2026 Latest Nipah Virus (Nipah virus) Research Review: Outbreak Updates, Molecular Structure, Pathogenesis, Development Progress, and abinScience Tool Recommendations
In January 2026, a new outbreak of Nipah virus (Nipah virus, NiV) emerged in West Bengal, India. As of January 26, five confirmed cases have been reported, including several healthcare workers involved in patient care. This confirms the risk of nosocomial human-to-human transmission, and nearly 100 close contacts have been placed under urgent home quarantine. The news has raised global alarm. Neighboring countries such as Nepal and Thailand have swiftly strengthened border screening measures, with international airports in Phuket and Chiang Mai implementing thermal imaging temperature checks and travel history verification. The World Health Organization (WHO) has once again issued a public health alert, highlighting the virus’s 40%–75% fatality rate and its potential for a large-scale pandemic. Nipah virus is more insidious than Ebola, with an extremely high mortality rate, long incubation period, and zoonotic transmission. First identified in Malaysia in 1998, it causes annual winter outbreaks in Bangladesh and has been recorded multiple times in India—this is the tenth outbreak in West Bengal since 2018.
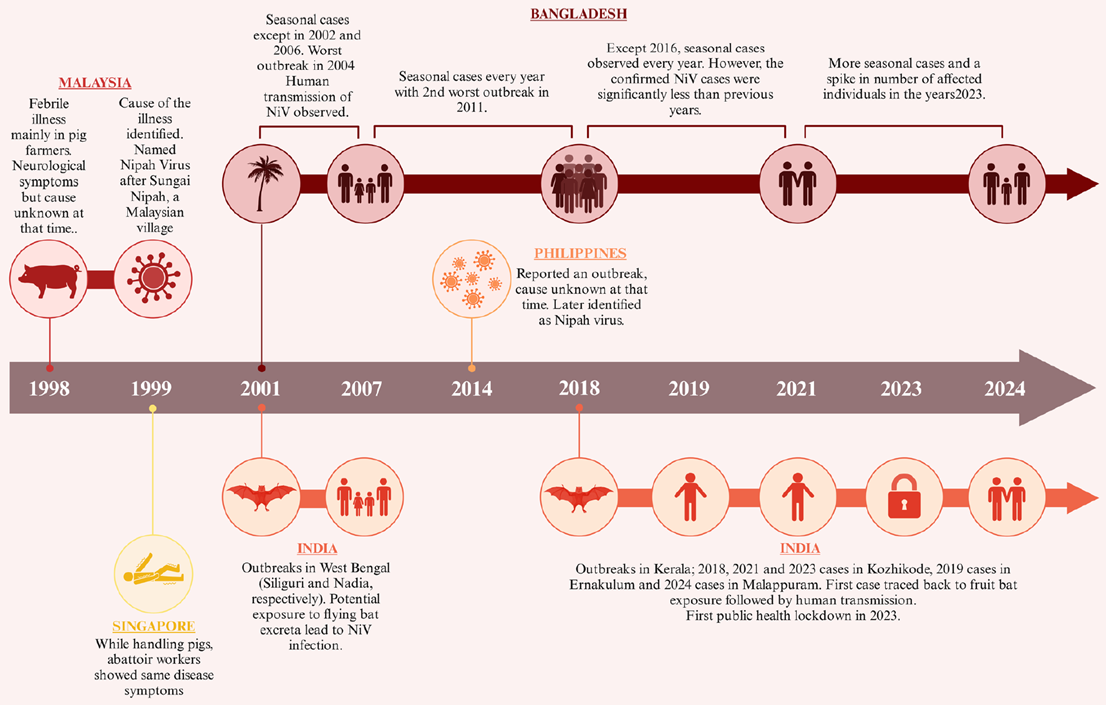
Figure 1. Timeline of Nipah virus outbreaks from 1998 to 2024
Viral Structure
Nipah virus belongs to the Paramyxoviridae family, genus Henipavirus and is closely related to Hendra virus (HeV). It has a negative-sense single-stranded RNA genome approximately 18.2 kb in length, encoding six structural proteins and three non-structural proteins. The virion consists of an envelope, nucleocapsid, and various functional proteins. The core structural proteins include nucleoprotein (N), phosphoprotein (P), matrix protein (M), fusion protein (F), glycoprotein (G), and large polymerase (L). Each plays a critical role in viral replication, transmission, and pathogenesis, contributing to its high pathogenicity and transmissibility.
