The Significance and Challenges of Humanized Mouse Models
Humanized mouse models serve as pivotal in vivo platforms for investigating human immune function and related diseases. However, their highly complex chimeric cellular environment poses unique challenges for flow cytometric analysis. OMIP-012 is specifically designed to systematically address the qualitative and quantitative characterization of cells in such models. Its core objectives are: to accurately distinguish the origin of human and murine leukocytes, achieve absolute counting of major human lymphocyte subsets, and establish a flexible analytical framework adaptable to diverse scientific inquiries.
Accurate Distinction Between Human and Murine Cell Origins
Precise differentiation of "human" versus "murine" cells in mixed cellular environments is a prerequisite for reliable immune analysis. OMIP-012 overcomes this hurdle through an innovative CD45 antibody combination, ensuring the purity of cell origins and significantly enhancing experimental credibility. This protocol enables efficient, precise phenotyping and counting of immune cells in humanized mice within a unified experimental framework.
A Customizable and Expandable Analytical Platform
Beyond resolving qualitative analysis challenges, OMIP-012 provides flexible scalability for future research via a modular design. Integrating standardized "anchor markers" and replaceable "functional modules," this panel is not only suitable for current studies but also adaptable to evolving scientific exploration needs.
Traditional flow cytometry faces numerous limitations in complex biological models (e.g., humanized mice), particularly when analyzing mixed human and murine immune cell populations. Through innovative antibody combinations and quantitative methodologies, the OMIP-012 protocol successfully resolves the challenge of distinguishing human from murine cells while delivering a stable, reproducible, and standardized framework for immune analysis. Its modular design allows researchers to flexibly adjust the panel, advancing new developments in immunology research.
1. OMIP-012 Panel
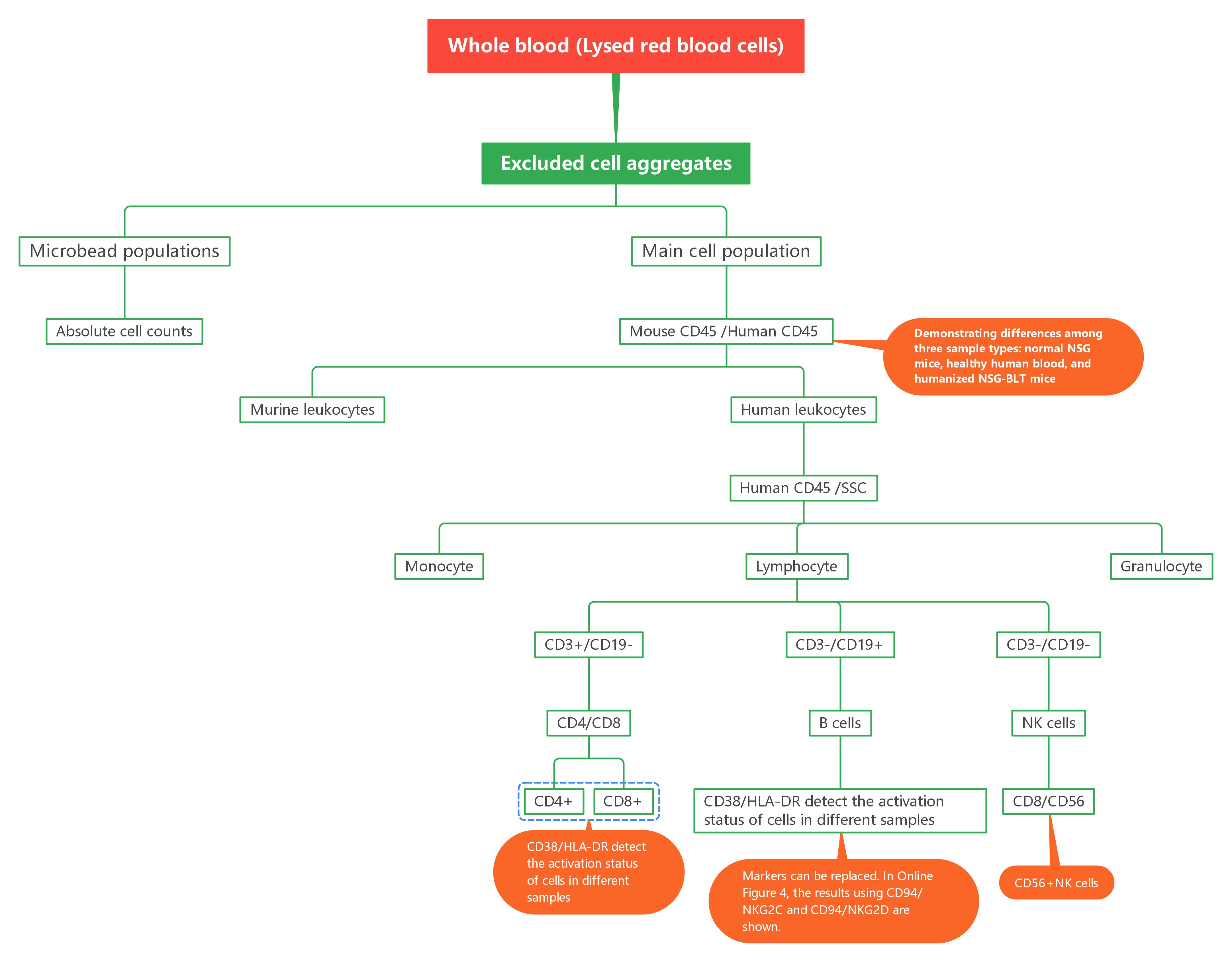
2. Gating Logic
After processing whole blood, microbead populations and the main cell population were gated to calculate absolute counts; species origin was distinguished using Mouse CD45/Human CD45, followed by analysis of cell subsets with core markers and detection of activation status via functional indicators—relevant markers can be flexibly replaced.
3. Experimental Results
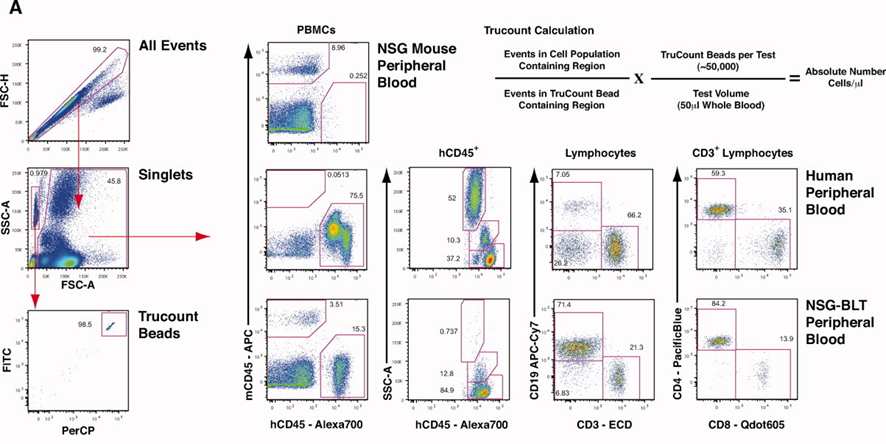
1). Lysed red blood cells, excluded cell aggregates.microbead populations and cell populations were gated separately. Absolute cell counts were calculated using bead populations; Mouse CD45/Human CD45 distinguished human and murine leukocytes (demonstrating differences among three sample types: normal NSG mice, healthy human blood, and humanized NSG-BLT mice). Human CD45, CD3, CD19, CD4, and CD8 were then used to analyze distinct cell populations in healthy human blood and humanized NSG-BLT mice.
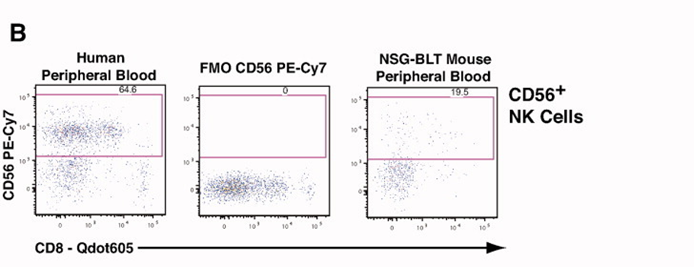
2). CD8/CD56 analysis identified NK cell populations within the CD3-/CD19+ cell subset.
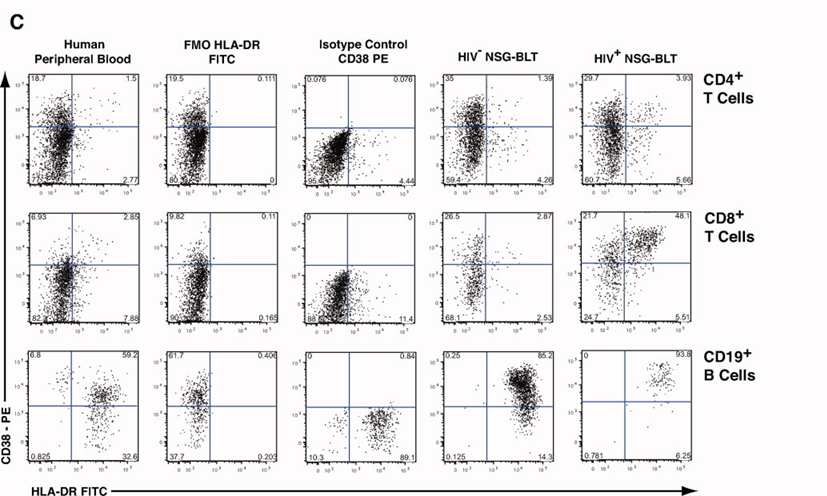
3). Cell activation status was evaluated in CD4+, CD8+, and CD3-/CD19+ cell populations across different samples.
4. Panel Interpretation
4.1 Precise Species Discrimination:
The core challenge of OMIP-012 lies in the absolute and reliable distinction between human and murine cells in mixed environments. This protocol addresses this by employing a cross-reactivity-validated pair of species-specific CD45 antibodies, establishing this as the foundational principle of panel design. The key innovation leverages the biological fact that "human and murine CD45 never co-express," boldly placing these two antibodies in highly spectrally overlapping channels. This counterintuitive approach not only avoided analytical interference but also transformed technical limitations into systematic advantages, fundamentally ensuring the purity of cell species origin within all subsequent gates and laying an unshakable foundation for the credibility of experimental data.
4.2 Quantitative Data Foundation
In humanized mouse models, the unstable hematopoietic system of the host renders relative data analysis based on percentages highly prone to distortion. OMIP-012 mandates microsphere-based absolute counting as the gold standard for this research field— the only pathway to obtaining reliable, comparable data. By directly reporting "absolute cell numbers per microliter of blood," it elevates immunophenotypic analysis from the ambiguous context of describing "relative abundance" to the objective level of "precise quantification," enabling direct and effective comparison of data across different individuals, time points, and laboratories.
4.3 Scalable Analysis Platform
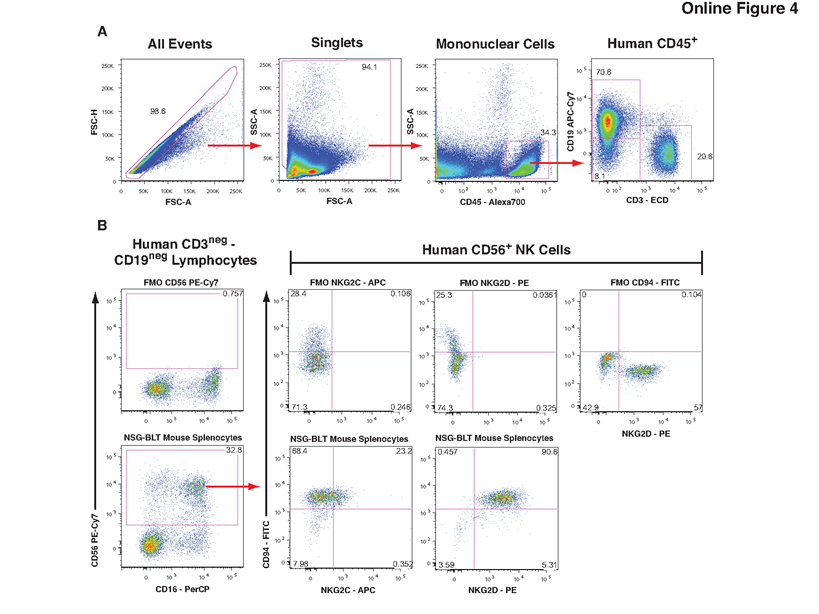
OMIP-012 transcends the limitations of "one-time" experimental protocols, constructing an expandable and customizable flow analysis platform centered on the concept of "modular design." It classifies antibodies into two categories: "anchor markers" for core immunophenotyping (e.g., CD45, CD3, CD4, CD8, CD19) and "functional modules" that can be flexibly replaced based on scientific questions. Its forward-looking design intentionally places "anchor markers" in non-conventional fluorochrome channels, thereby reserving mainstream channels such as PE, FITC, and APC as future "functional interfaces." As shown in Supplementary Figure 4: without modifying the core identification framework, a new NK cell phenotyping protocol was rapidly derived from the basic immunophenotyping panel by replacing three "functional modules"—HLA-DR (FITC), CD38 (PE), and Mouse CD45 (APC)—with CD94 (FITC), NKG2D (PE), and NKG2C (APC), plus the addition of CD16 (PerCP). This "plug-and-play" design significantly enhances research efficiency and system sustainability, allowing a single core protocol to support continuously evolving scientific exploration.
5. Applications
Humanized mouse immune monitoring Tumor immunology T cell response Autoimmune disorder immune analysis Clinical immune monitoring cell counting Vaccine-induced T/B cell analysis
6. Conclusion
Through three interlocking design elements, the OMIP-012 protocol provides robust methodological support for immune analysis in humanized mice. Its success lies in transforming a complex biological problem into an executable, reproducible, and expandable analytical workflow via clear flow cytometric logic. Beyond offering an antibody combination, it establishes a novel paradigm for analyzing chimeric models—prioritizing the absolute reliability of cell identity (species origin), followed by precise quantification of cell numbers, and ultimately facilitating flexible exploration of cell function. It is this systematic design philosophy that has made this protocol, published over a decade ago, remain a classic reference and source of inspiration for constructing similar research experimental schemes to this day.
Get OMIP-012 Compatible Flow Cytometry Antibodies
abinScience provides validated flow cytometry antibodies covering key targets in this panel, supporting your immunology research
References
[1] Long BR, Stoddart CA. OMIP-012: Phenotypic and numeric determination of human leukocyte reconstitution in humanized mice. Cytometry A. 2012 Aug;81(8):646-8.

About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human/Mouse/Rat/Dog/Hamster/Monkey, etc.). We provide stable and reliable support for scientific research.
Explore abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский