Candida auris (sometimes listed under the updated name Candidozyma auris) has become the most “operationally difficult” fungal pathogen in modern healthcare settings. The comprehensive review by Chowdhary and colleagues (Microbiology and Molecular Biology Reviews, 2025) frames the problem clearly: it is not only about drug resistance—C. auris couples resistance with traits that support persistent colonization, healthcare transmission, and diagnostic pitfalls. In other words, it behaves like a yeast that is unusually well-adapted to the hospital ecosystem rather than a sporadic opportunist.
That “facility-adapted” profile matters because it changes what teams need to measure. For many laboratories, the most useful questions are practical ones: Why does colonization last so long on skin? Why does it survive on surfaces? Why can it slip past routine identification? Why do treatment options narrow so quickly? This page summarizes the parts that are most actionable for research workflows: pathogenesis, protein targets that explain resistance, cell wall biology (especially β-1,3-glucan remodeling), and the virulence traits you can translate into assays.
2025 U.S. surveillance signal: Weekly table reporting in the National Notifiable Diseases Surveillance System lists 7,046 cumulative year-to-date confirmed clinical cases for Candida auris, clinical as of Week 51 (week ending December 20, 2025). Case counts are marked provisional in the weekly tables and may be updated as reporting is finalized.
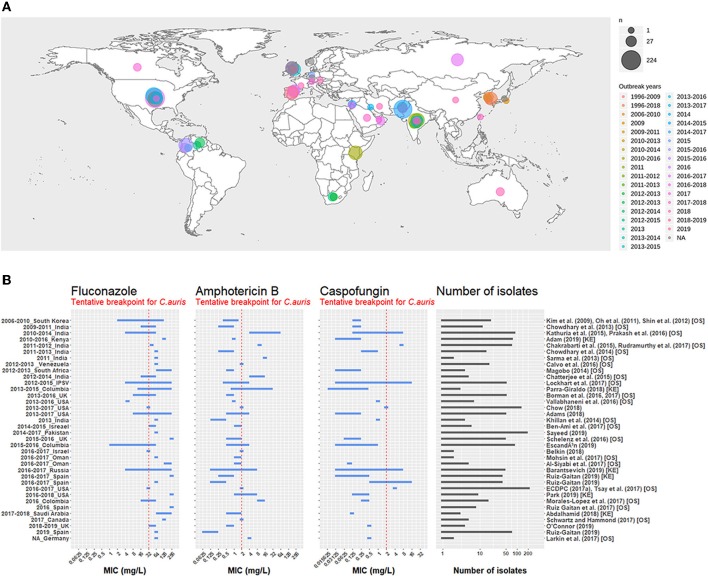
Fig. 1. Global distribution of C. auris and reported antifungal susceptibility ranges (MIC). Reproduced from Chaabane et al., Frontiers in Microbiology (2019), CC BY. [3]
Molecular Pathogenesis and Resistance Mechanisms
A helpful way to understand C. auris is to start with where it “likes to live.” Unlike many yeasts that are discussed mainly in the context of mucosal or gastrointestinal niches, C. auris is repeatedly linked to durable skin colonization (for example, axilla and groin), which becomes the engine for transmission. Colonization itself is often silent, but it creates a steady source of shedding—hands, linens, shared devices, and high-touch surfaces then become the bridge to new hosts.
From a control standpoint, the difficult part is that the organism can remain detectable even when the environment is not obviously “wet” or nutrient-rich. This is why dry-surface persistence and biofilm-like growth have attracted attention: they fit how real wards operate, and they help explain why routine cleaning can fail if the disinfectant choice, contact time, or surface context is mismatched. Ware and colleagues (APMIS, 2025) specifically describe dry surface biofilm formation supporting persistence and tolerance under sodium hypochlorite challenge.
