Clinical Value of Circulating Endothelial Cells (CECs)
Circulating Endothelial Cells (CECs) and bone marrow-derived Endothelial Progenitor Cells (EPCs) are extremely rare in peripheral blood, yet they possess high potential diagnostic value in cardiovascular diseases and angiogenesis-related disorders (such as cancer, ischemic diseases, and diabetes). Due to the weak expression of their cell surface markers, traditional marker detection methods often fail to reliably identify them, which increases the complexity of analysis. OMIP-011 proposes an innovative 8-color flow cytometry panel, providing a powerful tool for evaluating CECs in peripheral blood.
Addressing the Challenge of Weak Marker Expression
A major hurdle in CEC analysis lies in the weak and indistinct expression of their surface markers, which often results in inconsistent identification and unreliable detection outcomes—especially in conventional single-marker detection assays that lack sufficient specificity. OMIP-011 effectively overcomes this critical limitation by strategically combining key endothelial markers, including CD31, CD34, and CD146, thereby constructing a highly specific and sensitive assay for accurate CEC identification and initial characterization.
Precise CEC Quantification and Activation Status Analysis
To ensure the reliable identification and precise quantification of rare CECs in peripheral blood, OMIP-011 incorporates a dual-platform quantification method integrated with TruCount beads, which significantly enhances detection sensitivity for these scarce cells. Additionally, the inclusion of CD106 and CD117 enables in-depth analysis of CEC activation status—a critical factor, as the functional characteristics of activated CECs are closely associated with disease progression and prognosis, particularly in oncology and angiogenesis-related research fields.
Circulating Endothelial Cells (CECs) and bone marrow-derived Endothelial Progenitor Cells (EPCs) are extremely rare in peripheral blood but exhibit high potential diagnostic value in cardiovascular conditions and/or various angiogenesis-related diseases (e.g., cancer, ischemia, and diabetes). Analyzing CECs is challenging due to the fact that they are typically identified through a combination of multiple cell surface antigens, which display low, indistinct expression or continuous distribution. OMIP-011 presents an optimized 8-color panel for assessing CECs in peripheral blood.
1. OMIP-011 Panel
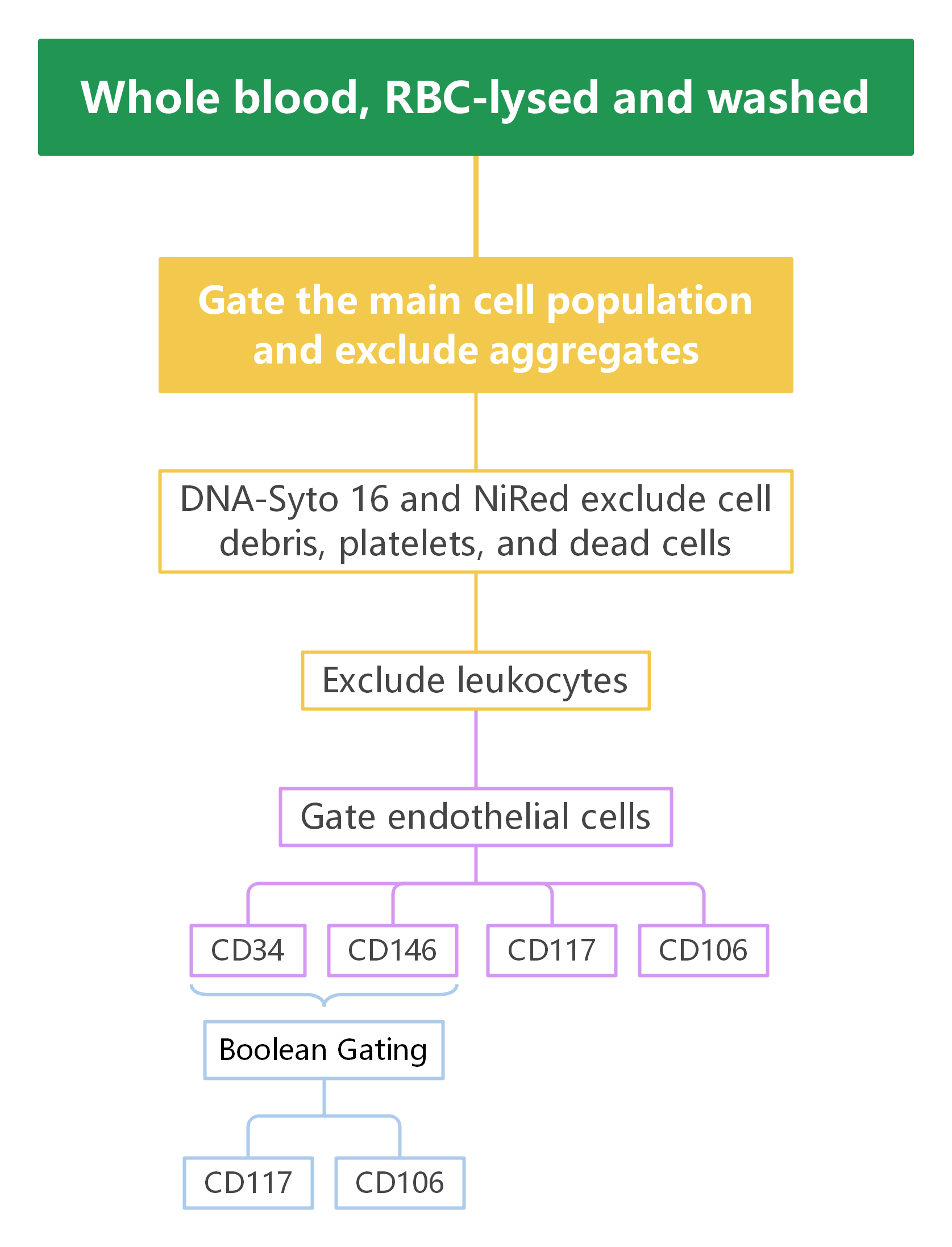
2. Gate Logic
1
Exclusion of Non-Target Cells and Debris
FSC-A/SSC-A was used to exclude cell debris, while FSC-A/FSC-H was used to exclude cell aggregates, ensuring the analysis of single-cell populations.
2
Dead Cell Exclusion
The DNA dye Syto 16 and NiRed (a live/dead marker) were employed to exclude dead cells, microparticles, and platelets, guaranteeing data accuracy.
3
Endothelial Cell Population Selection
CD31 was used to gate endothelial cell populations, which were further characterized using CD34, CD146, CD106, and CD117 to analyze endothelial cell subsets.
4
B Cell and T Cell Population Analysis
CD45 labeling excluded leukocytes, and additional markers such as CD3, CD4, CD8, and CD19 were used to further sort T cell, B cell, and NK cell subsets.
5
CEC Activation Analysis
CD106 and CD117 were combined to analyze the activation status of CECs, facilitating investigations into their roles in different diseases.
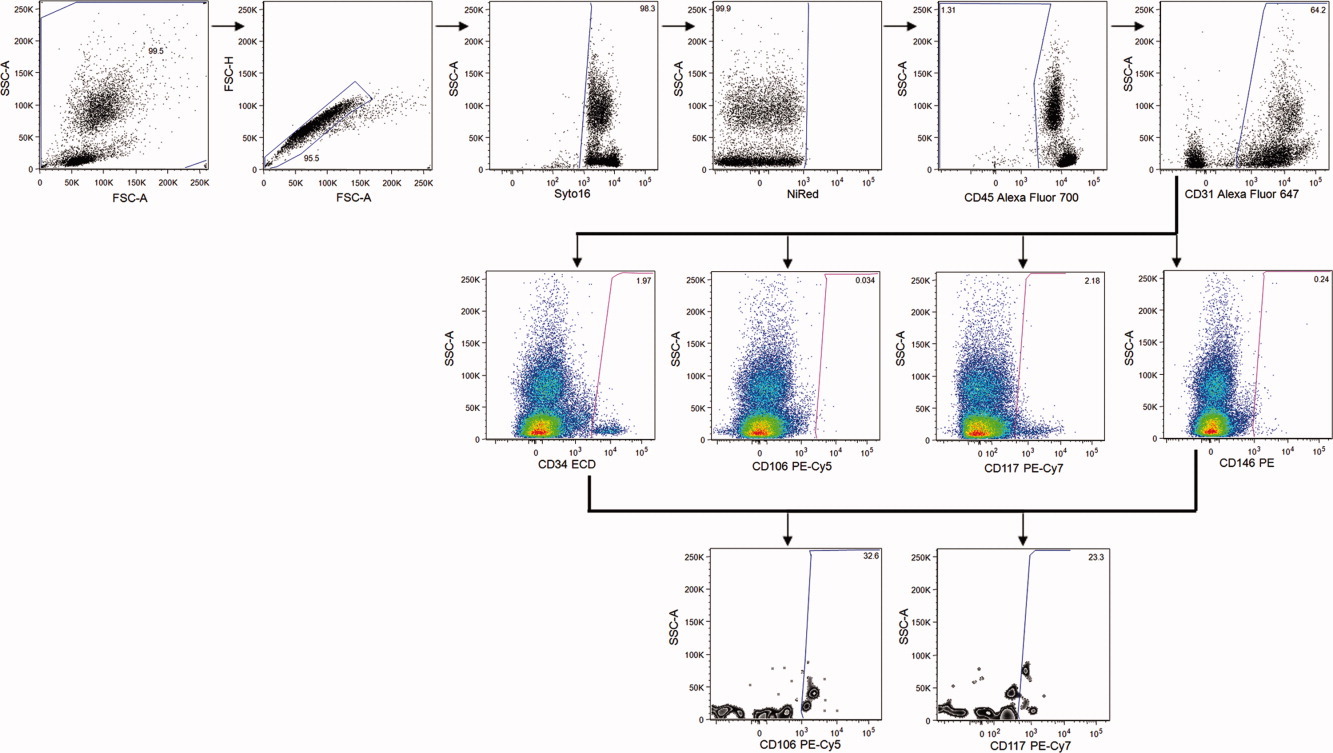
3. Experimental Results
- ① FSC-A/SSC-A was used to exclude abnormal artifacts in the upper left and lower right corners, while FSC-A/FSC-H was used to exclude cell aggregates.
- ② Syto 16+ (DNA dye) and NiRed- (live/dead marker) were used to exclude dead cells, microparticles, and platelets; CD45- was used to exclude leukocytes.
- ③ CD31 gated the endothelial cell population, which was further analyzed for distribution using CD34, CD146, CD106, and CD117 in combination with SSC.
- ④ CD34+CD146+ cell populations were analyzed via Boolean Gating, with their distribution displayed in CD106/SSC and CD117/SSC plots.
4. Panel Interpretation
4.1 Optimized Endothelial Marker Combination
A key challenge in CEC analysis is the weak expression of their markers, which often leads to unreliable identification in traditional single-marker detection. This panel combines CD31, CD34, and CD146 to more accurately distinguish CECs, overcoming the problem of weak marker expression.
4.2 Dual-Platform Quantification Method
To ensure the accuracy of CEC enumeration, the study adopts a dual-platform quantification method combined with TruCount beads. This method not only improves the detection rate of CECs but also enhances the comparability between samples—especially when CECs are extremely rare in blood samples.
4.3 Analysis of Activated CEC Subpopulations
The inclusion of CD106 and CD117 as activation markers allows the panel to not only identify CECs but also further analyze their activation status. This is crucial for understanding the role of CECs in different diseases, particularly in cancer and angiogenesis-related research, where activated CECs may be closely associated with disease progression and prognosis.
5. Applications
Detection and immune monitoring of CECs Diagnosis of angiogenesis-related diseases Immune monitoring of cardiovascular diseases Research on CEC activation status and tumor progression Quantitative CEC analysis in clinical immune testing
6. Conclusion
By carefully selecting endothelial markers and integrating a dual-platform approach, OMIP-011 provides an effective tool for the accurate analysis and quantification of CECs in peripheral blood. The proposal of this panel not only addresses issues such as weak marker expression and CEC rarity in traditional methods but also supports diagnosis and monitoring in fields related to angiogenesis and cardiovascular diseases.
Get OMIP-011 Compatible Flow Cytometry Antibodies
abinScience provides validated Flow Cytometry Antibodies covering key targets in this panel, supporting your CECs analysis and research
References
[1] Lachmann R, Lanuti P, Miscia S. OMIP-011: Characterization of circulating endothelial cells (CECs) in peripheral blood. Cytometry A. 2012 Jul;81(7):549-51.

About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human/Mouse/Rat/Dog/Hamster/Monkey, etc.). We provide stable and reliable support for scientific research.
Explore abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский