The accuracy of flow cytometry results depends not only on antibody selection and instrument settings but also critically on the state of the sample itself. Dead cells, due to compromised membrane integrity, often lead to non-specific antibody binding. Additionally, they can exhibit enhanced autofluorescence, generating false-positive signals and blurring the boundaries of cell populations. This interference is particularly pronounced when analyzing rare cell populations or low-abundance antigens.
1. Impact of Viability Staining on Flow Cytometry Results
1.1 Non-specific Antibody Binding
When a cell loses membrane integrity, intracellular components with high charge density and non-specific antigenic sites are exposed. These can bind fluorescently labeled antibodies non-specifically, producing false-positive signals.
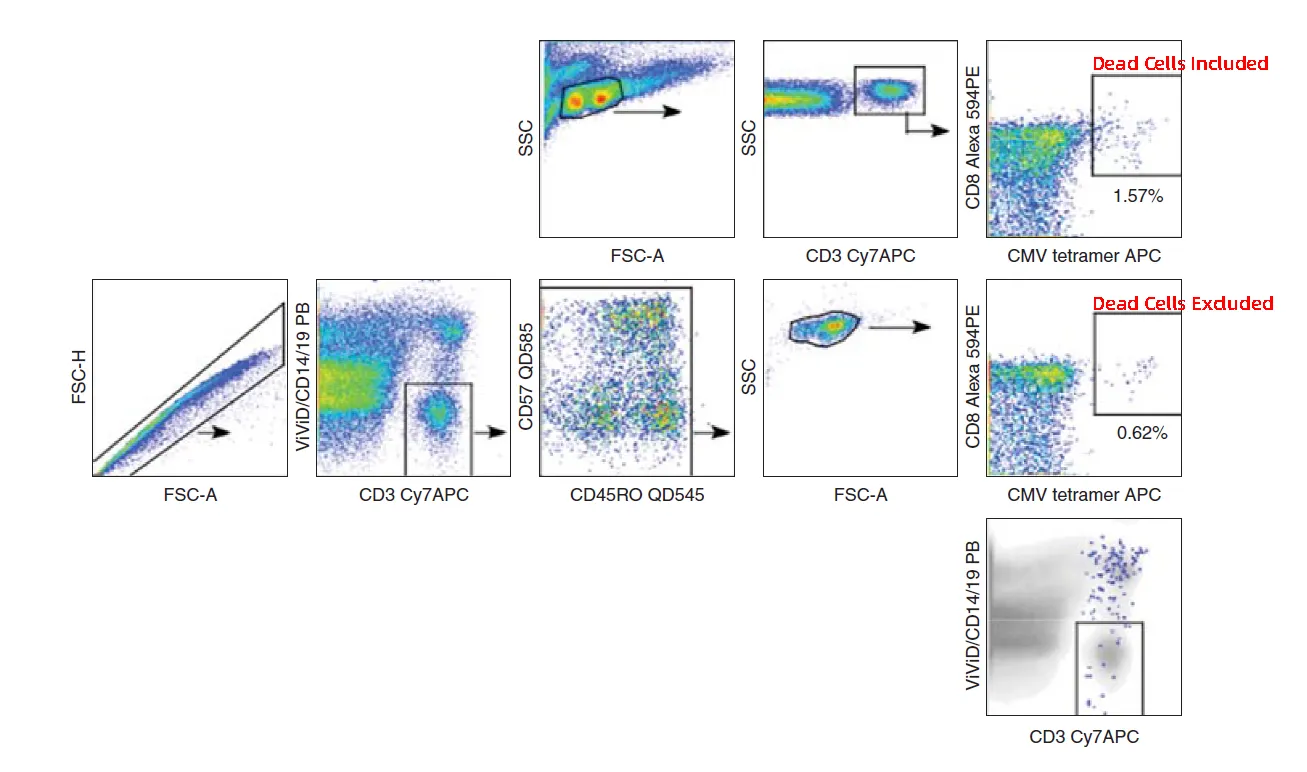
Figure 1. False-positive signals caused by dead cells
1.2 Increased Autofluorescence
During cell death, organelles such as mitochondria and lysosomes release metabolic byproducts (e.g., flavins, lipofuscin) that can significantly increase autofluorescence. This autofluorescence is broad-spectrum and may spill into multiple detection channels, raising background signals. In some cases, it manifests as diagonal spreading in dot plots—a key feature for identifying dead cell interference.
