2025 Latest Treponema pallidum Research Review
In recent years, syphilis infections have surged dramatically worldwide. According to data from Japan's Ministry of Health, Labour and Welfare, confirmed syphilis cases have exceeded 13,000 for four consecutive years, reaching a 25-year peak of approximately 15,000 cases in 2023, with over 80% of female cases occurring in women in their 20s. Social media-fueled indiscriminate sexual behaviors combined with distorted perceptions—such as "showing off" infections—have allowed this ancient infectious disease to make a strong comeback, with the true "deadly culprit" lurking in intimate contact being Treponema pallidum.


Figure 1. Social media "infection showcase" (image sourced from the web)
Introduction to Treponema pallidum
Syphilis is a chronic, systemic sexually transmitted infection caused by the spirochete bacterium Treponema pallidum subsp. pallidum (commonly abbreviated as T. pallidum). It can also be transmitted vertically from mother to child, resulting in congenital syphilis. The disease progresses through distinct stages: primary (painless chancre at the infection site, typically appearing 3–6 weeks after sexual contact), secondary (disseminated rash, fever, lymphadenopathy, and mucosal lesions, often accompanied by systemic symptoms such as fatigue and weight loss), latent (asymptomatic, but the bacteria persist in the body, potentially for years), and tertiary (if untreated, may involve cardiovascular complications like aortitis or neurological issues such as tabes dorsalis or gummas, leading to severe disability or death). Diagnosis relies on serological tests, such as nonspecific VDRL and specific TPHA assays, since T. pallidum cannot be routinely cultured. Early penicillin treatment is highly effective, but neurosyphilis can occur at any stage and requires lumbar puncture for confirmation. Untreated tertiary syphilis can cause life-threatening cardiovascular and neurological damage.
Molecular Structure of Treponema pallidum
T. pallidum is a slender, spiral-shaped bacterium, approximately 6–15 μm in length and 0.1–0.2 μm in diameter, exhibiting the classic spirochete morphology with periplasmic flagella (axial filaments) that enable its corkscrew-like motility. These flagella are located between the cell wall and outer membrane, allowing the bacterium to penetrate mucosal tissues. Unlike typical Gram-negative bacteria, it lacks lipopolysaccharide in its outer membrane but possesses lipoproteins and sparsely distributed outer membrane proteins, which aid in immune evasion. Under electron microscopy, T. pallidum appears as a tightly coiled spiral, resembling a corkscrew, with a smooth surface and no capsule.

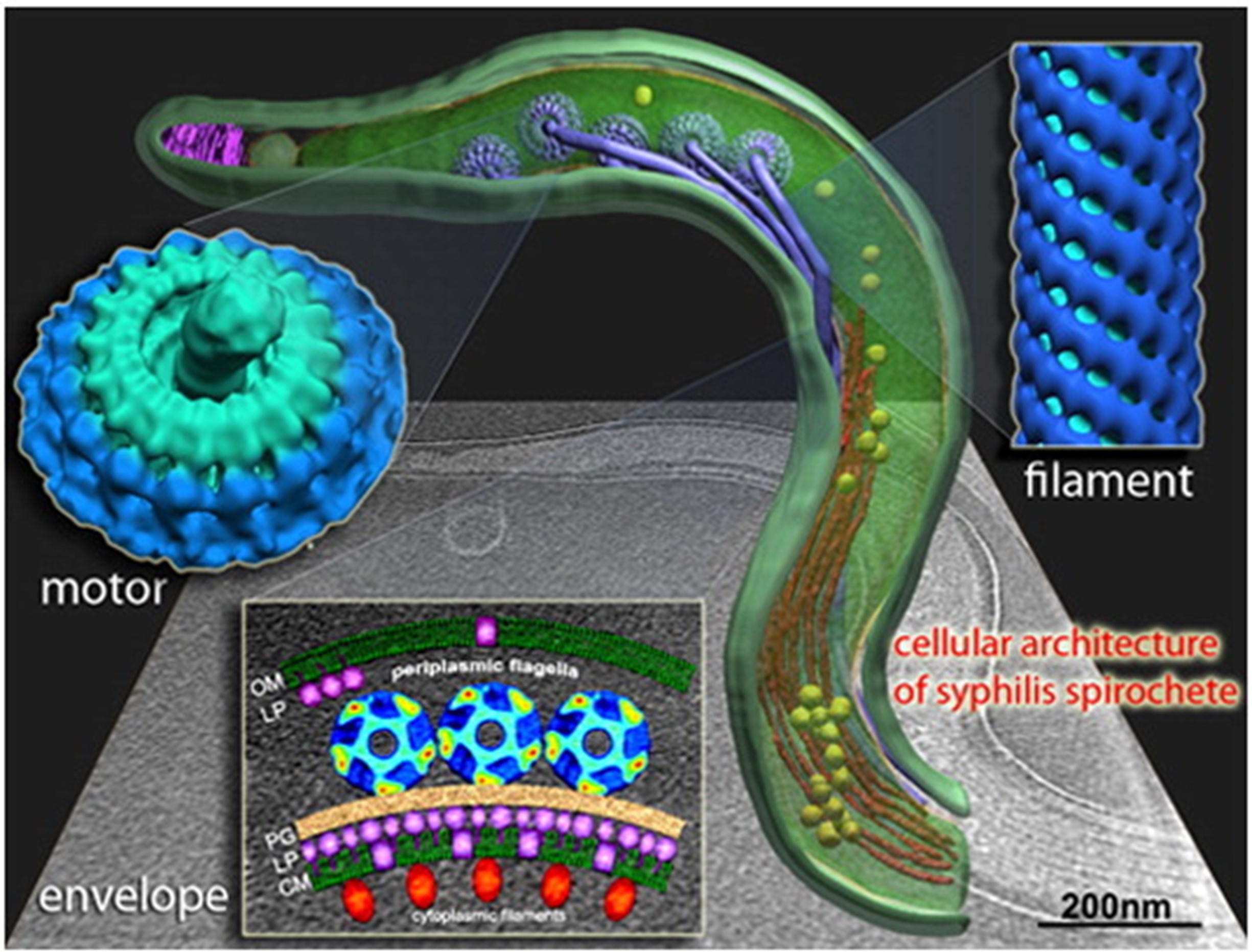
Figure 2. High-resolution cryo-electron tomography visualization of the syphilis pathogen Treponema pallidum
The outer membrane of T. pallidum contains limited transmembrane proteins, such as Tp0751 and Tp92, which are involved in host cell adhesion and nutrient uptake. The bacterial genome is relatively small (~1.1 Mb) and encodes about 1,041 genes, many related to metabolic simplification and a parasitic lifestyle. The lipoprotein layer (e.g., Tp47) in the outer membrane forms a protective barrier that reduces immune recognition. Studies show that surface antigen variation occurs through gene recombination, enhancing persistence. Molecular structure models typically highlight its three-layer cell envelope: outer membrane, periplasmic space (containing flagella), and inner membrane.
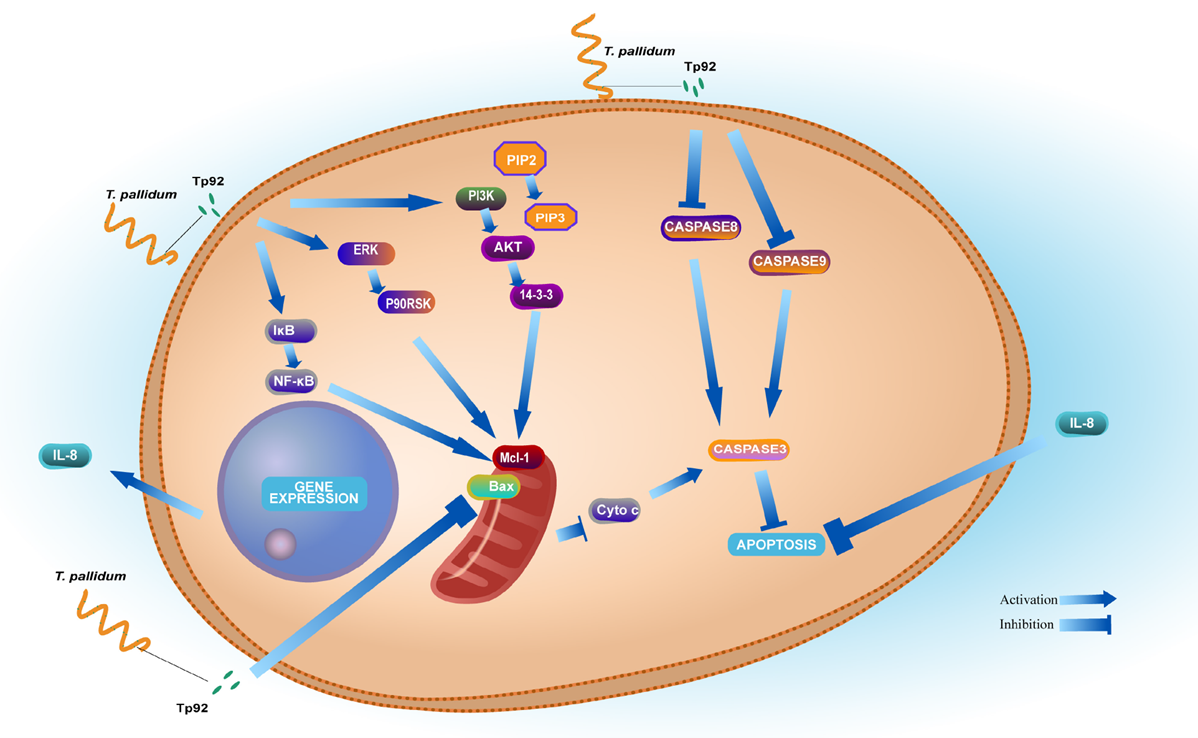

Figure 3. Tp92 delays human neutrophil apoptosis via ERK, PI3K/Akt, and NF-κB pathways
Pathogenesis Mechanisms
During transmission, T. pallidum breaches mucosal or epithelial barriers, adheres to host cells via surface proteins such as Tp0751, and disseminates hematogenously to cause multi-system damage. The bacterium evades host immunity through antigen variation, limited outer membrane protein exposure, and induction of anti-inflammatory responses, allowing persistent infection in tissues like the central nervous system (CNS) and placenta, leading to long-term latent infection. Pathogenesis involves pro-inflammatory cytokine secretion by macrophages, facilitating blood-brain barrier (BBB) breach and neurosyphilis, or placental crossing leading to congenital infection. Its corkscrew motility and low metabolic rate further enable migration and survival within the host.
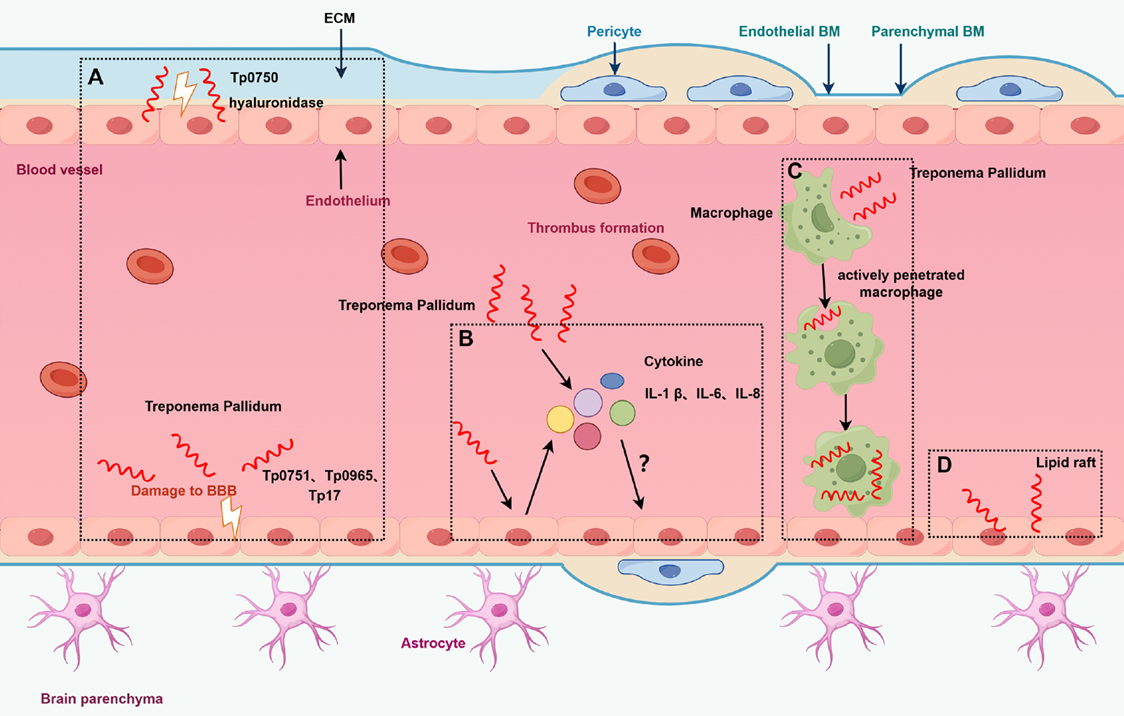

Figure 4. Schematic of potential mechanisms by which T. pallidum breaches the blood-brain barrier
Notably, T. pallidum's immune evasion mechanisms are central to its ability to establish persistent infection. On one hand, the sparse exposure of outer membrane proteins limits immune recognition targets; on the other, studies have shown that the bacterium can weaken host clearance by modulating antigen expression and inducing apoptosis in immune cells. Additionally, the membrane-associated protein TmpA plays a key role in bacterial adhesion to host cells, tissue recognition, and immune response activation—dysfunction in TmpA may further promote infection progression and dissemination.
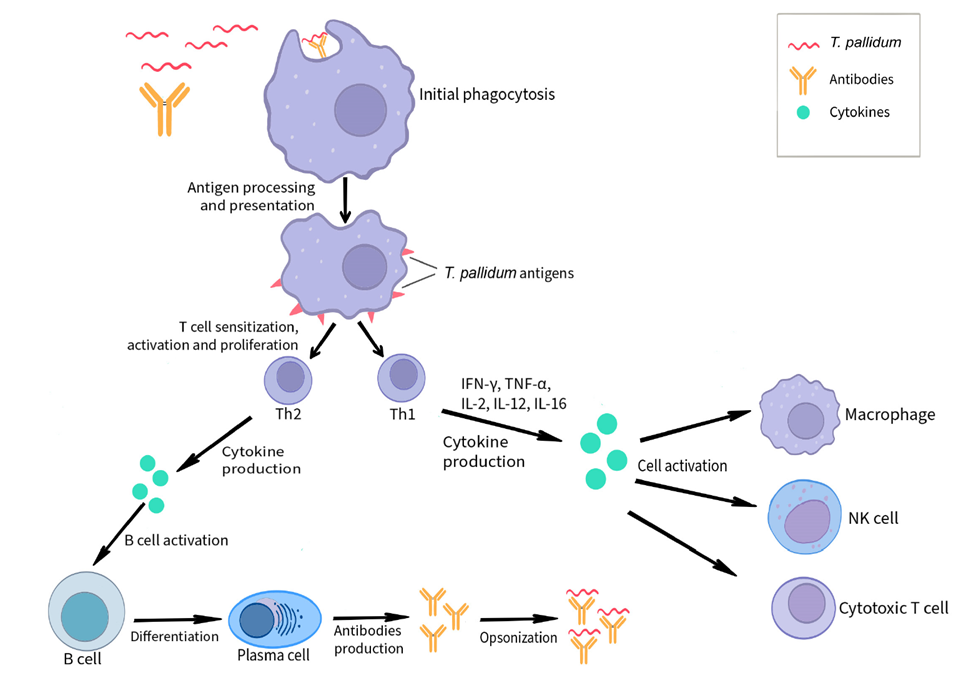

Figure 5. Schematic of the immune response during Treponema pallidum infection
Core Pathogenesis and Evasion Mechanisms of T. pallidum (2025 Latest Research Consensus)
- Adhesion and Invasion: Tp0751/TmpA mediate mucosal barrier penetration; helical axial filaments drive tissue migration
- Immune Evasion: Sparse outer membrane proteins (Tp92/Tp47), antigen variation, induction of neutrophil anti-apoptosis (ERK/PI3K/NF-κB pathways)
- Systemic Dissemination: Pro-inflammatory cytokines facilitate BBB/placental breach, leading to neurosyphilis/congenital infection
- Persistent Latency: Low metabolism + genome simplification support asymptomatic survival for years
Clinical Diagnosis of Treponema pallidum
For clinical diagnosis, common methods include dark-field microscopy, silver staining, nucleic acid amplification tests (NAATs), nontreponemal serological tests (e.g., RPR, TRUST), treponemal serological tests (e.g., TPPA, TP-ELISA), and cerebrospinal fluid analysis. NAATs offer high sensitivity and specificity and are useful for early diagnosis, treatment monitoring, and congenital syphilis confirmation, while CSF testing is primarily for confirming neurosyphilis. Treatment-wise, the World Health Organization states that syphilis can be completely cured with standard therapy; benzathine penicillin is the first-line drug, and early treatment is key to preventing progression and breaking transmission chains.
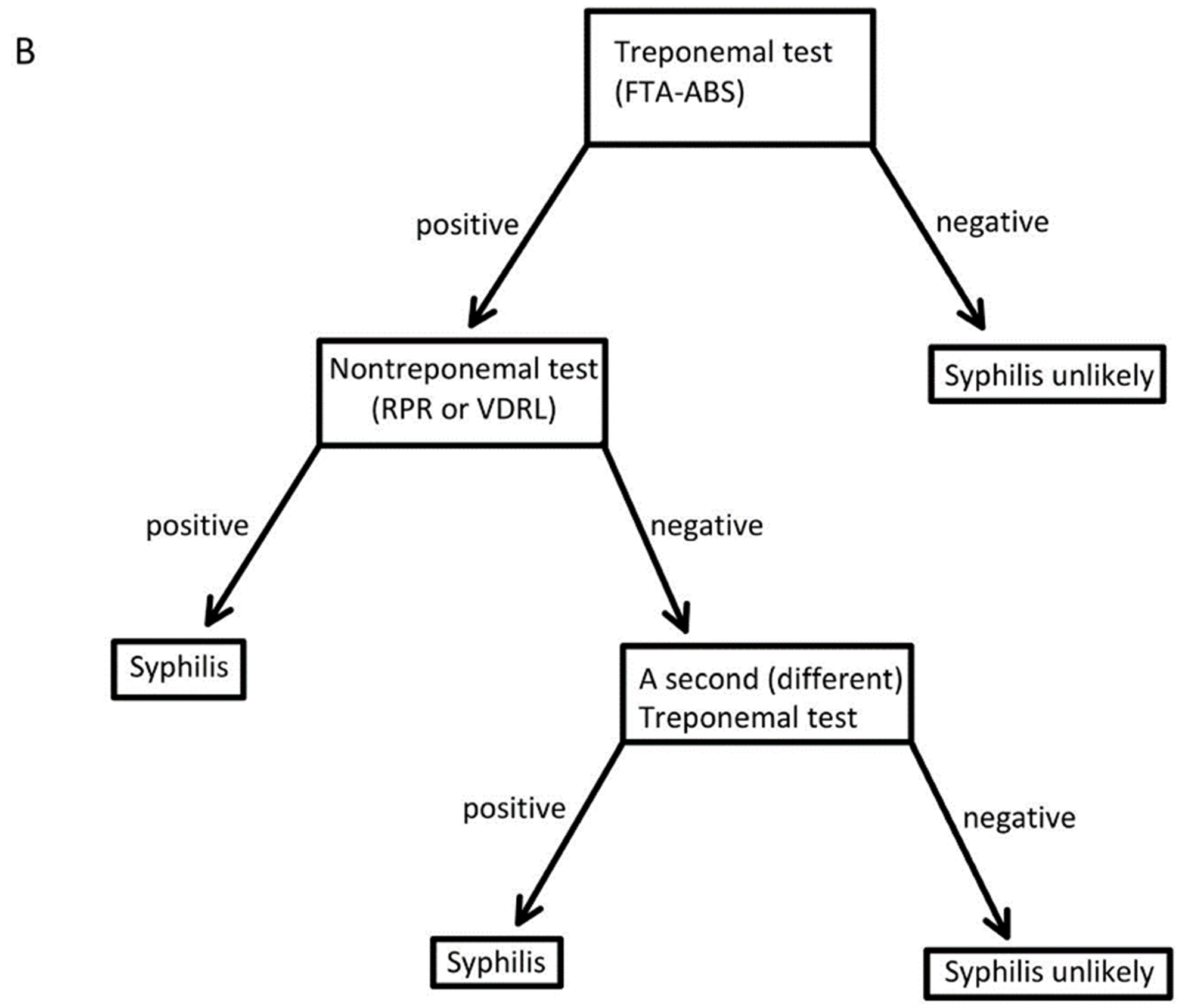

Figure 6. Diagnostic algorithm for syphilis testing
Key Clinical Points: Traditional serology (RPR/TPPA) combined with NAAT/CSF for neurosyphilis confirmation; benzathine penicillin achieves >95% cure rate in early stages, but antimicrobial resistance monitoring and vaccine development are urgently needed.
In 2025, NIAID-funded R21 projects encourage the use of in vitro culture, gene editing, and guinea pig congenital syphilis models to advance basic pathogenesis research. Long-term goals include effective vaccines, higher-sensitivity diagnostics, and enhanced surveillance systems to address the global resurgence. In resource-limited settings, priority should be given to point-of-care tools and equitable data collection.
abinScience Empowering Syphilis Research: Tp antigen recombinant proteins/polyclonal antibodies/ELISA kits covering key immune evasion targets (TPF1, TPP17, tmpA, TPP47, FlaB3), supporting mechanism validation, diagnostic development, and vaccine screening.
abinScience Latest Product Recommendations for Treponema pallidum
The following are abinScience's latest recombinant proteins and antibodies related to Treponema pallidum. Catalog numbers link directly to product pages.
Protein (Recombinant Proteins)
| Catalog No. |
Product name |
| JN906012 |
Recombinant Treponema pallidum/TP TPF1/Antigen 4D Protein, C-His |
| JN980012 |
Recombinant Treponema pallidum/TP TPP17/17 kDa lipoprotein Protein, N-GST |
| JN894012 |
Recombinant Treponema pallidum/TP TPP15 & tmpA & TPP47 Protein, C-His |
| JN038012 |
Recombinant Treponema pallidum/TP TPP47 Protein, C-His |
| JN858012 |
Recombinant Treponema pallidum/TP tmpA Protein, C-His |
| JN939012 |
Recombinant Treponema pallidum FlaB3 Protein, N-His-SUMO |
Antibody (Antibodies)
| Catalog No. |
Product name |
| JN906014 |
Anti-Treponema pallidum TPF1/Antigen 4D Polyclonal Antibody |
| JN939014 |
Anti-Treponema pallidum FlaB3 Polyclonal Antibody |
| JN858014 |
Anti-Treponema pallidum tmpA Polyclonal Antibody |
| JN038014 |
Anti-Treponema pallidum TPP47 Polyclonal Antibody |
| JN980014 |
Anti-Treponema pallidum TPP17 Polyclonal Antibody |
Kit (ELISA Kits)
| Catalog No. |
Product name |
| JN894018 |
Treponema pallidum TPP15 ELISA Kit |
| JN980018 |
Treponema pallidum TPP17 ELISA Kit |
| JN038018 |
Treponema pallidum TPP47 ELISA Kit |
Need help selecting reagents for your syphilis research project? Contact us: info@abinscience.com | +86-27-65523339
References
- Zhou Y, Xie Y, Xu M. Potential mechanisms of Treponema pallidum breaching the blood-brain barrier. Biomed Pharmacother. 2024;180:117478. doi:10.1016/j.biopha.2024.117478
- Ramírez-Amador V, Castillejos-García I, Maldonado-Mendoza J, et al. Exposing the Great Imitator: Proposal for a Holistic Diagnosis of Oral Secondary Syphilis in People Living with HIV. Head Neck Pathol. 2022;16(3):773-784. doi:10.1007/s12105-022-01446-5
- Ávila-Nieto C, Pedreño-López N, Mitjà O, et al. Syphilis vaccine: challenges, controversies and opportunities. Front Immunol. 2023;14:1126170. doi:10.3389/fimmu.2023.1126170
- Kaminiów K, Kiołbasa M, Pastuszczak M. The Significance of the Cell-Mediated Host Immune Response in Syphilis. Microorganisms. 2024;12(12):2580. doi:10.3390/microorganisms12122580
- Li W, Li S, Wang J, et al. The outer membrane protein Tp92 of Treponema pallidum delays human neutrophil apoptosis via the ERK, PI3K/Akt, and NF-κB pathways. Mol Microbiol. 2023;120(5):684-701. doi:10.1111/mmi.15164
- Liu J, Howell JK, Bradley SD, et al. Cellular architecture of Treponema pallidum: novel flagellum, periplasmic cone, and cell envelope as revealed by cryo electron tomography. J Mol Biol. 2010;403(4):546-561. doi:10.1016/j.jmb.2010.09.020
- Chaudhry S, Akinlusi I, Shi T, Cervantes J. Secondary Syphilis: Pathophysiology, Clinical Manifestations, and Diagnostic Testing. Venereology. 2023;2(2):65-75. doi:10.3390/venereology2020006

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский














