Crohn’s disease (CD) is one of the major subtypes of inflammatory bowel disease (IBD). It can affect any part of the gastrointestinal tract from the mouth to the rectum, most commonly involving the terminal ileum and adjacent colon. Typical histological features include mucosal erosion, transmural inflammation, granuloma formation, and secondary fibrosis and fistula development. Clinically, it often presents with chronic abdominal pain, diarrhea, weight loss, anemia, and nutritional deficiencies, and may be accompanied by extraintestinal manifestations involving multiple systems, such as immune-related lesions in joints, skin, and eyes.
The pathogenesis of CD remains incompletely understood but is widely recognized to involve genetic susceptibility, gut microbiota dysbiosis, and immune dysfunction. Genetically, more than 200 risk loci have been identified, with mutations in genes such as NOD2 and ATG16L1 impairing intestinal barrier function and bacterial recognition. Gut microbiota imbalance—characterized by enrichment of Enterobacteriaceae and reduced mucosal microbial diversity—triggers inflammation through mucosal penetration and metabolic disruption. Immune abnormalities primarily manifest as aberrant myeloid-stromal cell crosstalk, excessive activation of TH1/TH17 cells, and massive release of proinflammatory cytokines (TNF, IL-17, OSM, etc.), ultimately leading to chronic intestinal inflammation and tissue damage.


Figure 1. Crohn’s disease-associated genetic mutations, intestinal abnormalities and pathobionts.
Core Targets and Mechanisms in Crohn’s Disease Research
The pathological network of CD involves multiple pathways, including intestinal barrier function, immune regulation, and microbial metabolism. The following key targets provide critical directions for disease intervention:
| Target |
Normal Biological Function |
Pathological Role in CD |
Research Applications |
| TNFSF15 |
Regulates immune responses and inflammation |
Specific SNPs (rs6478108/rs4574921) associated with stenotic phenotype and perianal fistula formation |
Disease phenotype prediction, fistula mechanism studies |
| TNFA |
Regulates immune responses and inflammation |
Overactivation amplifies intestinal inflammation and promotes tissue damage |
Anti-TNF therapy validation, inflammation activity detection |
| KLRK1 |
Expressed on natural killer cells; involved in immune surveillance and cytotoxicity |
Impaired immune cell killing function and compromised mucosal defense barrier |
Innate immunity studies, mucosal protection mechanism validation |
| TGFBR1 |
Mediates TGF-β signaling; regulates cell proliferation, differentiation, and repair |
Signaling defects lead to impaired intestinal tissue repair and promote fibrosis |
Fibrosis mechanism research, tissue repair interventions |
| S1PR1 |
Regulates lymphocyte trafficking, vascular homeostasis, and immune cell activation |
Mediates abnormal lymphocyte homing to the gut, exacerbating mucosal inflammation |
Migration signaling studies, immune cell homing interventions |
| BTK |
Involved in B-cell activation and signaling; regulates immune complex formation |
Drives aberrant B-cell activation and autoantibody production, worsening intestinal immune dysregulation |
BTK inhibitor validation, humoral immunity analysis |
| RIPK1 |
Regulates apoptosis and necroptosis; participates in inflammatory signaling |
Mediates programmed necrosis of intestinal epithelial cells, amplifying mucosal inflammation and tissue damage |
Cell death mechanism studies, anti-inflammatory target screening |
| GIRDIN |
Involved in cytoskeletal remodeling, cell migration, and signaling |
Regulates intestinal epithelial cell migration and repair; dysfunction exacerbates mucosal injury |
Epithelial barrier repair studies, tissue regeneration interventions |
| NLRP3 |
Forms inflammasome; mediates proinflammatory cytokine release |
Excessive inflammasome activation increases IL-1β secretion, aggravating intestinal inflammation |
Inflammasome inhibitor screening, innate immune regulation studies |
| NOD2 |
Recognizes bacterial peptidoglycan; initiates innate immunity and intestinal barrier regulation |
Mutations impair bacterial clearance and cause microbiota dysbiosis, increasing disease susceptibility; associated with stenotic phenotype and surgical risk |
Genetic mechanism studies, host-microbiota interaction validation, precision medicine target |
Recent Breakthroughs in Crohn’s Disease Research
In recent years, technologies such as single-cell sequencing and multi-omics have led to several groundbreaking discoveries in CD research:
- Identification of a specific LND epithelial cell subset highly expressing LCN2, NOS2, and DUOX2. This subset expands significantly in active CD, drives mucosal inflammation by recruiting immune cells, and its proportion can predict response to anti-TNF therapy.
- Confirmation that aberrant myeloid-stromal cell crosstalk is central to perianal fistula pathogenesis, with CHI3L1+ fibroblasts co-expressing fibrotic and destructive gene signatures; the AP-1 transcription factor family regulates key inflammatory genes.
- Discovery of pathobionts in the gut microbiota (e.g., adherent-invasive Escherichia coli [AIEC] and Ruminococcus species) that promote inflammation by degrading the mucus layer and cross-feeding metabolites, providing a basis for microbiota-targeted therapies.
- Evidence of ethnic differences in CD pathogenesis: African-descended patients show abnormal myeloid cell differentiation, elevated CHI3L1 and OSM expression, higher fistula incidence, and more severe disease.
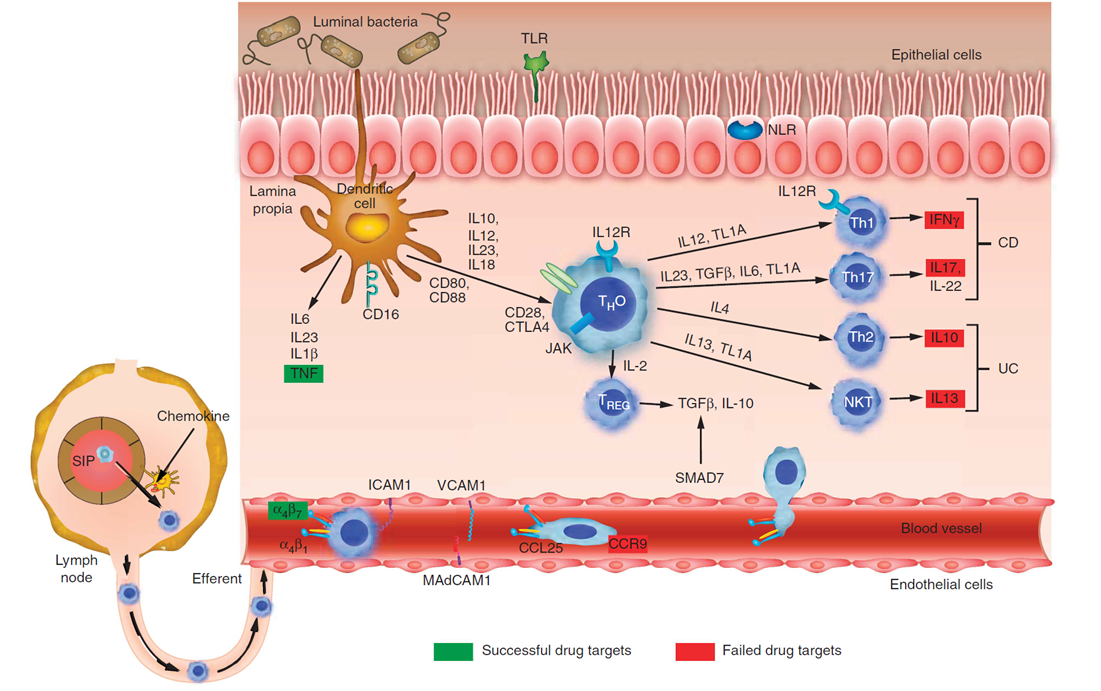
Fig. 2. Drug targets in inflammatory bowel disease (IBD).
Core Challenges in Crohn’s Disease Research
Crohn’s disease research continues to face several unresolved challenges:
- At the etiological level, the “cause-versus-consequence” paradox of gut microbiota dysbiosis remains unsolved—it is unclear whether dysbiosis triggers disease or results from inflammation. High inter-individual variability and the lack of universal “disease-specific” microbial markers complicate the issue.
- In precision diagnostics and treatment, validated prognostic biomarkers are lacking, making it difficult to predict disease progression (e.g., strictures or fistulas) or drug response (e.g., anti-TNF failure risk). Diagnosis relies on nonspecific inflammatory markers (CRP, fecal calprotectin), small bowel lesions are easily missed, and global diagnostic and management protocols lack standardization, particularly for complications such as intestinal fibrosis or cutaneous manifestations.
- Therapeutically, current options (biologics, surgery) offer limited long-term efficacy. Median duration of anti-TNF response is only 2–3 years. Fecal microbiota transplantation (FMT) shows promise but suffers from inconsistent results due to small sample sizes and methodological variability. Head-to-head trial data for newer agents (e.g., IL-23 inhibitors) are insufficient to establish optimal treatment sequencing.
Additionally, research has historically focused on European and North American populations, leaving data gaps for regions with rising incidence (Asia, Africa) and failing to capture ethnic/regional disease heterogeneity. Interactions between environmental triggers (diet, timing of microbial exposure) and genetic susceptibility also remain poorly defined. These challenges underscore the need for multi-target, multi-dimensional research tools—abinScience provides targeted reagents for core pathways to help overcome these bottlenecks.
Research Significance: The dual driving mechanism of LND cell abnormalities and gut microbiota dysbiosis explains the core mystery of CD recurrence, offering new insights for developing novel combination intervention strategies.
Below is the latest selection of abinScience recombinant proteins, antibodies, and kits relevant to Crohn’s disease research. Catalog numbers link directly to product pages.
Recombinant Proteins
| Catalog No. |
Product name |
| HF879012 |
Recombinant Human TNFa/TNF-alpha Protein, N-His |
| HY213011 |
Recombinant Human CSF2/GM-CSF Protein, C-His |
| HY499012 |
Recombinant Human CD3D Protein, N-His |
| HY328021 |
Recombinant Human IL6 Protein, C-His |
| HY596012 |
Recombinant Human CD49d/ITGA4 Protein, N-His |
| HB829012 |
Recombinant Human JAK1 Protein, N-His |
| HB769011 |
Recombinant Human IL12B/IL-12 p40/NKSF2 Protein, C-Flag |
| HW621012 |
Recombinant Human Ki67/MKI67 Protein, N-His |
| HC260012 |
Recombinant Human BTK Protein, N-His |
| HC317012 |
Recombinant Human RIPK1 Protein, N-His |
View More Recombinant Proteins
| Catalog No. |
Product name |
| HS856011 |
Recombinant Human IL17A Protein, C-His |
| HV430012 |
Recombinant Human AHI1 Protein, N-His |
| HV466012 |
Recombinant Human IL23A Protein, N-His |
| HV077012 |
Recombinant Human NOD1 Protein, N-His |
| HW774012 |
Recombinant Human TGFBR1 Protein, N-His |
| HV001012 |
Recombinant Human NLRP3 Protein, N-His |
| HB123011 |
Recombinant Human CD314/KLRK1 Protein, C-His |
| HY328031 |
Recombinant Human IL6 Protein, C-Fc |
| HY213022 |
Recombinant Human CSF2/GM-CSF Protein, C-His |
| HW774011 |
Recombinant Human TGFBR1 Protein, C-Fc |
| HF879021 |
Recombinant Human TNFa/TNF-alpha Protein, N-His-Avi |
| HB829022 |
Recombinant Human JAK1 Protein, C-His |
| HY328521 |
Biotin-labeled Recombinant Human IL6 Protein, C-His |
Antibodies
View More Antibodies
| Catalog No. |
Product name |
| HV466056 |
Research Grade Mirikizumab |
| HF879106 |
Research Grade Lenercept |
| HF879116 |
Research Grade Opinercept |
| HF879126 |
Research Grade Tulinercept |
| HF879036 |
Research Grade Infliximab |
| HF879026 |
Research Grade Adalimumab |
| HV466026 |
Research Grade Tildrakizumab |
| HY213046 |
Research Grade Gimsilumab |
| HY213066 |
Research Grade Anti-Human CSF2/GM-CSF (KB002) |
| HS856136 |
Research Grade Anti-Human IL17A (ABM59) |
| HV466116 |
Research Grade Anti-Human IL23A/IL-23p19 (QX 004N) |
| HB769023 |
Anti-Human IL12B/IL-12 p40/NKSF2 Antibody (SAA2017) |
Kits
View More Kits
| Catalog No. |
Product name |
| AF879518 |
Anti-Etanercept Neutralizing Antibody ELISA kit |
| AB769518 |
Anti-Ustekinumab Neutralizing Antibody ELISA kit |
| AS856518 |
Anti-Secukinumab Neutralizing Antibody ELISA kit |
| AF879058 |
Anti-Etanercept ELISA Kit |
| AF879068 |
Anti-Golimumab ELISA Kit |
For research use only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский





