Lung cancer remains the leading cause of cancer-related deaths worldwide. Most patients are diagnosed at an advanced stage, and overall survival rates still need improvement. In recent years, immune checkpoint inhibitors (ICIs) have brought new hope to some lung cancer patients, yet a large proportion show no response to ICI therapy, and the underlying resistance mechanisms are not fully understood. Effective anti-tumor immune responses rely on antigen-presenting cells (APCs, including dendritic cells (DCs) and macrophages) to efficiently capture, process, and present tumor antigens, thereby activating T cell-mediated anti-tumor effects. However, dysfunction of APCs in the tumor microenvironment impairs T cell activation, ultimately promoting tumor progression and resistance to immunotherapy.
Serum amyloid A (SAA) is an acute-phase pro-inflammatory protein that is highly expressed in various cancers and contributes to pro-tumor responses. Previous studies have shown that SAA secreted by lung cancer stem cells promotes tumor progression by suppressing TH1 immunity, but the specific mechanisms by which SAA regulates APC function and T cell activation remain to be fully elucidated. A research team from Nanjing Medical University and other institutions recently published an original study in Frontiers in Immunology that systematically reveals the molecular mechanism by which SAA inhibits antigen presentation and weakens T cell-mediated anti-tumor immunity via the CD36 pathway, providing a new target and strategy for lung cancer immunotherapy.
Linking SAA to the Immunosuppressive Microenvironment
The research team first analyzed single-cell RNA sequencing data from lung adenocarcinoma (LUAD) patient samples using the TISCH2 database. Results showed that SAA is significantly overexpressed in malignant cell populations in LUAD, and the proportion of SAA-high subsets in metastatic lesions was higher than in primary tumors among both malignant cells and macrophages. Functional analysis revealed a close association between SAA-high groups and differential expression of antigen presentation-related genes. Cell-cell communication analysis further confirmed strong interactions between SAA-high malignant subsets and various APCs. These bioinformatics findings suggest that SAA signaling is closely linked to the immunosuppressive microenvironment in lung adenocarcinoma and may regulate antigen presentation processes.
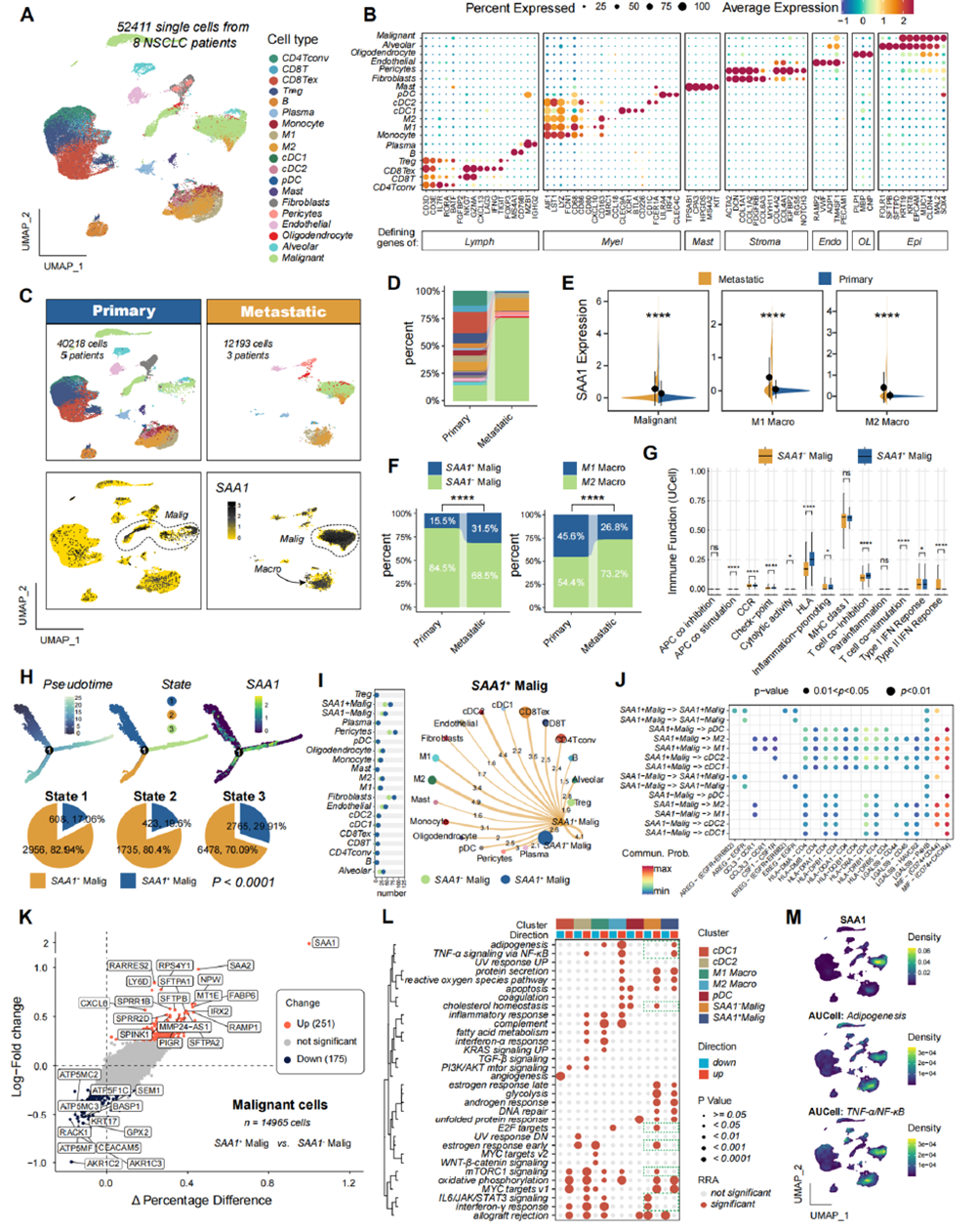
Figure 1. Single-cell transcriptome analysis and characterization of SAA1+ malignant cells in the NSCLC microenvironment
Elucidating the Direct Impact of SAA on T Cell Differentiation
To clarify SAA’s regulatory role in T cell function, the team established an in vitro autologous T cell-tumor organoid co-culture system, with control, recombinant SAA-treated, and SAA-neutralizing antibody (a-SAA)-treated groups. Flow cytometry showed that recombinant SAA significantly reduced the proportions of CD4+ and CD8+ effector T cells while markedly increasing CD8+ central memory T cells. In contrast, SAA neutralization enhanced effector T cell responses and suppressed memory cell formation. These results indicate that SAA directly modulates T cell subset differentiation in the tumor microenvironment, inhibiting effector T cell generation and thereby weakening anti-tumor immune responses.
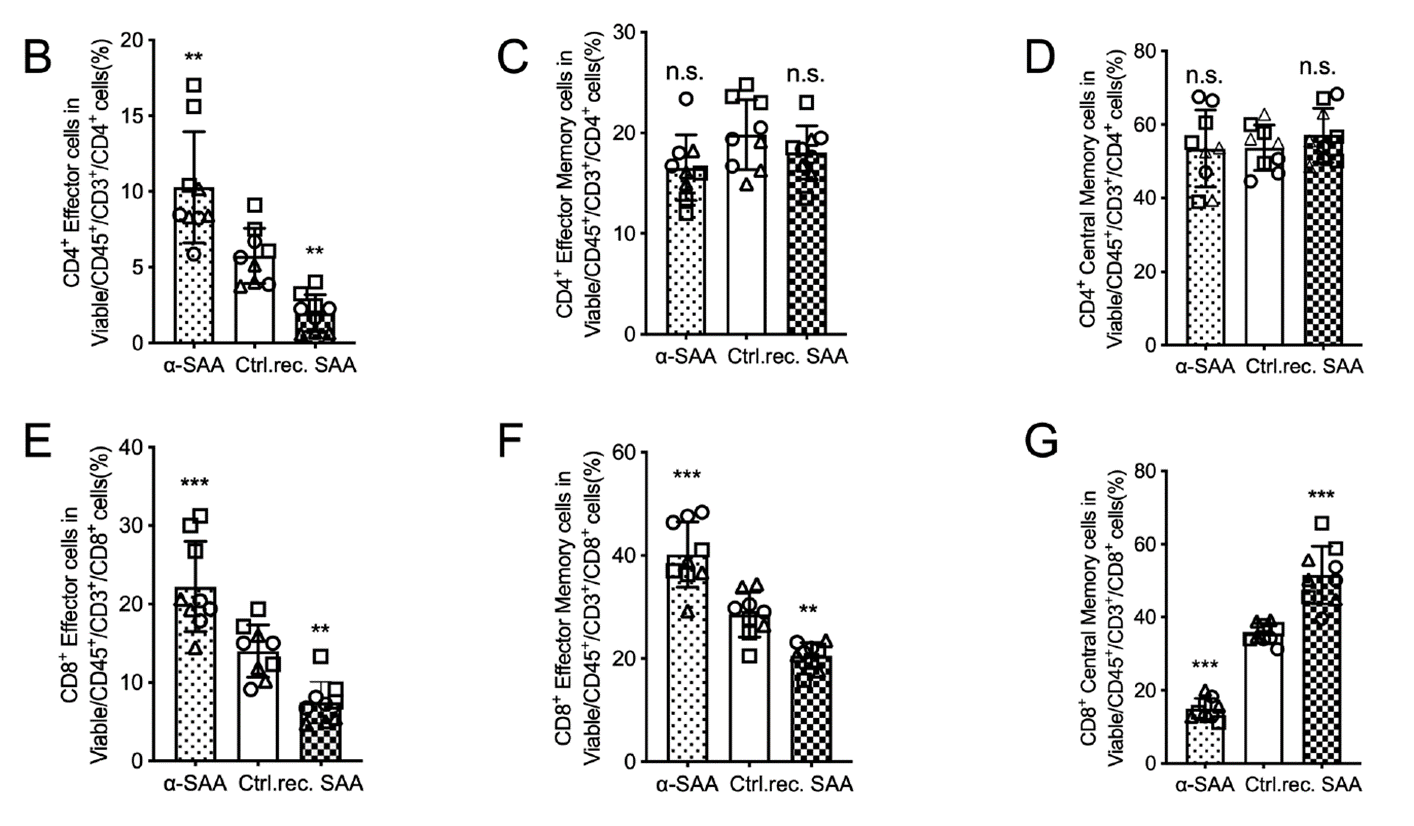
Figure 2. SAA exposure alters T cell subset distribution in tumor organoid co-cultures
Revealing Dual Inhibition of APC Function by SAA
1. At the Dendritic Cell (DC) Level
Using a co-culture system of human peripheral blood mononuclear cells (PBMCs) with tumor organoids, the team found that recombinant SAA treatment significantly reduced the proportion of DCs among surviving CD45+ cells and decreased the activated (CD80+) phenotype within the DC population. In contrast, a-SAA treatment restored or even enhanced DC expansion and activation. Cytokine profiling further showed that SAA signaling markedly affected the release of IL-1β, IFN-γ, and other cytokines critical for T cell maturation and anti-tumor immunity. These findings confirm that SAA inhibits DC expansion and activation, impairing their antigen-presenting capacity.
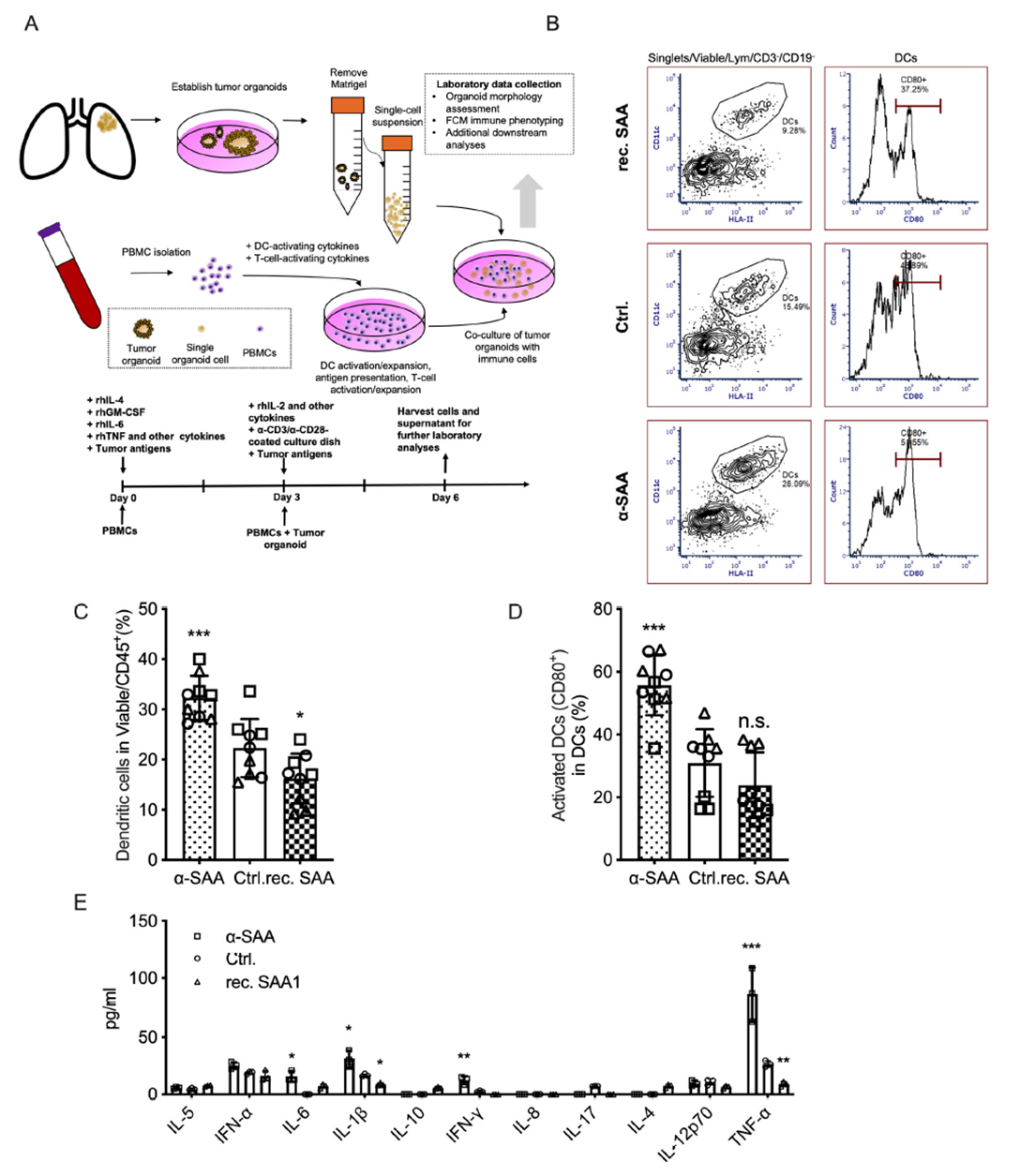
Figure 3. SAA inhibits dendritic cell expansion and activation
2. At the Macrophage Level
PBMCs were differentiated into macrophages using M-CSF and treated in a physiological activation environment simulated by tumor antigens and low-dose LPS. Results showed that recombinant SAA reduced the proportion of macrophages among surviving CD45+ cells, significantly decreased M1-type (CD86+) macrophages, and increased M2-type (CD206+) macrophages. Proliferation assays further indicated that SAA drove M1 macrophages toward a quiescent state, while a-SAA reversed this effect. These data suggest that SAA inhibits macrophage activation and promotes polarization toward the immunosuppressive M2 phenotype.