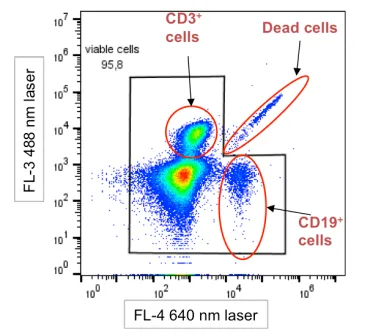
Figure 2. Schematic of diagonal distribution of dead cells
1.3 Blurred Population Boundaries
The combination of non-specific binding and increased autofluorescence can result in diagonal tails or poorly defined boundaries of target populations in scatter plots. This compromises gating accuracy and reproducibility. Rare populations or weakly expressed antigens are particularly susceptible, potentially leading to undercounting or misidentification.
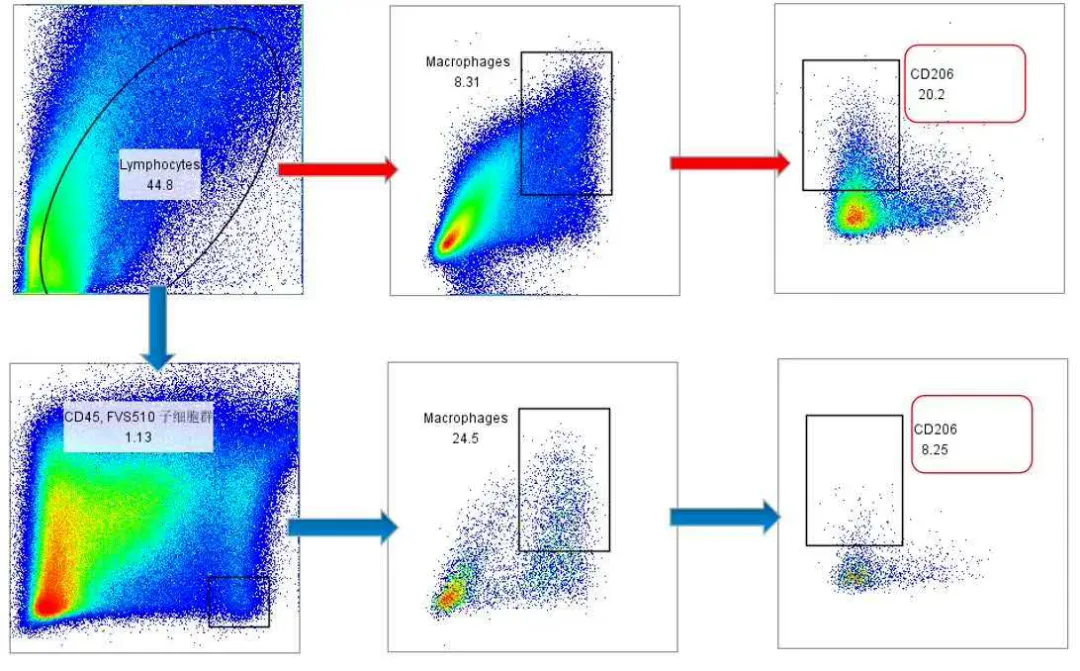
Figure 3. Without viability staining and CD45 gating, CD11b⁺F4/80⁺ macrophage populations appear poorly resolved, and CD206 signals are abnormally elevated
2. Types of Viability Dyes and Selection Guidelines
Common viability dyes can be categorized into two types based on experimental needs:
2.1 Nucleic Acid Dyes (e.g., PI, 7-AAD)
Nucleic acid dyes distinguish live and dead cells based on membrane integrity. Live cells exclude the dye, while dead cells with compromised membranes allow dye entry and DNA binding, generating fluorescence. These dyes are suitable for unfixed samples. After fixation, membrane permeability changes, and dyes may enter live cells, preventing accurate discrimination.
