Date de publication :
2026-03-25 Nombre de vues : 1
In multicentre vaccine studies and infection-immunity monitoring, generating functionally comparable T-cell data across different species and laboratory environments remains a central technical challenge. OMIP-016 offers a 10-colour functional flow cytometry panel validated for both human and macaque PBMCs. Through the incorporation of a post-fixation freezing strategy, a permissive but well-controlled gating approach and a refined combination of activation markers, the panel substantially improves the sensitivity and inter-laboratory consistency of antigen-specific T-cell measurements.
1. OMIP-016 Panel
|
Target
|
Fluorochrome
|
Function
|
|
Dead Cells
|
Blue fluorescent dye
|
Exclude dead cells
|
|
CD3
|
APC-Cy7
|
T cell lineage
|
|
CD4
|
PerCP-Cy5.5
|
|
CD8
|
V500
|
|
CD45RA
|
PE-Cy7
|
Memory/differentiation marker
|
|
CD154
|
FITC
|
CD4 T-cell activation
|
|
MIP-1β
|
PE
|
Function
|
|
IFN-γ
|
V450
|
|
TNF-α
|
Alexa 700
|
|
IL-2
|
APC
|
2. Gate Logic
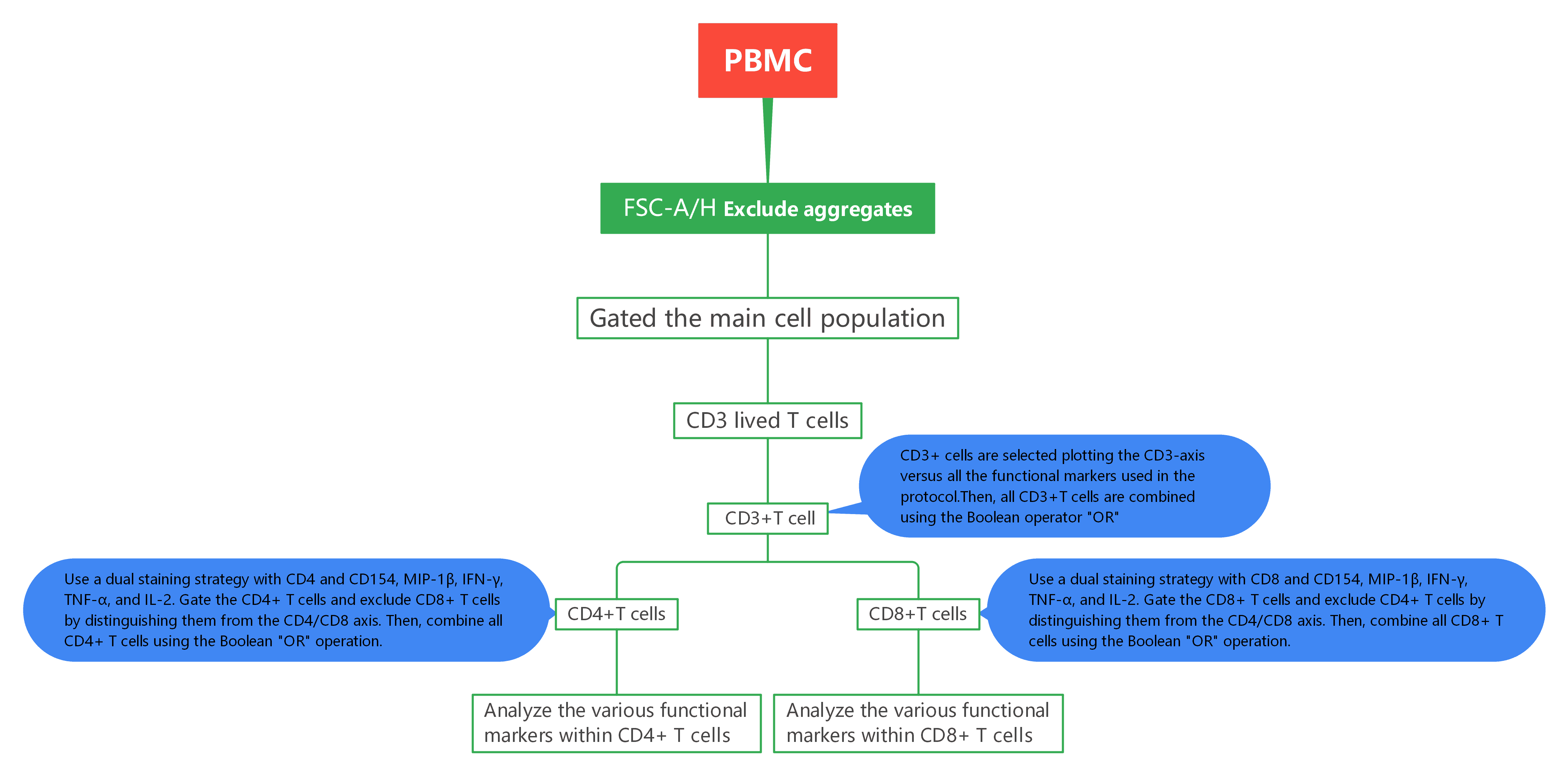
3. Experimental Results
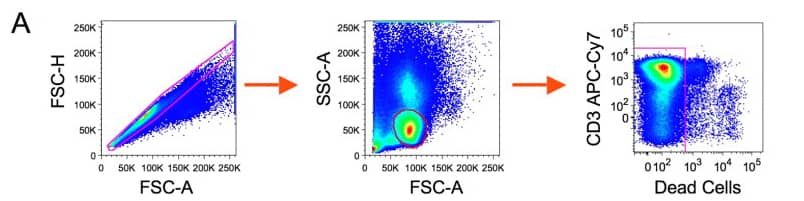
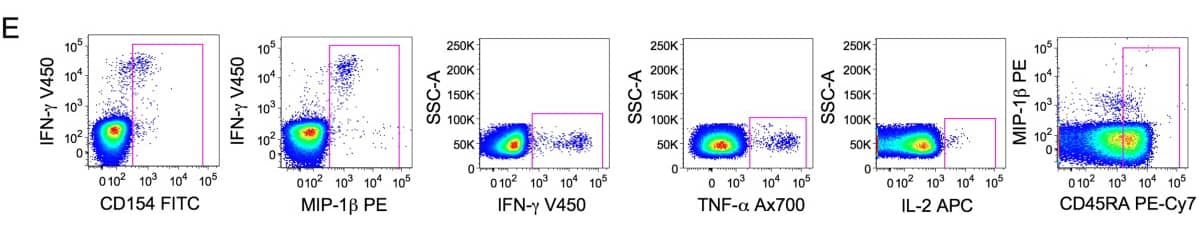
1). Exclude aggregates and dead cells, then identify live CD3 T cells.
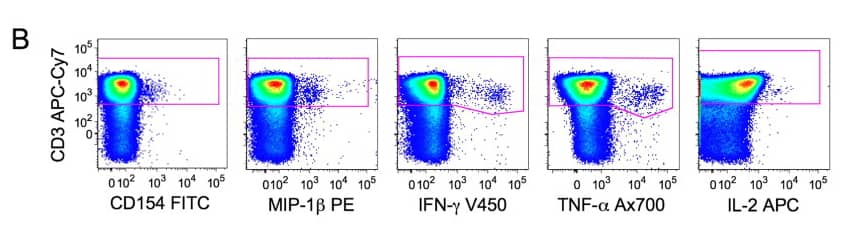
2). CD3+ cells are selected plotting the CD3-axis versus all the functional markers used in the protocol. The CD3+ population is calculated combining all the gates shown in (B) using the Boolean operator ‘‘OR’’.
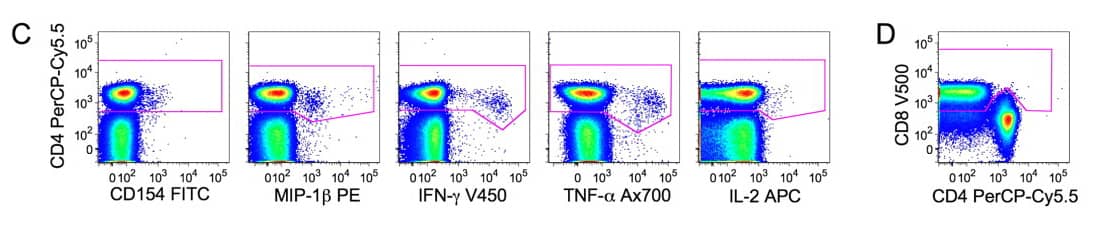
3). CD4+cells are selected taking in account the CD4 downregulation. The CD8 gate in (D) was used to discriminate functional positive CD4 T cells with a downregulated CD4 from functional positive CD8. Total CD4+cells are then obtained by gates in (C) combined by the operator ‘‘OR’’ and the exclusion of the CD8 cells identified in (D).
4). Live lymphocytes expressing CD3 and CD4 are then analyzed according to the expression of five functional markers and the differentiation marker CD45RA. Combination of axis was chosen to allow easy positioning of gates and optimal discrimination between positive and negative events.
4. Protocol Interpretation
1). Parallel optimisation across species: improving human–macaque translational relevance OMIP-016 was developed through simultaneous titration and validation of antibodies in both human and macaque samples. Reagents that performed reliably at the same working concentration in both species were prioritised, minimising signal discrepancies caused by species-dependent binding differences. This allows more reliable “horizontal” comparisons within a single panel and workflow, directly strengthening the translational value of findings between preclinical and clinical stages.
2). Incorporation of CD154 and MIP-1β to broaden the detection of CD4 and CD8 responses
OMIP-016 intentionally incorporates CD154 (CD40L) to identify activated antigen-specific CD4 T cells, and includes MIP-1β to enhance the detection of CD8 responses. The value here does not lie in adding extra markers per se, but in the functional complementarity between them. CD154 reveals activated CD4 subsets that may not secrete classical cytokines yet are antigen-responsive, while MIP-1β strengthens sensitivity relative to traditional IFN-γ and TNF-α readouts. Together, these markers provide a more comprehensive assessment of the breadth of T-cell reactivity.
3). Post-fixation freezing strategy: a practical solution for multicentre workflows
A key operational feature of OMIP-016 is the introduction of a freeze interval after fixation and permeabilisation. Samples can be frozen in Perm/Wash buffer, transported to a central facility equipped for multiparametric flow cytometry, and subsequently thawed for staining and acquisition. OMIP-016 compared rapid 40-second thawing in a 37°C water bath with longer thawing at 37°C, demonstrating that rapid, uniform thawing reduces edge effects and sample-volume-related inconsistencies, thereby enhancing reproducibility in large cohorts. However, rapid thawing may lead to selective loss of fragile cells, producing an apparent rise in viability despite a reduction in total events. Quantitative comparisons—especially when absolute counts are required—should therefore report both absolute event numbers and relative frequencies.
5. Conclusion
The value of OMIP-016 does not stem from the addition of a single marker or minor staining adjustments. Instead, it integrates cross-species compatibility, tolerance for activation-induced downregulation of lineage markers, and a centralisation-friendly workflow enabled by post-fixation freezing. For vaccine evaluation, infection-immunity research and large-scale cohort studies, this ICS panel provides a high-fidelity, reproducible framework that improves the detection of genuine antigen-specific responses while offering clear validation guidelines for consistent implementation across study sites.
References:
[1] Guenounou S, Bosquet N, Dembek CJ, Le Grand R, Cosma A. OMIP-016: Characterization of antigen-responsive macaque and human T-cells. Cytometry A. 2013 Feb;83(2):182-4.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human, Mouse, Rat, Dog, Hamster, Monkey, etc.). We provide stable and reliable support for scientific research. For more information on abinScience flow cytometry antibodies, please click:
abinScience Flow Cytometry (FACS) Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский







