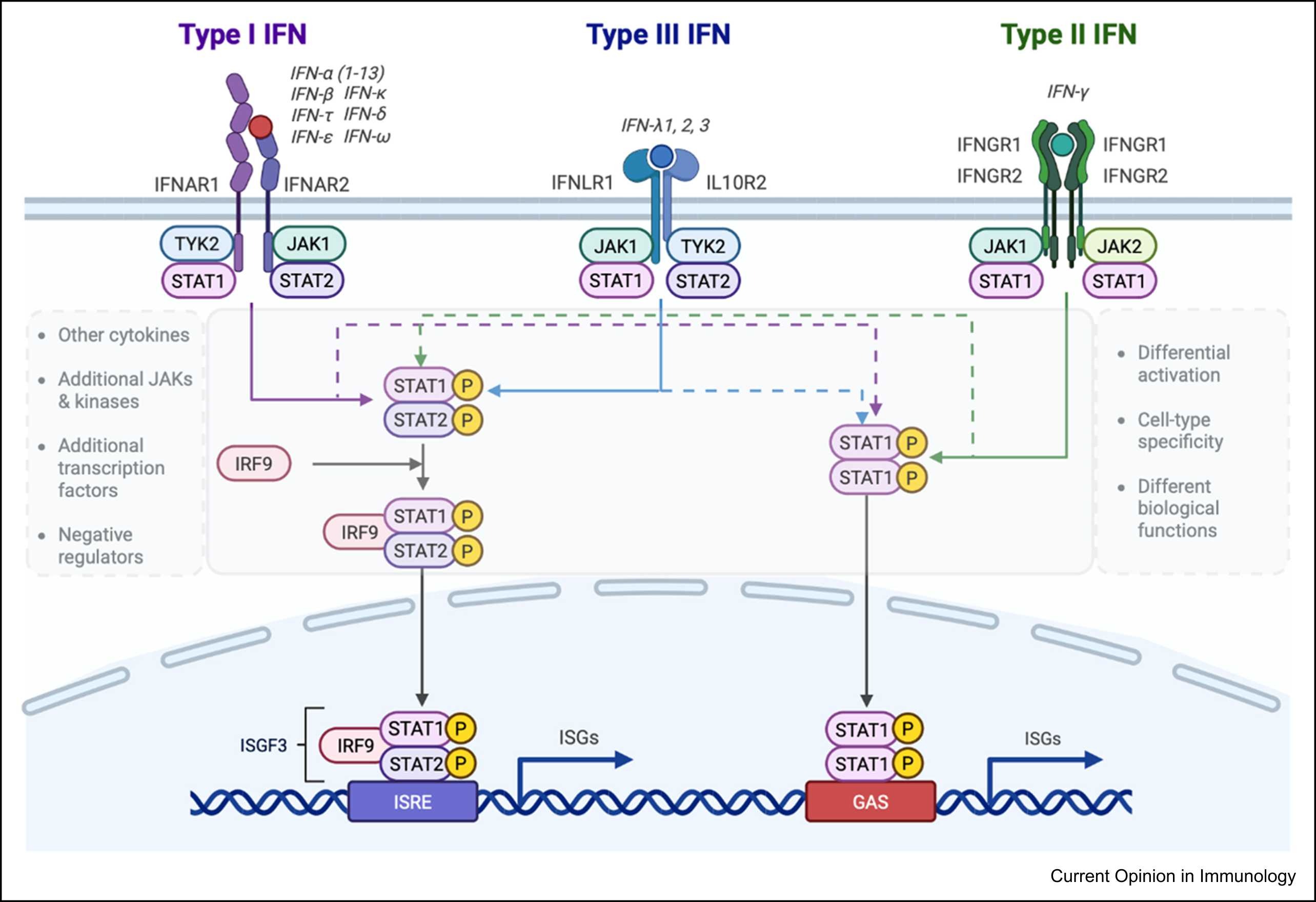
Figure 3. Schematic illustration of signal transduction by type I, II, and III IFN receptors
Among the identified immune abnormalities, the axis formed by type I IFN signaling and aberrant B cell activation represents one of the most consistently observed immunological features of SS.
Transcriptomic and proteomic studies have consistently demonstrated a prominent IFN signature in SS patients, whose intensity correlates closely with disease activity, autoantibody profiles, and the extent of systemic involvement. Type I IFN upregulates key molecules such as BAFF and CXCL13 via the JAK–STAT pathway, significantly enhancing the survival, migration, and differentiation of B cells.
In this context, autoreactive B cells escape immune tolerance, undergo clonal expansion and affinity maturation within the glands, and continuously produce autoantibodies such as anti-Ro/SSA and anti-La/SSB. Beyond contributing to serological characteristics, this process contributes to glandular tissue damage and is closely associated with an increased risk of SS-related lymphoproliferative disorders.
T cells play a crucial role in "sustaining and amplifying" this axis: follicular helper T cells support the persistence of aberrant B cell responses through co-stimulatory signals, while Th1 and Th17 cells exacerbate local inflammation by secreting IFN-γ and IL-17. In contrast, insufficient function of regulatory T cells impairs the termination of immune responses, leading to chronicity.
III. Glandular Microenvironment and Metabolic Regulation: Key Drivers of Chronic Inflammation
Emerging research indicates that the chronicity of SS is not solely determined by immune cells themselves; the glandular microenvironment plays a pivotal role in organizing and sustaining immune responses over time.
Spatial transcriptomics and single-cell RNA sequencing have revealed distinct spatial distribution patterns of immune cells within the salivary glands of SS patients. Specific subsets of endothelial cells and fibroblasts regulate immune cell recruitment through chemokine networks, endowing inflammatory responses with lymphoid-like tissue organization that enhances their stability and persistence.
Meanwhile, metabolic pathways have emerged as an important complementary research perspective. Metabolic signaling pathways such as PI3K/Akt/mTOR are highly activated in both immune cells and the glandular microenvironment, and are tightly coupled with inflammatory signals. This finding suggests that metabolic reprogramming may be a key mechanism sustaining immune cell hyperreactivity and chronic inflammation, opening new avenues for research beyond traditional immune targets.
IV. Core Significance of Recent Research Advances: From "Describing Abnormalities" to "Immune Stratification"
Recent advances in SS research are not characterized by the discovery of individual novel molecules, but rather by a fundamental shift in research strategy. Multi-omics integration analyses have revealed significant molecular heterogeneity among SS patients, with variations in IFN-driven intensity, B cell abnormal features, and immune network composition.
This recognition has propelled the development of immune stratification studies and provided a rational framework for explaining the inconsistent results of previous targeted therapy trials. Current research is moving away from a "one-size-fits-all mechanistic hypothesis" toward "stratified analysis of dominant immune factors," offering more refined insights for subsequent target research and model construction.
V. Prominent Therapeutic Targets and Their Immunological Rationale
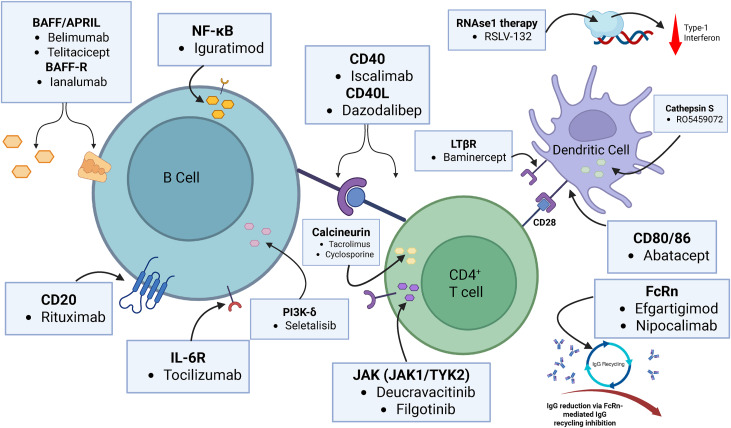
Figure 4. Novel agents targeting the main pathways involved in Sjögren syndrome pathophysiology
Targeted research in SS is highly focused, centered around the disease’s immune network architecture.
B cell-related targets (e.g., BAFF and its receptor, CD19, CD20, BTK) have long been the focus of research, as they lie at critical nodes of the IFN–B cell axis, governing the persistence of autoreactive B cells. The limited efficacy of single-target interventions has prompted investigators to explore combination or sequential treatment strategies.
T cell co-stimulatory pathways (CD40–CD40L, CD28–CD80/CD86), as hub nodes connecting T cell regulation and aberrant B cell responses, have also garnered widespread attention. Their regulatory value lies in their ability to modulate multiple immune layers rather than a single cell type.
At the signaling pathway level, the JAK–STAT pathway is regarded as a key hub in the immune network due to its integration of IFN and multiple inflammatory cytokine signals. Pathways such as NF-κB, STING, and the NLRP3 inflammasome are primarily involved in amplifying and sustaining immune responses, representing important directions in basic research.
In summary, SS research has evolved from symptom-oriented and single-pathway hypotheses to a systematic field centered on immune network dissection. Although its etiology remains incompletely understood, studies focusing on the IFN–B cell axis, T cell regulation, and the tissue microenvironment have established a relatively clear pathological framework. With the deepening of multi-omics and immune stratification research, SS not only advances the mechanistic understanding of its own pathogenesis but also serves as a valuable reference model for other chronic autoimmune diseases.
abinScience Sjögren’s Syndrome Research Solutions
1. Antibodies
| Product name |
Catalog No. |
| Anti-Human CD257/BAFF/TNFSF13B Antibody (BlySmAb-1.1) |
HV212107 |
| Anti-IL17A Polyclonal Antibody |
HS856014 |
| InVivoMAb Anti-Human IL17A (Iv0029) |
HS856010 |
| Anti-JAK1 Polyclonal Antibody |
HB829014 |
| Anti-Human CD40/TNFRSF5 Antibody (SAA2006) |
HB782023 |
| Anti-Human HLA-DRB1 Antibody (MP12) |
HM837013 |
| Anti-Human CD20/MS4A1 Antibody (SAA2511) |
HY257033 |
| Anti-Human CD278/ICOS Antibody (SAA0092) |
HV702207 |
| Anti-Human CD126/IL6R/IL-6RA Nanobody (SAA1278) |
HY583013 |
| Anti-Human CD182/CXCR2 Antibody (SAA1360) |
HB243053 |
View more Sjögren’s Syndrome Antibodies
2. Assay Kits
View more Sjögren’s Syndrome Assay Kits
3. Research Biosimilars
| Target |
Product name |
Catalog No. |
| CD20 |
Research Grade Ofatumumab |
HY257466 |
| Research Grade Rituximab |
HY257446 |
| CD40 |
Research Grade Iscalimab |
HB782016 |
| Research Grade ciltistotug |
HB782346 |
| IL17A |
Research Grade Secukinumab |
HS856026 |
| Research Grade betinukibart |
HS856236 |
| Research Grade turenkibart |
HS856246 |
| CD257 |
| Research Grade Blisibimod |
HV212056 |
| Research Grade Belimumab |
HV212016 |
| IFNAR1 |
| Research Grade Anifrolumab |
HB823016 |
| Research Grade Anti-Human IFNAR1 (QX 006N) |
HB823036 |
View more Sjögren’s Syndrome Research Biosimilars
abinScience provides high-quality antibodies, assay kits, and other research tools covering key directions in Sjögren’s syndrome research. These tools support studies on IFN signaling, aberrant B cell activation, and related immune pathways, offering reliable technical support for in-depth mechanistic exploration and advancement of basic and translational research in SS.
References
[1] Mariette X, Barone F, Baldini C, Bootsma H, Clark KL, De Vita S, Gardner DH, Henderson RB, Herdman M, Lerang K, Mistry P, Punwaney R, Seror R, Stone J, van Daele P, van Maurik A, Wisniacki N, Roth DA, Tak PP. A randomized, phase II study of sequential belimumab and rituximab in primary Sjögren's syndrome. JCI Insight. 2022 Dec 8;7(23):e163030. doi: 10.1172/jci.insight.163030. PMID: 36477362; PMCID: PMC9746921.
[2] Zhao T, Zhang R, Li Z, Qin D, Wang X. A comprehensive review of Sjögren's syndrome: Classification criteria, risk factors, and signaling pathways. Heliyon. 2024 Aug 15;10(17):e36220. doi: 10.1016/j.heliyon.2024.e36220. PMID: 39286095; PMCID: PMC11403439.
[3] Carnazzo V, Rigante D, Restante G, Basile V, Pocino K, Basile U. The entrenchment of NLRP3 inflammasomes in autoimmune disease-related inflammation. Autoimmun Rev. 2025 Jun 24;24(7):103815. doi: 10.1016/j.autrev.2025.103815. Epub 2025 Apr 13. PMID: 40233890.
[4] Price E, Bombardieri M, Kivitz A, Matzkies F, Gurtovaya O, Pechonkina A, Jiang W, Downie B, Mathur A, Mozaffarian A, Mozaffarian N, Gottenberg JE. Safety and efficacy of filgotinib, lanraplenib and tirabrutinib in Sjögren's syndrome: a randomized, phase 2, double-blind, placebo-controlled study. Rheumatology (Oxford). 2022 Nov 28;61(12):4797-4808. doi: 10.1093/rheumatology/keac167. PMID: 35377447; PMCID: PMC9707320.
[5] Raftopoulou S, Bakasis AD, Mavragani CP. Interferons in Sjogren's disease: current status and future prospects. Curr Opin Immunol. 2026 Feb;98:102703. doi: 10.1016/j.coi.2025.102703. Epub 2025 Dec 5. PMID: 41352246.
[6] Tang X, Shih PC, Hu J, Ma D, Zhang L. JAK Inhibitors: Therapeutic Prospects and Clinical Challenges in Sjögren's Syndrome. Int J Rheum Dis. 2025 Jul;28(7):e70377. doi: 10.1111/1756-185x.70377. PMID: 40692262.
[7] Gandolfo S, Bombardieri M, Pers JO, Mariette X, Ciccia F. Precision medicine in Sjögren's disease. Lancet Rheumatol. 2024 Sep;6(9):e636-e647. doi: 10.1016/S2665-9913(24)00039-0. Epub 2024 May 6. PMID: 38723653.
[8] Baldini C, Fulvio G, La Rocca G, Ferro F. Update on the pathophysiology and treatment of primary Sjögren syndrome. Nat Rev Rheumatol. 2024 Aug;20(8):473-491. doi: 10.1038/s41584-024-01135-3. Epub 2024 Jul 9. PMID: 38982205.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