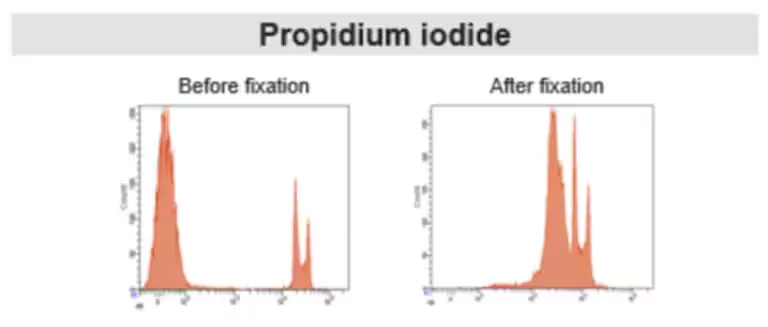
Figure 4. Comparison of PI staining before and after fixation
2.2 Amine-Reactive Dyes (Fixable Viability Dyes)
Amine-reactive dyes covalently bind cellular proteins. Dead cells allow the dye to penetrate and label abundant intracellular proteins, producing fluorescence much higher than that of live cells labeled only on the surface. These dyes tolerate subsequent fixation and permeabilization, making them suitable for complex experiments that include intracellular staining.
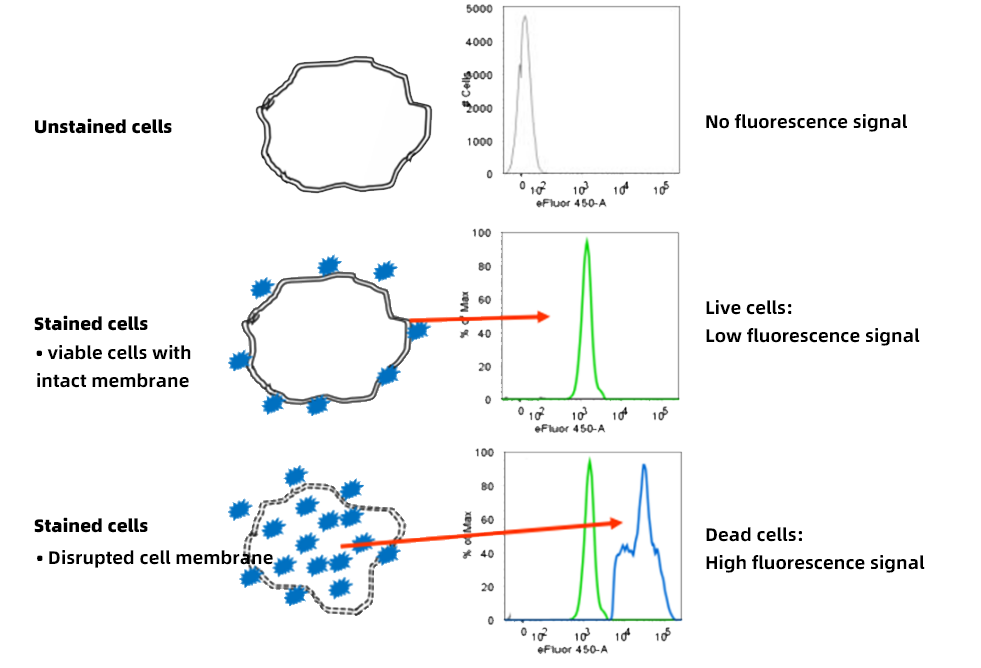
Figure 5. Principle of live/dead discrimination using amine-reactive dyes
When designing experiments, it is essential to determine whether fixation or permeabilization is required, guiding the appropriate choice of dye.
| Feature |
Nucleic Acid Dyes |
Amine-Reactive Dyes |
| Staining Principle |
Membrane-impermeable nucleic acid dyes enter cells with compromised membranes and bind dsDNA/RNA; intact cells are excluded. |
Membrane-impermeable amine-reactive dyes covalently bind primary amines on cellular proteins. Weak signal from surface labeling; strong signal when dye penetrates dead cells. |
| Staining Timing |
After surface staining, before acquisition |
Before surface staining |
| Post-fixation Staining |
No |
Yes |
| Fluorescence Options |
Limited, channel selection constrained |
Multiple, flexible for panel design |
| Common Dyes |
PI, 7-AAD, DAPI |
Invitrogen™ LIVE/DEAD Fixable Dyes, eBioscience™ Fixable Viability Dyes |
| Other Considerations |
Broad emission spectrum can interfere with other channels; PI is sticky and unsuitable for fixed-cell assays |
Multiple fluorophores available, flexible panel integration; reduces cell aggregation and flow cytometer clogs |
3. Operational Considerations for Viability Staining
3.1 Titration Optimization
Amine-reactive dyes require titration to determine the optimal working concentration. The goal is to maximize separation between live and dead cells (stain index), not simply to increase fluorescence intensity. Excessive dye can raise background in live cells; insufficient dye fails to label dead cells adequately. Each new dye batch should be titrated to ensure consistency.
3.2 Buffer Selection
Since amine-reactive dyes bind proteins, high protein concentrations (e.g., serum or BSA) can compete for dye binding, weakening positive signals. Staining should be performed in protein-free buffers (e.g., PBS), followed by washing steps with protein-containing buffers to block non-specific binding.
3.3 Spectral Panel Design
When designing multiparameter panels, consider potential spreading errors caused by the viability dye channel. Thoughtful dye combinations help minimize interference with detection of weakly expressed antigens.
Conclusion
Viability staining is a critical quality control step in flow cytometry. Proper dye selection, optimized titration, and controlled staining conditions effectively exclude dead cell interference, improving data accuracy and reproducibility while providing a reliable foundation for scientific conclusions.
About Us
As a strategic venture of AtaGenix (established 2011), abinScience specializes in the development and production of high-quality life science reagents, and is committed to providing researchers with high-performance, highly reliable Flow Cytometry Antibodies, we offer:
1. Stringently validated flow cytometry antibodies including CD3, CD4, CD8 ensuring high sensitivity and minimal background noise;
2. A comprehensive portfolio of immunology research tools for multiple species, including Human, Mouse, Rat, Dog, Hamster;
3. Expert technical support and panel design consultation to help you accurately resolve complex immune cell populations.
For more information on abinScience flow cytometry antibodies, please visit:
abinScience Flow Cytometry Antibodies
References
[1] Perfetto SP, Chattopadhyay PK, Lamoreaux L, Nguyen R, Ambrozak D, Koup RA, Roederer M. Amine-reactive dyes for dead cell discrimination in fixed samples. Curr Protoc Cytom. 2010 Jul;Chapter 9:Unit 9.34. doi: 10.1002/0471142956.cy0934s53. PMID: 20578108; PMCID: PMC2915540.
[2] Perfetto SP, Chattopadhyay PK, Lamoreaux L, Nguyen R, Ambrozak D, Koup RA, Roederer M. Amine reactive dyes: an effective tool to discriminate live and dead cells in polychromatic flow cytometry. J Immunol Methods. 2006 Jun 30;313(1-2):199-208. doi: 10.1016/j.jim.2006.04.007. Epub 2006 May 19. PMID: 16756987.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский