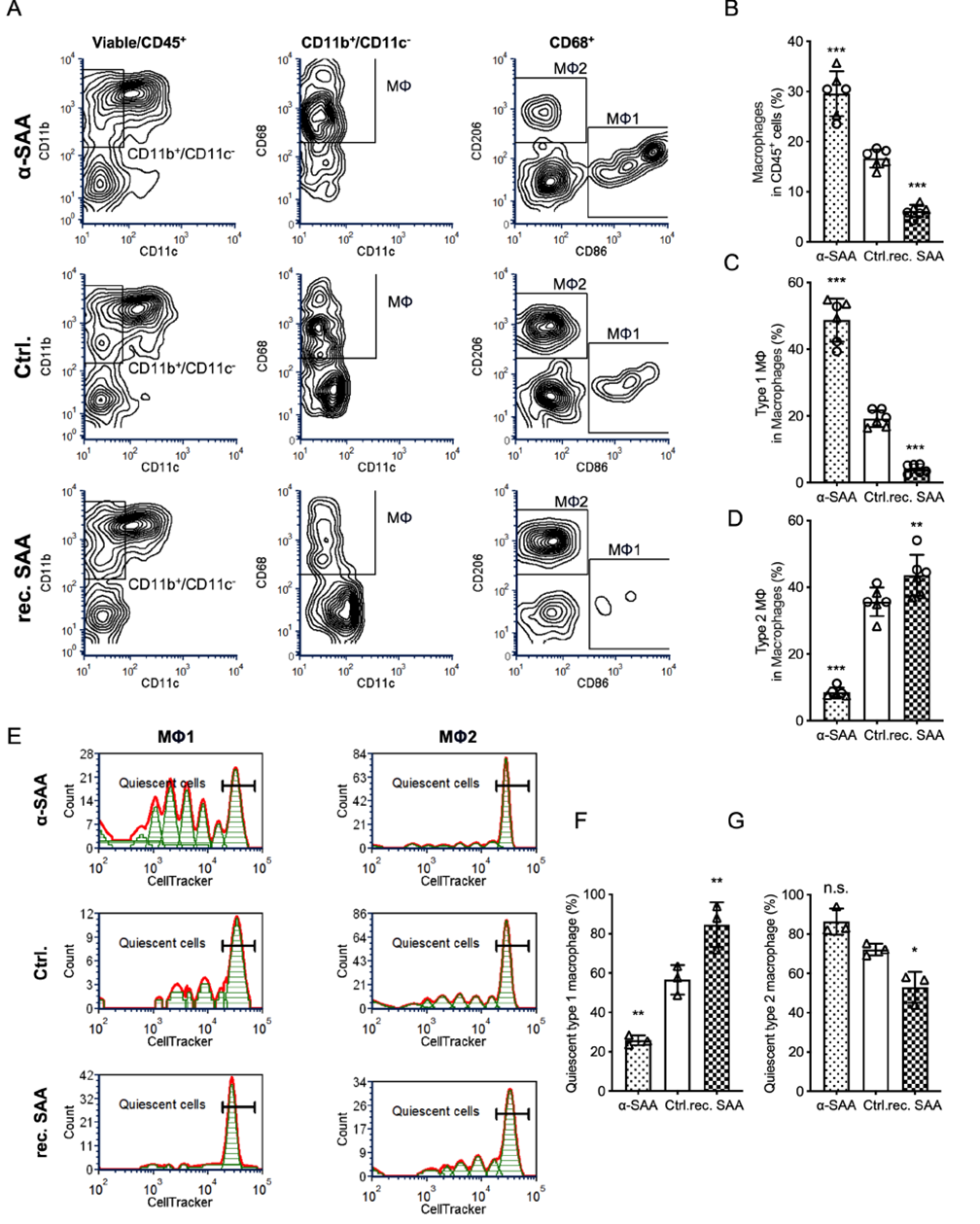
Figure 4. SAA inhibits macrophage activation and promotes M2 polarization
Validating SAA’s Regulation of Tumor Growth and T Cell Infiltration
The team established subcutaneous tumor models in mice using SAA-overexpressing (SAAOE) and wild-type LLC cells, treated with PD-1 antibody combined with a-SAA or isotype control. Tumors in the SAAOE group were significantly larger and heavier than controls, while SAAOE + a-SAA treatment markedly slowed tumor growth. Flow cytometry confirmed that SAA overexpression dramatically reduced T cell and CD8+ T cell infiltration in the tumor microenvironment, whereas a-SAA restored CD8+ T cell infiltration. Subset analysis showed decreased effector CD8+ T cells (CD44+CD62L−), increased naïve T cells, and reduced granzyme B+ cytotoxic T cells in the SAAOE group—all reversed by a-SAA. Immunohistochemistry further validated a-SAA’s restoration of CD8+ T cell infiltration. These in vivo data demonstrate that SAA promotes tumor growth by limiting cytotoxic T cell recruitment and functional differentiation, while SAA neutralization effectively enhances anti-tumor immunity.
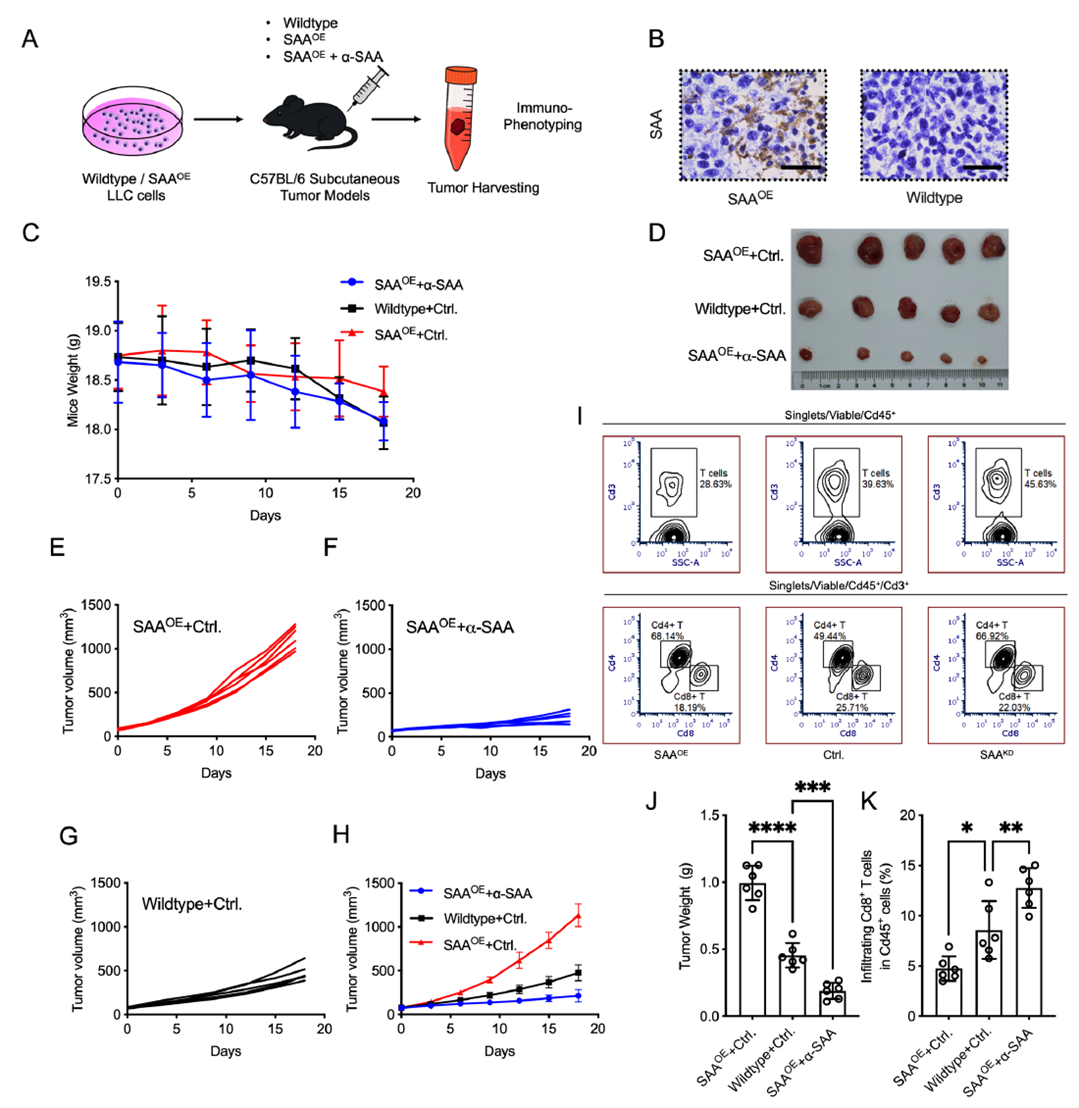
Figure 5. SAA expression enhances in vivo tumor growth
Confirming SAA’s Inhibitory Effect on Macrophage Polarization
Given macrophages’ central role in initiating and sustaining T cell responses, the team performed detailed flow cytometry on tumor-infiltrating macrophages. Compared to wild-type controls, the SAAOE + Ctrl group showed significantly reduced total macrophage (Cd45+Cd11b+F4/80+) infiltration, which was restored by SAAOE + a-SAA. Polarization analysis revealed that SAAOE markedly lowered M1 (CD86+) frequency and activated MHC-II+ M1 subsets, while a-SAA promoted M1 polarization and upregulated MHC-II expression. Conversely, M2 macrophage proportions were relatively higher in the SAAOE group and reduced by a-SAA. These in vivo results confirm that SAA suppresses macrophage infiltration, M1 polarization, and activation, thereby exacerbating T cell activation deficits.
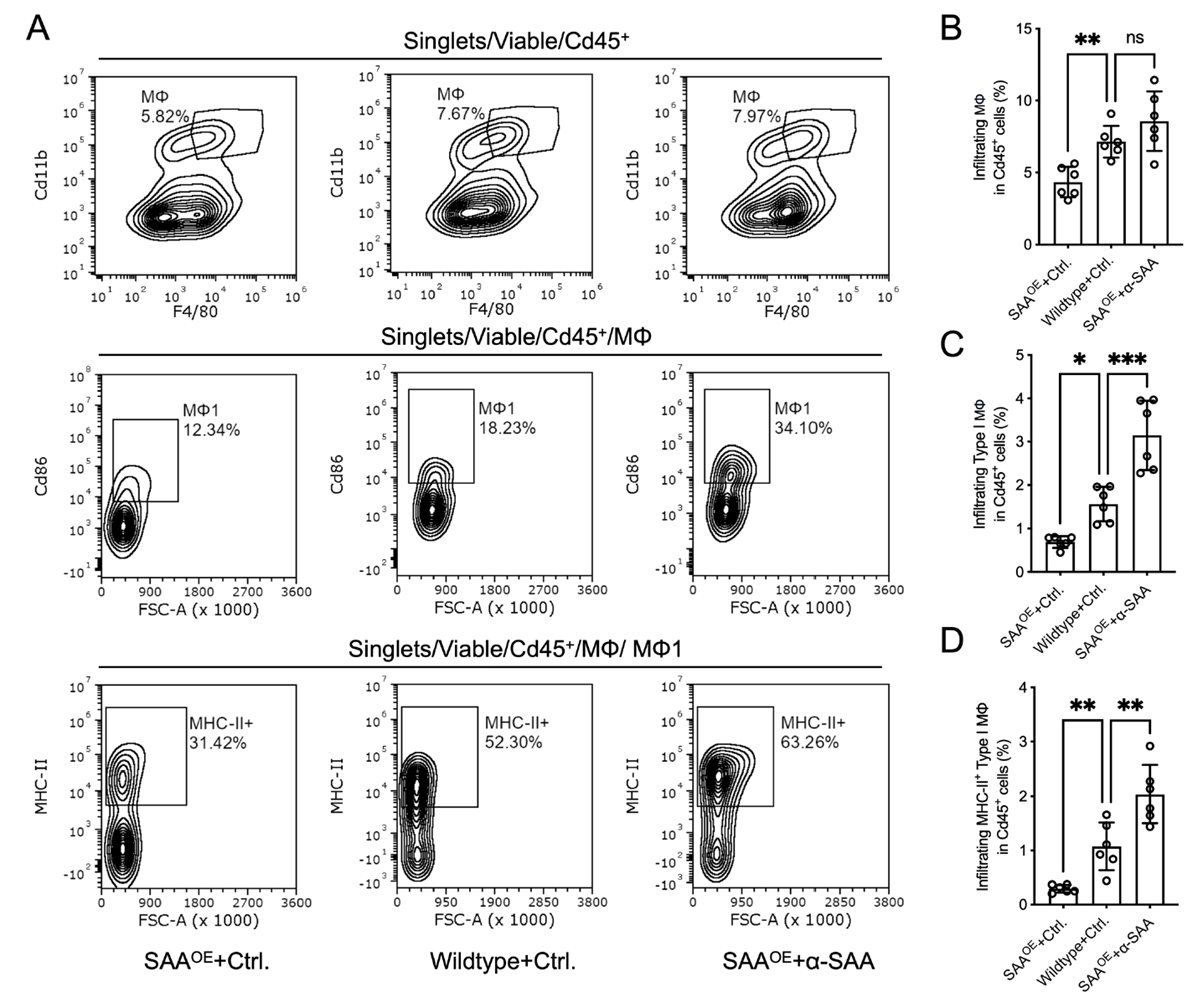
Figure 6. SAA inhibits macrophage infiltration and M1 antigen-presenting activation in vivo
Defining the SAA-Macrophage-T Cell Regulatory Axis
To directly test whether SAA affects T cell activation via macrophage regulation, the team conducted macrophage pretreatment followed by co-culture experiments. PBMC-derived macrophages were pretreated with recombinant SAA, a-SAA, or control for 5 days, then co-cultured with autologous non-adherent PBMCs and tumor cells under IL-2 and CD3/CD28 stimulation. SAA-pretreated macrophages significantly reduced CD8+ T cell proportions, granzyme B+ cytotoxic T cell frequency, and tumor cell killing (measured by LDH release). In contrast, a-SAA-pretreated macrophages restored T cell activation and cytotoxicity to levels comparable to or exceeding controls. These results directly confirm that SAA indirectly suppresses CD8+ T cell activation and anti-tumor activity by shaping an immunosuppressive macrophage phenotype, while SAA neutralization relieves this suppression.
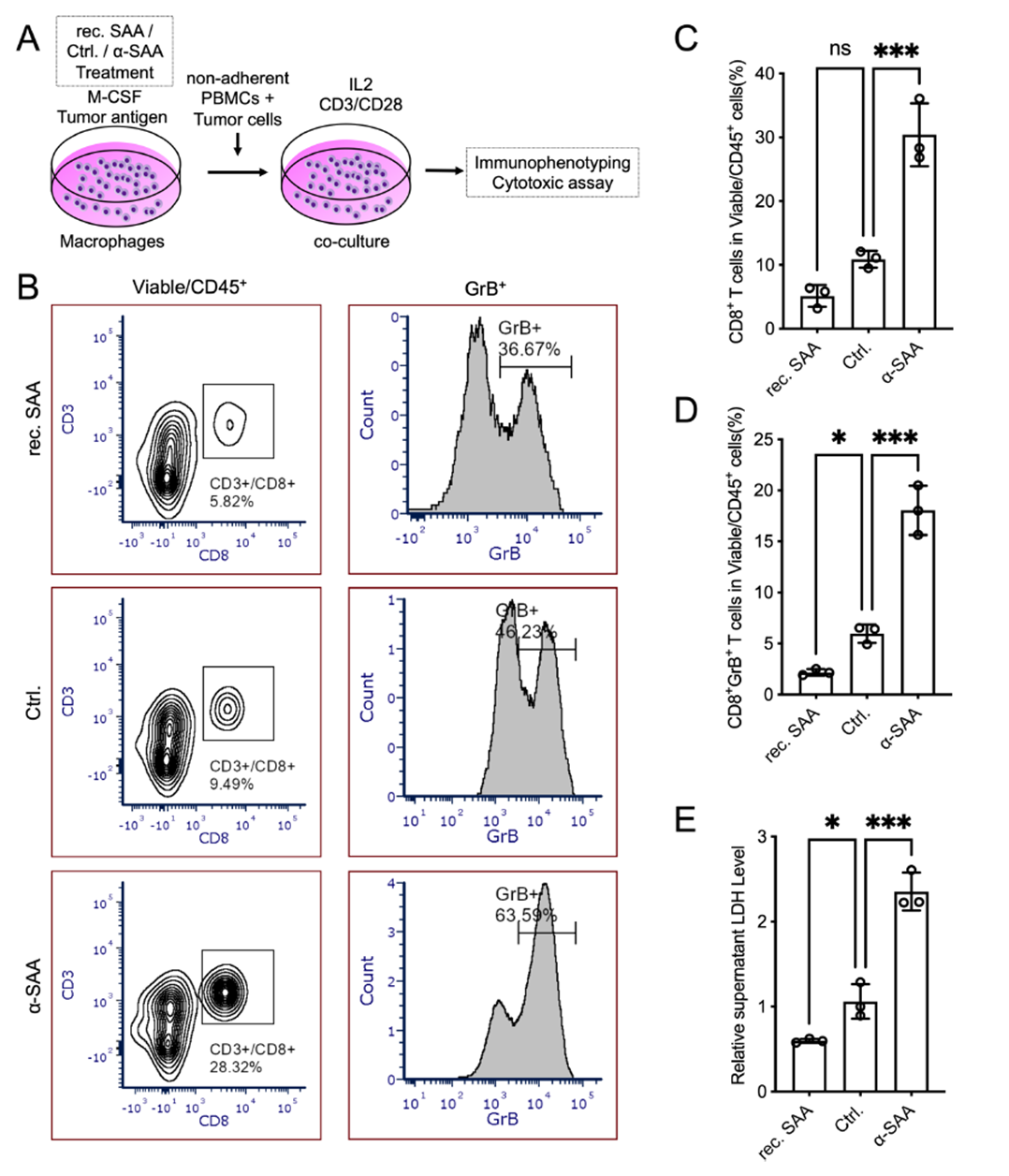
Figure 7. SAA-treated macrophages suppress T cell activation and cytotoxicity in co-culture
Identifying the Central Role of the SAA-CD36 Axis
To uncover the molecular pathway underlying SAA’s regulation of APCs, the team performed receptor inhibitor experiments in THP1 cells. The TLR2/4 pathway inhibitor TIRAPi failed to significantly reverse SAA’s inhibition of HLA-DR expression, whereas the CD36 inhibitor SMS121 restored HLA-DR+ cell proportions in a dose-dependent manner, alleviating SAA’s suppressive effect. This indicates that SAA primarily inhibits macrophage maturation and activation through the scavenger receptor CD36 rather than TLR2/4, establishing the SAA-CD36 axis as critical for macrophage regulation.
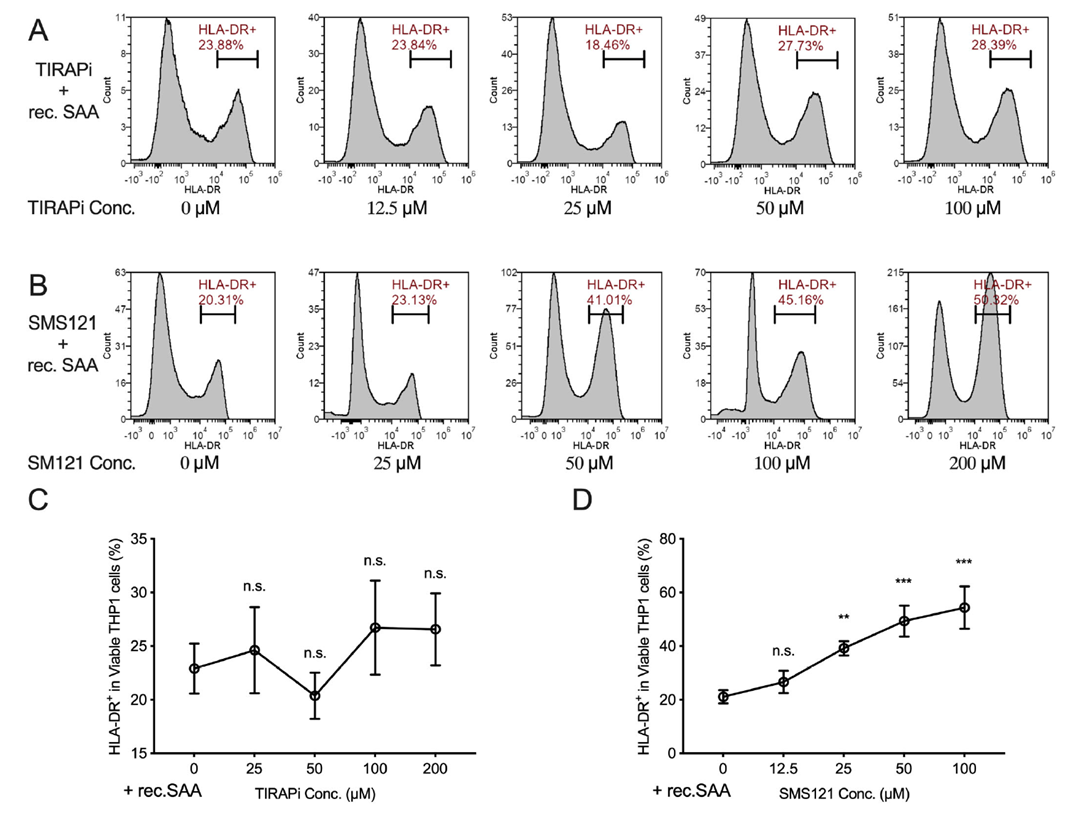
Figure 8. SAA restricts macrophage maturation through interaction with CD36
Through comprehensive bioinformatics analysis, in vitro cellular experiments, and in vivo animal models, this study provides the first complete elucidation of the molecular mechanism underlying SAA-mediated immunosuppression in lung cancer: Tumor-derived SAA binds CD36 to dually inhibit DC and macrophage activation and antigen presentation, drive macrophage M2 polarization, and subsequently impair CD8+ T cell recruitment, differentiation, and cytotoxicity—ultimately promoting tumor progression and immunotherapy resistance. The study clarifies SAA’s dual regulation of the APC-T cell axis in the tumor microenvironment, identifies CD36 as the key receptor for its immunosuppressive function, and demonstrates that SAA-neutralizing antibodies effectively restore anti-tumor immunity, offering a new therapeutic target for lung cancer. Combination with PD-1 inhibitors or improved efficacy in SAA-high patients requires further clinical validation.
abinScience provided the critical SAA-neutralizing antibody (catalog no.: HY110010) for this study. It not only neutralized recombinant SAA function in in vitro co-culture experiments—validating specificity for T cell differentiation, DC activation, and macrophage polarization inhibition—but also, when administered intraperitoneally in tumor-bearing mice, successfully reversed SAA overexpression-induced tumor growth acceleration and immunosuppressive phenotypes. This directly confirmed the therapeutic potential of targeting SAA and provided essential support for elucidating the SAA-macrophage-T cell axis and the role of the CD36 pathway, serving as a key tool for validating the study’s core hypotheses.
Disclaimer: This article is a summary based on publicly available literature. The product is intended for research use only.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский










