In the history of cancer diagnostics, the epithelial tumor marker SerpinB3 (also known as SCCA-1) has been recognized for nearly half a century. As early as 1977, Japanese researchers Kato and Torigoe first detected it in the serum of patients with cervical squamous cell carcinoma and named it tumor antigen TA-4. Since then, SerpinB3 has become a near-universal “alarm” for epithelial tumors: elevated levels are closely linked to increased metastasis risk, treatment resistance, and poor prognosis in head and neck, lung, esophageal, liver, pancreatic, and even skin squamous cell carcinomas. Today, SCCA testing remains widely used for monitoring epithelial tumors.
However, a research team from Arizona State University has proposed a paradigm-shifting hypothesis: SerpinB3’s “cancerous” behavior may simply reflect its native skin-repair function being inappropriately activated. Published in PNAS (https://doi.org/10.1073/pnas.2415164122), the study reveals that SerpinB3 actually serves as an “endogenous switch” in the skin injury response, promoting epithelial regeneration and tissue remodeling. Starting from the question “Is SerpinB3 an intrinsic component of the skin injury response?”, the work uncovers its central role in wound healing—inducing an EMT-like transition in keratinocytes to accelerate re-epithelialization and collagen remodeling—providing an evolutionarily conserved physiological explanation for its “runaway activation” in cancer and chronic wounds.
Unexpected Signal in Wound Models
The study began with a serendipitous observation. While analyzing previously published transcriptomic data from silk-gold nanorod (Silk-GNR) dressings that promote wound healing, the team noticed an overlooked gene—Serpinb3a (the mouse homolog). On day 4 post-treatment, Serpinb3a expression surged in mouse wounds, hinting at a role in repair. Mining multiple public transcriptomic datasets confirmed the pattern: after skin injury, Serpinb3a is strongly induced early on and appears exclusively in activated keratinocytes (Krt16⁺), not in basal cells or fibroblasts. In human skin donors, SERPINB3 and SERPINB4 were synchronously upregulated 3–7 days post-surgery. Protein validation showed Serpinb3a peaking on day 2 in mouse wounds and returning to baseline by day 7; in human HaCaT keratinocytes, it rose rapidly within 24 hours of scratch stimulation. Together, these data establish SerpinB3/Serpinb3a as a classic skin injury response factor involved in early epithelial migration.
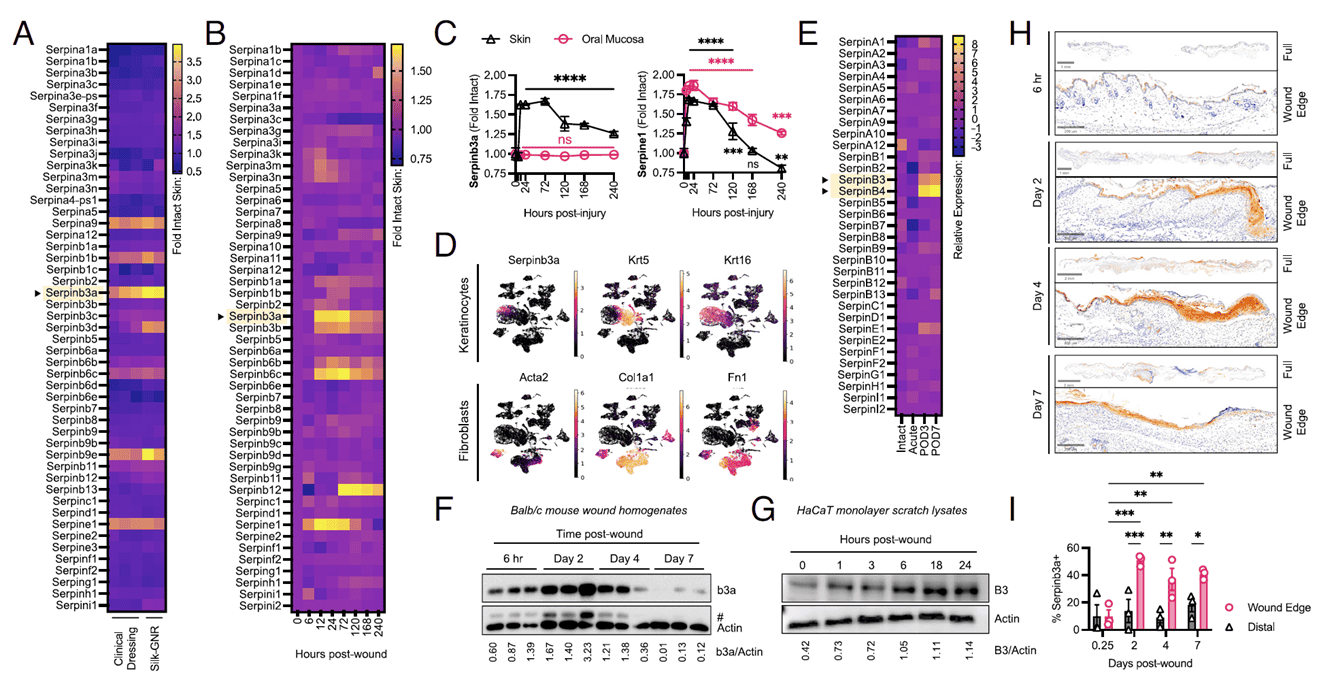
Figure 1. SerpinB3/b3a is an epidermal wound-response element
The Molecule That Gets Epithelial Cells “Moving”
Given SerpinB3’s high expression in migrating wound-edge epithelium, the researchers hypothesized it directly modulates cell behavior. Overexpressing SerpinB3 in HaCaT cells dramatically altered morphology: E-cadherin plummeted, pSTAT3 signaling intensified, and cells became elongated with enhanced polarity. This is a hallmark epithelial-to-mesenchymal transition (EMT)-like state typical of migrating and repairing epithelial cells. Adding recombinant Serpinb3a externally triggered similar effects, with upregulated fibronectin, modestly reduced proliferation, and boosted migration. Scratch assays showed Serpinb3a-treated HaCaT cells re-epithelialized at a rate comparable to EGF-treated controls within 24 hours, while fibroblasts (NIH/3T3) were unresponsive. These findings demonstrate that SerpinB3 drives EMT-like changes to make keratinocytes more migratory, thereby hastening wound closure.
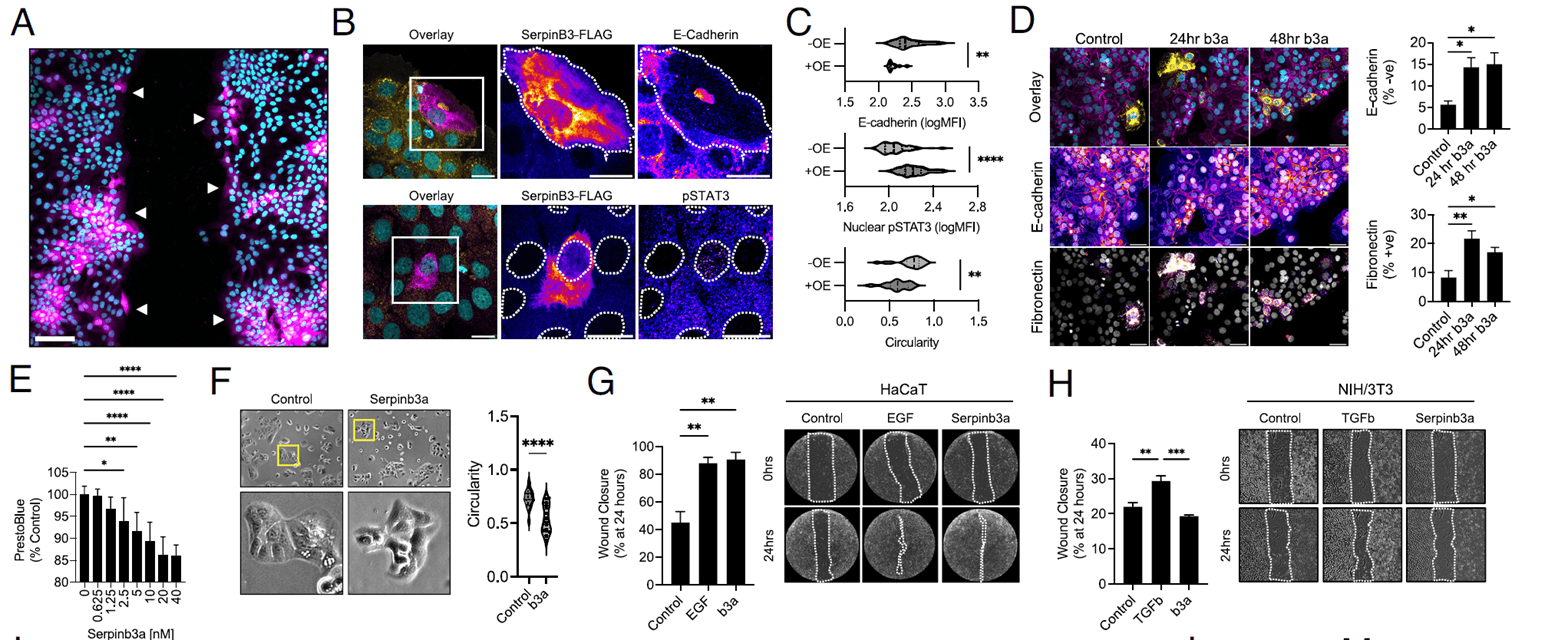
Figure 2. SerpinB3 promotes re-epithelialization by inducing an EMT-like phenotype in keratinocytes
In Vivo Validation in Mice: Faster Closure and Better Collagen Remodeling
To test physiological relevance, the team applied exogenous Serpinb3a in a mouse skin wound model. Early-stage closure accelerated markedly in the Serpinb3a group; although differences narrowed after day 7 due to wound contraction, by day 14 regenerating epithelium still expressed high Serpinb3a, suggesting a “lingering primed” state. Histology revealed collagen remodeling in the Serpinb3a group more closely resembled healthy skin: fibers were thicker, more uniformly oriented, and formed the classic basket-weave pattern. Notably, this improvement did not involve fibroblast activation—α-SMA and Collagen I levels were unchanged. The authors speculate that remodeling factors released by epithelial cells (e.g., MMP10, Relm-α) drive the effect; these molecules degrade scar tissue while also fueling epithelial tumor progression. Thus, SerpinB3 acts as a “signal coordinator” in wound repair, orchestrating epithelial behavior and intercellular communication to promote orderly tissue reconstruction.
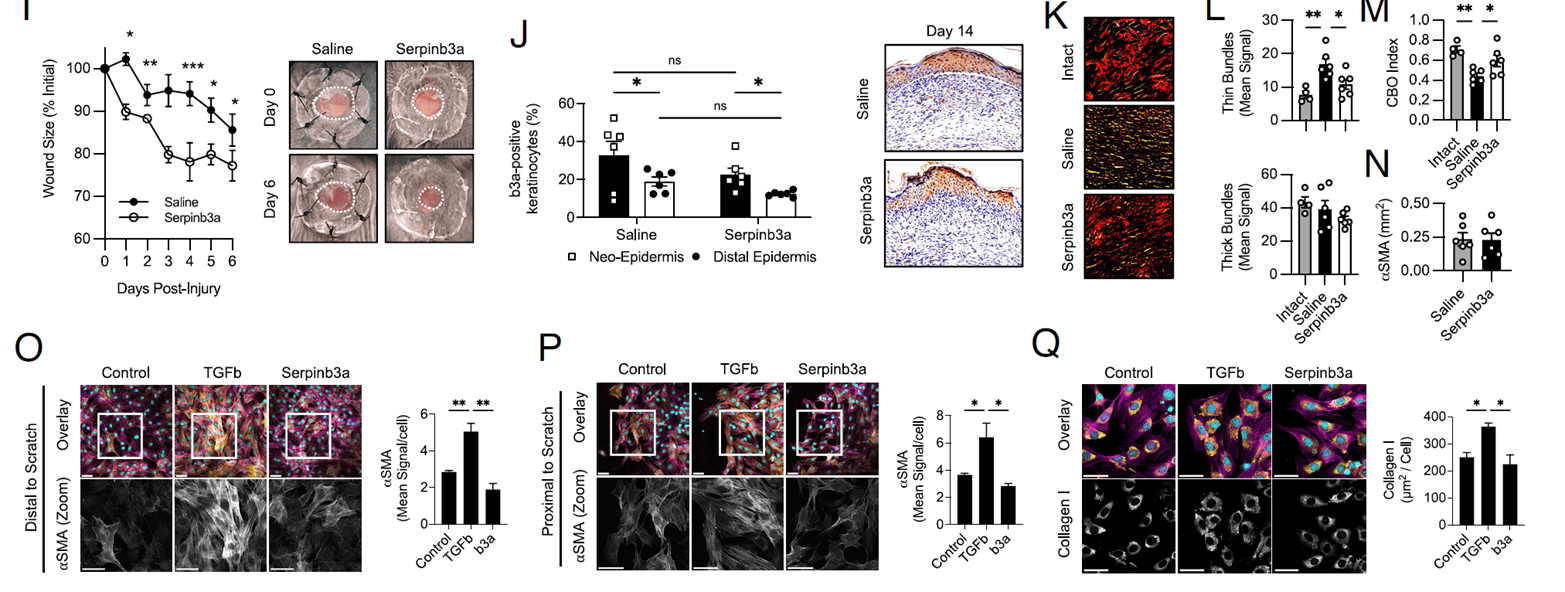
Figure 3. Serpinb3a accelerates wound closure and optimizes collagen remodeling in vivo
This study offers a fresh explanation for the long-standing controversy surrounding SerpinB3 in cancer. Its high expression in tumors may not reflect an “aberrantly activated oncogene” but rather a wound-repair program hijacked by cancer cells. As early as 2014, Sheshadri et al. showed SCCA1/SERPINB3 promotes EMT and invasion via UPR and IL-6 pathways; in 2015, Sivaprasad et al. demonstrated SERPINB3/B4’s involvement in inflammation and skin barrier disruption in animal models. Now, this work uncovers its primordial physiological function—preserving epithelial regeneration and barrier integrity during tissue damage. Going forward, SerpinB3 could serve simultaneously as a disease biomarker and therapeutic target in oncology and wound healing, depending on how we interpret and modulate it.
About abinScience
abinScience was founded in Strasbourg, France, leveraging the region’s outstanding research and innovation ecosystem to focus on developing and producing high-quality life-science reagents. Guided by the vision “Empowering Bioscience Discovery,” abinScience is dedicated to delivering efficient, reliable experimental solutions to researchers worldwide, fueling cutting-edge life-science advances.
Below is the list of SERPINB3-related proteins and antibodies offered by abinScience:
| Type |
Catalog No. |
Product name |
| Protein |
HB632012 |
Recombinant Human SERPINB3 Protein, N-His |
| Antibody |
HB632014 |
Anti-SERPINB3 Polyclonal Antibody |
| HB632025 |
Anti-Human SCCA Monoclonal Antibody (1A294) |
| HB632015 |
Anti-Human SCCA Monoclonal Antibody (1A293) |
Disclaimer: This article is based on publicly available literature; products are for research use only.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский