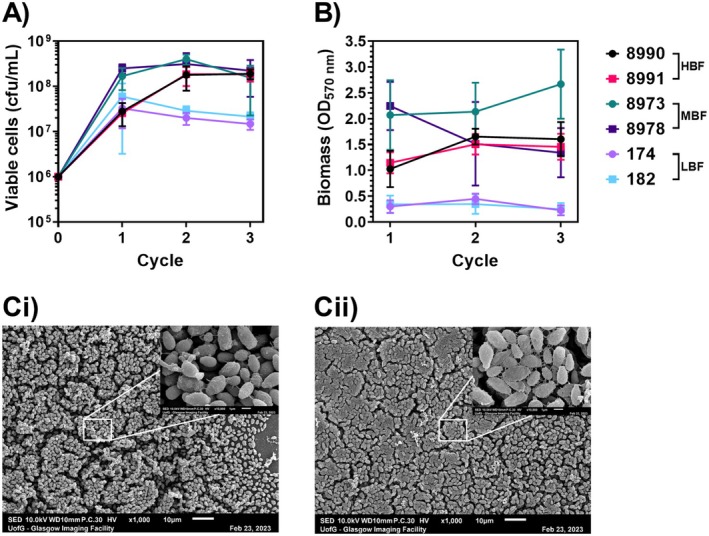
Fig. 2. Dry surface biofilm model and hypochlorite tolerance phenotypes in C. auris. Reproduced from Ware et al., APMIS (2025), CC BY 4.0. [4]
When infections do occur, they usually arise in the familiar high-risk context (for example, invasive devices, prolonged hospitalization, immune compromise). The key research takeaway is that C. auris does not rely on a single dramatic toxin. Its advantage is more cumulative: colonization + persistence + stress tolerance + resistance mechanisms that are often multi-layered rather than single-gene.
Key Protein Targets: Functions and Research Applications
In practice, “protein structure” discussions around C. auris most often mean one thing: how target proteins and their substitutions reshape drug susceptibility. Two anchors appear across clinical and experimental literature—ERG11 (azole target in ergosterol biosynthesis) and FKS1 (echinocandin target in β-1,3-glucan synthesis). Even when you do not run structural assays directly, thinking in a structure-informed way helps you connect sequencing and minimum inhibitory concentration shifts to downstream phenotypes.
| Target | Function & Characteristics | Research Utility |
|---|---|---|
| ERG11 | Lanosterol 14α-demethylase (azole target) in ergosterol biosynthesis. Reduced azole susceptibility is often discussed alongside broader programs (for example, efflux and stress responses), not only one substitution. | Azole mechanism work; genotype–minimum inhibitory concentration interpretation; pathway validation experiments. |
| FKS1 | Catalytic subunit of β-1,3-glucan synthase (echinocandin target). Hot-spot mutations (for example, S639F in some reports) can elevate echinocandin minimum inhibitory concentrations and coincide with measurable wall remodeling. | Echinocandin resistance mapping; link “target change” to wall remodeling phenotypes. |
| β-1,3-Glucan (exposure/accessibility) | Core cell wall scaffold. Exposure can shift with stress, biofilm growth, and remodeling in resistant backgrounds. This makes it useful as a “state readout,” not merely a presence marker. | Assay readout for remodeling and biofilms; supports microscopy, flow cytometry, and comparative phenotype profiling. |
| Adhesins / Biofilm Matrix | Surface attachment and persistence traits that support colonization and environmental survival. Species-specific adhesins have been reported as determinants of surface colonization and virulence. | Adhesion screens; dry-surface persistence assays; intervention testing on realistic substrates. |
If you are building a “minimal but credible” experimental story, a good pattern is to pair one upstream driver (drug pressure or target genotype) with one downstream phenotype. β-1,3-glucan exposure is often a strong downstream choice because it sits at the intersection of growth, stress adaptation, biofilm biology, and innate immune visibility.
About abinScience
abinScience develops research-grade recombinant proteins and antibodies for infectious disease and microbial pathogenesis research. For Candida auris projects, we focus on tools that map to high-intent phenotypes researchers actually measure: cell wall remodeling, biofilm-associated behavior, and resistance-linked biology.
abinScience C. auris Research Reagents
If you currently have one core reagent, the key is to make the page solve a specific lab problem rather than “cover all knowledge.” β-1,3-glucan is especially useful here because it behaves like a readout of wall state: it can shift with biofilm growth, stress conditions, and resistance-associated remodeling. That makes it a practical handle for experiments that aim to explain persistence or treatment pressure responses. For research use only | Bulk available.
| Type | Catalog # | Product Name |
|---|---|---|
| Antibody | JN903016 | Research Grade Anti-Candida auris β-1, 3 glucans Antibody (2G8) |
Where clone 2G8 is most useful (simple starting points)
Contact: support@abinscience.com | Inquiries for custom reagents welcome.
References
+86-027-65523339
中国武漢市深敦寺路666号C棟、武漢、430206

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский