The UK Health Security Agency (UKHSA) continues to investigate an unprecedented outbreak of invasive meningococcal disease (IMD) in the Canterbury area of Kent. As of March 22 (12:30 pm), a total of 29 cases have been reported — 20 laboratory-confirmed (19 identified as MenB) and 9 still under investigation. Two deaths have been confirmed: a 21-year-old University of Kent student and 18-year-old sixth-form student Juliette Kenny. The total peaked at 34 on March 20 before some cases were reclassified following further laboratory testing. No new cases have been reported since March 23. The outbreak has been traced to Canterbury's Club Chemistry nightclub (March 5–7), with cases linked to the University of Kent, Canterbury Christ Church University, and several secondary schools. One case connected to the University of Kent was confirmed in France, involving a 19-year-old exchange student. Health Secretary Wes Streeting described the outbreak as "unprecedented" and announced a targeted MenB vaccination program. As of March 22, over 9,000 MenB vaccines and 12,500 doses of preventive antibiotics had been administered across six clinic sites in Kent. The UKHSA has stated the outbreak is "not yet over" and remains under active surveillance.

Figure 1. University of Kent students queuing for antibiotics. Image sourced from The Guardian.
Transmission and Pathogenesis of Neisseria meningitidis
Meningococcal disease is caused by Neisseria meningitidis and spreads primarily through respiratory droplets or close contact (such as kissing or sharing drinks, e-cigarettes, or cups). Between 10% and 20% of healthy individuals carry the bacteria asymptomatically in the nasopharynx, with carriage rates reaching 20–30% in university settings — particularly among adolescents and young adults. Once the bacteria invade the bloodstream or meninges, they can trigger sepsis or meningitis.
Although traditionally classified as an extracellular pathogen, recent research has revealed that N. meningitidis possesses a robust intracellular lifestyle — a feature critical to IMD pathogenesis. The bacterium first colonizes the nasopharyngeal mucosa, adhering to epithelial cells via type IV pili (T4P, the pilE pilus protein), Opa/Opc adhesins, and other virulence factors. This triggers microvilli-like protrusion formation and bacterial internalization into endosomes. The bacteria then cross the nasopharyngeal epithelial barrier (NEB) via transcellular or paracellular routes to enter the bloodstream.
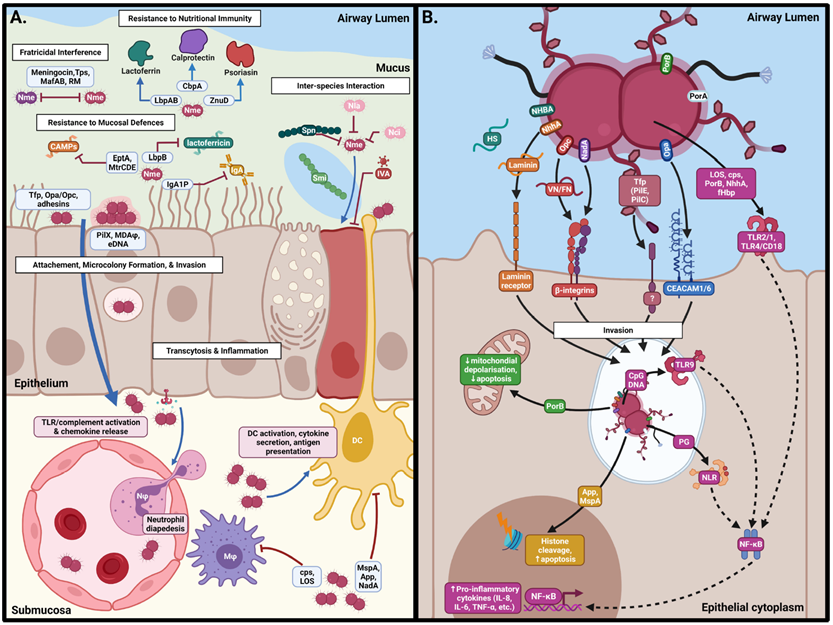
Figure 2. Neisseria meningitidis interactions at the nasopharyngeal surface
Once in the bloodstream, the bacteria can further invade endothelial cells and cross the blood-brain barrier (BBB), leading to sepsis or meningitis. The key lies in the intracellular escape mechanism: type IV pili bind host CD147/PAFr receptors, triggering calcium release and LAMP1 redistribution. IgA protease cleaves LAMP1, blocking endosome maturation. Meanwhile, the PARK9 transporter elevates intravacuolar Mn²⁺ concentrations, activating HrpA hemolysin activity and ultimately lysing the vacuolar membrane to release bacteria into the cytosol. Cytosolic bacteria bind dynein, modulate NF-κB, inhibit apoptosis, and activate inflammasomes (caspase-1/4), triggering pyroptosis and amplifying inflammation.
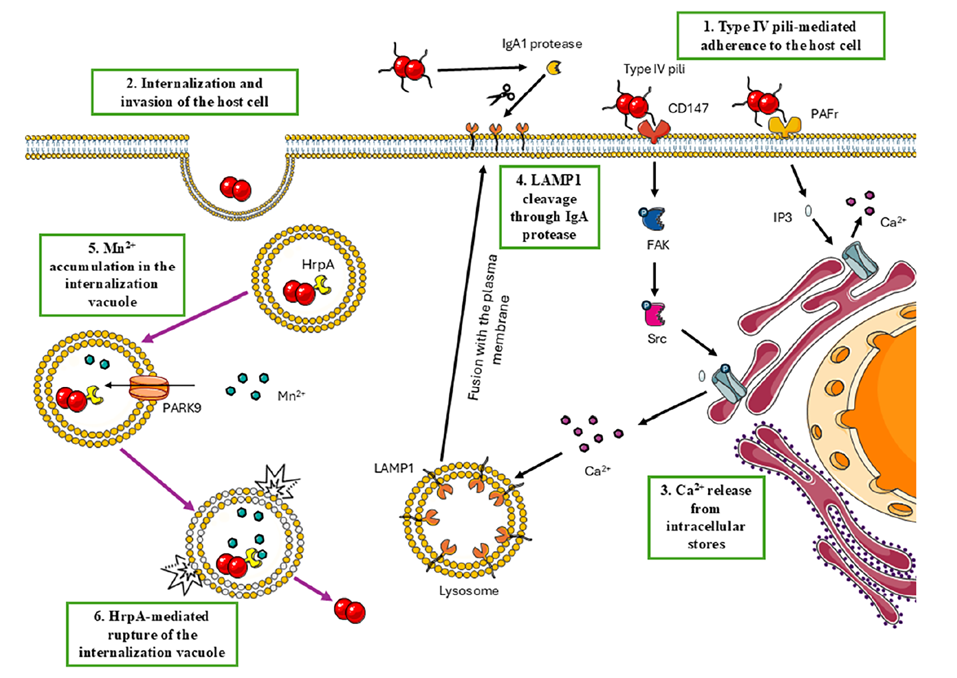
Figure 3. Proposed mechanism of Neisseria meningitidis vacuolar escape
Key Targets
Vaccine and therapeutic development centers on surface antigens and virulence factors. abinScience offers recombinant proteins and antibodies covering the major targets listed below (see product recommendations at the end of this article):
| Target |
Full Name / Description |
Key Function / Mechanism |
| FHbp |
Factor H-binding protein (also GNA1870 / LP2086) |
Core antigen of 4CMenB and pentavalent vaccines; binds complement factor H to evade immune killing |
| NHBA |
Neisserial Heparin Binding Antigen (formerly GNA2132) |
Surface-exposed; promotes adhesion; induces bactericidal antibodies; cross-protection against N. gonorrhoeae |
| NadA |
Neisseria adhesin A |
Promotes adhesion to and invasion of epithelial/endothelial cells |
| PorA |
Porin A (outer membrane protein) |
Major OMV antigen; provides strain-specific protection |
| TbpB |
Transferrin-binding protein B |
Iron acquisition system; essential for bacterial survival in the host |
| MtrE |
Mtr efflux pump outer membrane channel |
Antibiotic efflux; contributes to antimicrobial resistance |
| TCS |
Two-Component Systems (only 6 pairs; highly conserved) |
Regulate virulence factor expression, antibiotic response, resistance, and environmental adaptation |
Latest Research Advances
Pentavalent vaccine breakthroughs: The FDA has approved two pentavalent MenABCWY vaccines — PENBRAYA (Pfizer, 2023) and PENMENVY (GSK, February 2025) — that cover serogroups A/C/W/Y/B in a single formulation, substantially simplifying immunization schedules. Both vaccines incorporate FHbp recombinant protein as a core MenB antigen. In Africa, the Men5CV (NmCV-5) has received WHO prequalification and is being administered widely to infants.
Novel platform vaccines: The University of Oxford's ChAdOx1 MenB.1 — an adenoviral vector expressing MenB outer membrane proteins — reported Phase I/IIa results in Science Translational Medicine (2025). After two doses, 100% of participants generated protective human serum bactericidal antibody (hSBA) responses against the target MenB strain. The trial confirmed that viral vector platforms can be successfully applied to bacterial vaccine development.
Cross-protection and emerging targets: Clinical evidence now supports 33–47% cross-protection of 4CMenB against Neisseria gonorrhoeae. NHBA combined with OMV induces bactericidal antibodies, and human challenge trials are underway. Two-component systems (TCS) have been proposed as novel anti-virulence and anti-resistance targets.
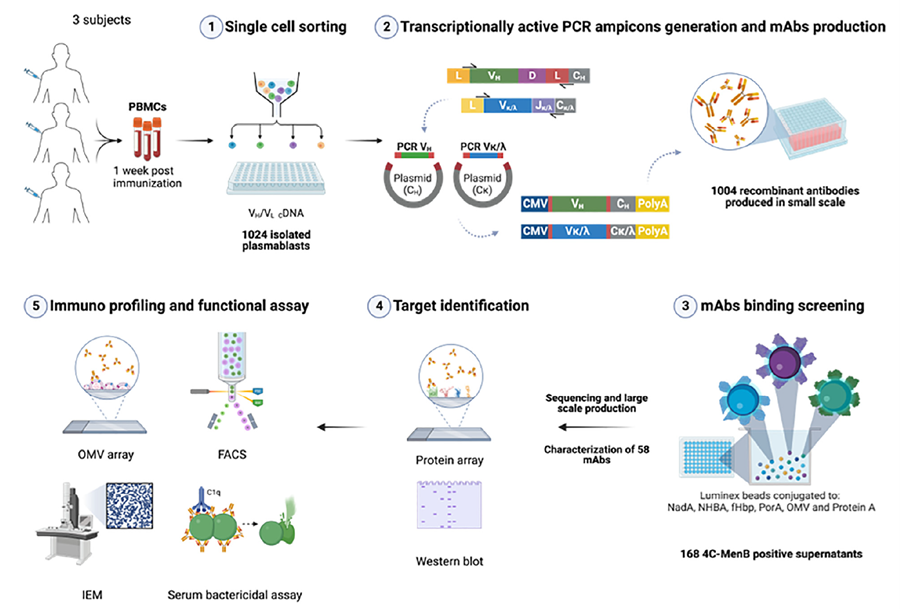
Figure 4. Monoclonal antibodies isolated from 4CMenB vaccinees — PorB and LOS identified as major OMV components driving cross-strain protection
abinScience Products for Meningococcal Research
abinScience provides recombinant proteins and antibodies covering the key N. meningitidis virulence and vaccine targets. Featured products organized by target are listed below (click catalog numbers for full product details):
Recombinant Proteins
| Target |
Catalog No. |
Product Name |
| FHbp |
JN824072 |
Recombinant N. meningitidis FHbp Protein, N-His-KSI |
| FHbp |
JN824012 |
Recombinant N. meningitidis FHbp Protein, C-His |
| NHBA |
JN891022 |
Recombinant N. meningitidis NHBA Protein, N-His |
| NadA |
JN971012 |
Recombinant N. meningitidis NadA Protein, N-His |
| pilE |
JN057012 |
Recombinant N. meningitidis pilE Protein, N-His |
| neuA |
JN976012 |
Recombinant N. meningitidis neuA Protein, C-His |
Antibodies
| Target |
Catalog No. |
Product Name |
| FHbp |
JN824013 |
Anti-N. meningitidis FHBP Antibody (1A12) |
| FHbp |
JN824014 |
Anti-N. meningitidis FHbp Polyclonal Antibody |
| NHBA |
JN891013 |
Anti-N. meningitidis NHBA Antibody (10C3) |
| NadA |
JN971013 |
Anti-N. meningitidis NadA Antibody (G9) |
| PorA |
JN957013 |
Anti-N. meningitidis porA Antibody (MN16C13F4) |
| LPS |
JN067403 |
Anti-N. meningitidis Group B LPS Antibody (SAA1462) |
| pilE |
JN057024 |
Anti-N. meningitidis pilE Polyclonal Antibody |
Browse All N. meningitidis Products →
Covering FHbp / NHBA / NadA / pilE / neuA / PorA / LPS — recombinant proteins, monoclonal & polyclonal antibodies
Need Reagents for Meningococcal Research?
abinScience provides manufacturer-direct recombinant proteins and antibodies for N. meningitidis research. Bulk pricing, custom specifications, and technical support available.
Email: info@abinscience.com | Tel: +86-27-65523339
References
1. Mikucki A, McCluskey NR, Kahler CM. The Host-Pathogen Interactions and Epicellular Lifestyle of Neisseria meningitidis. Front Cell Infect Microbiol. 2022;12:862935. doi: 10.3389/fcimb.2022.862935
2. Resta SC, Talà A, Conte R, Calcagnile M, Bucci C, Alifano P. Neisseria meningitidis: a traditional extracellular pathogen with an intense intracellular lifestyle. Front Cell Infect Microbiol. 2025;15:1733264. doi: 10.3389/fcimb.2025.1733264
3. Vezzani G, Viviani V, Audagnotto M, et al. Isolation of human monoclonal antibodies from 4CMenB vaccinees reveals PorB and LOS as the main OMV components inducing cross-strain protection. Front Immunol. 2025;16:1565862. doi: 10.3389/fimmu.2025.1565862

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