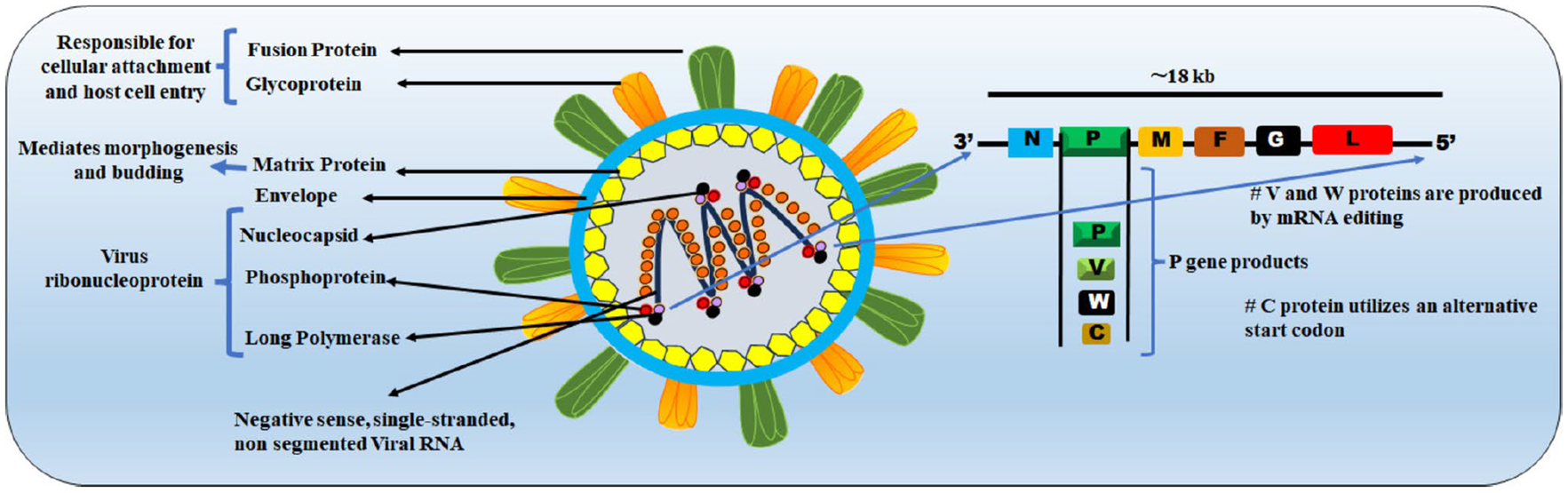
Figure 2. Schematic representation of Nipah virus structure and proteins
| Protein |
Full Name |
Function |
| Nucleoprotein N |
Nucleocapsid protein |
Encapsulates the viral RNA genome to form the nucleocapsid complex, protecting genetic material from host enzymes and directly participating in viral replication and transcription—an essential component of the viral life cycle. |
| Phosphoprotein P |
Phosphoprotein |
Acts as a cofactor for the large polymerase (L), stabilizing it. Through RNA editing, it generates V and W proteins that help the virus evade the host immune response. |
| Matrix protein M |
Matrix protein |
Links the nucleocapsid to envelope glycoproteins, providing structural support for virion assembly, mediating budding from host cells, and participating in viral morphogenesis via nucleocytoplasmic shuttling. |
| Fusion protein F |
Fusion protein |
Initially synthesized as an inactive F₀ precursor, it is cleaved into F₁ and F₂ subunits. It mediates fusion between the viral envelope and host cell membrane, a critical step for viral entry; conformational changes directly affect infection efficiency. |
| Glycoprotein G |
Glycoprotein |
Forms homotetramers that specifically bind host Ephrin-B2/B3 receptors, determining host range and tissue tropism. It serves as the “key” for viral entry and is the primary target for neutralizing antibodies. |
| Large polymerase L |
Large polymerase |
The core enzyme for replication and transcription (~250 kDa), containing RNA-dependent RNA polymerase and methyltransferase domains. It catalyzes mRNA synthesis and genome replication, essential for viral proliferation. |
Pathogenesis
The high pathogenicity of Nipah virus results from its unique infection mechanisms and immune evasion strategies, which together cause severe pathological damage:
Core Pathogenic Mechanisms of Nipah Virus
- Receptor Binding and Entry: The viral G protein binds host Ephrin-B2/B3 receptors, triggering a conformational change in the F protein from a pre-fusion to a fusion-active state, enabling fusion of the viral envelope with the host cell membrane and release of the nucleocapsid into the cytoplasm.
- Immune Evasion: Once inside the cell, P protein and its derivatives (C, W, V) block interferon signaling pathways such as JAK-STAT, suppressing innate immunity and allowing rapid viral replication. N-glycans on G protein act as decoys, shielding the envelope from antibody neutralization.
- Cell-to-Cell Spread and Tissue Damage: The F protein induces fusion of infected cells with adjacent uninfected cells, forming multinucleated syncytia and enabling direct spread while evading immune clearance. The virus has broad tissue tropism, crossing the blood-brain barrier to cause acute encephalitis and infecting lung endothelial and parenchymal cells, leading to acute respiratory distress. Severe cases can progress to coma and death within 24–48 hours.
- Long-Term Sequelae: Approximately 20% of survivors experience persistent neurological issues, such as personality changes and epilepsy, due to lasting central nervous system damage.
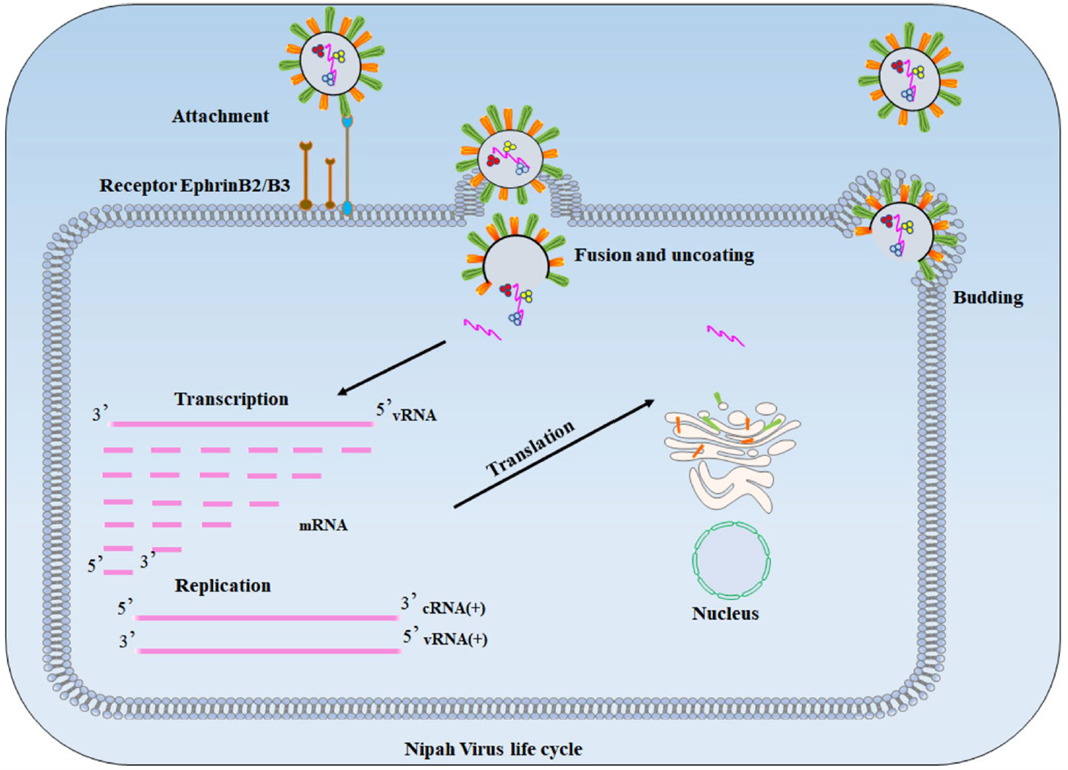
Figure 3. Nipah virus life cycle
Latest Research Progress
No licensed vaccines or specific treatments exist for Nipah virus, but significant breakthroughs have been achieved in recent years:
Vaccine Development
- In January 2024, the University of Oxford initiated the first human trial of the ChAdOx1 NipahB vaccine to evaluate safety and immunogenicity.
- Phylex Biosciences’ mRNA nanoparticle vaccine showed strong neutralizing antibody responses with a single dose in mice.
- A G protein head domain nanoparticle vaccine developed by Wuhan Institute of Virology provides broad protection against three pathogenic henipaviruses.
- The U.S. PHV02 vaccine has completed Phase I trials with confirmed safety and immunogenicity and is advancing to Phase II.
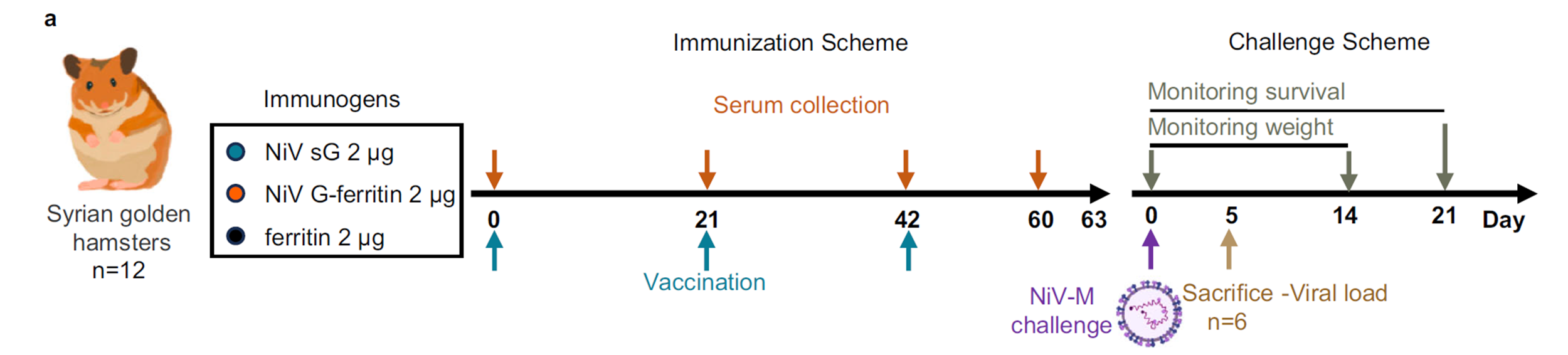
Figure 4. NiV G-ferritin protects Syrian golden hamsters from lethal NiV Malaysia strain infection
Antibody and Drug Research
- In July 2025, a bispecific antibody D590-m102.4 developed by the University of Queensland achieved 100% prophylactic protection in hamsters.
- The human monoclonal antibody m102.4, which blocks G protein-receptor binding, has completed Phase I trials and is approved for emergency use in India.
- Nucleoside analogs such as remdesivir demonstrate antiviral activity in vitro and in animal models, reducing mortality.
Diagnostic Technologies
- Diagnosis currently relies on RT-PCR for viral nucleic acids and ELISA for specific antibodies.
- A Chinese team’s CRISPR/Cas12a assay rapidly detects the N gene with high sensitivity and is suitable for field use.
- The Truenat™ Nipah PoC kit, validated with 97% sensitivity and 100% specificity, has been deployed in Indian outbreaks.
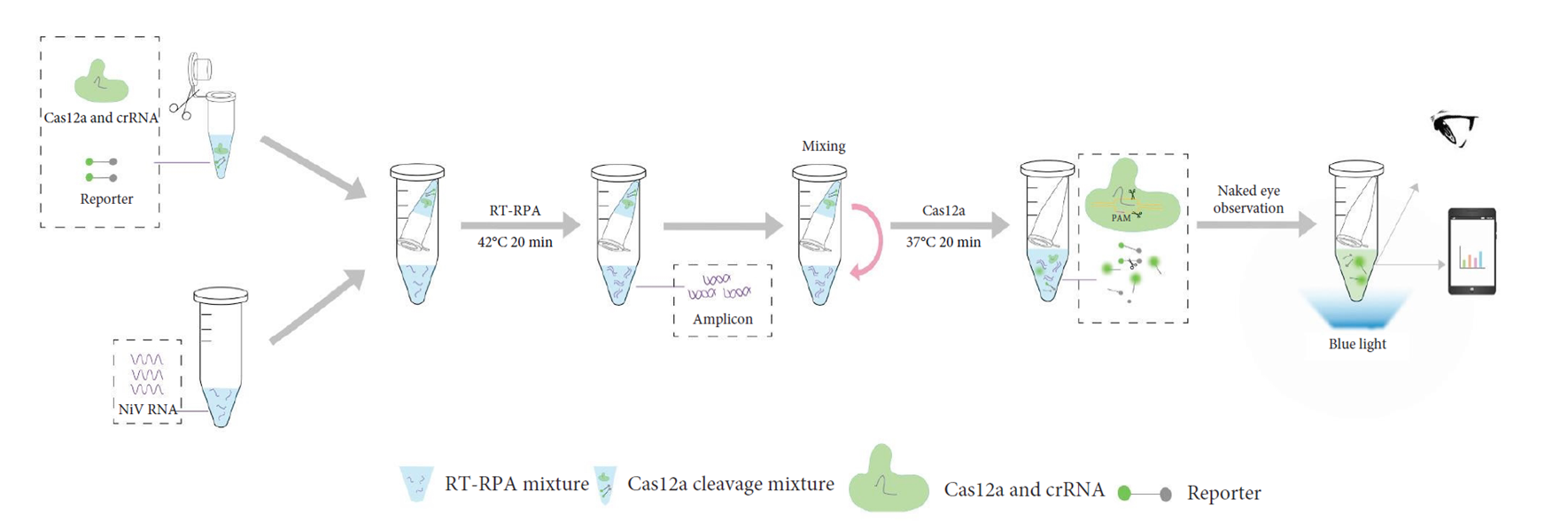
Figure 5. Schematic of a one-step visual RT-RPA-CRISPR/Cas12 assay for NiV detection
Epidemiological Features and Latest Global Outbreak Updates
Nipah virus outbreaks are concentrated in India and Bangladesh, primarily linked to consumption of date palm sap contaminated by fruit bats. Transmission also occurs directly from bats or via intermediate hosts such as pigs. The current West Bengal outbreak highlights diverse transmission routes, including foodborne, droplet, saliva, and human-to-human spread, with nosocomial infections among healthcare workers underscoring the risk.
The incubation period ranges from 4 to 14 days (up to 45 days), and asymptomatic carriers can transmit the virus, complicating control efforts. Strains vary in virulence; Indian and Bangladeshi strains (NiV-B) have fatality rates of 70%–100% and high human-to-human transmission (~75%), compared to the Malaysian strain (NiV-M, ~8%).
In 2022, a collaborative study published in the New England Journal of Medicine (NEJM) reported a new henipavirus—Langya virus (LayV)—isolated from febrile patients with animal contact in eastern China. The genome is similar to other henipaviruses. A total of 35 acute cases were identified (26 mono-infections), with no fatalities. No secondary transmission was observed among 15 close contacts of 9 patients, suggesting low human-to-human risk. Shrews are suspected as the natural reservoir.
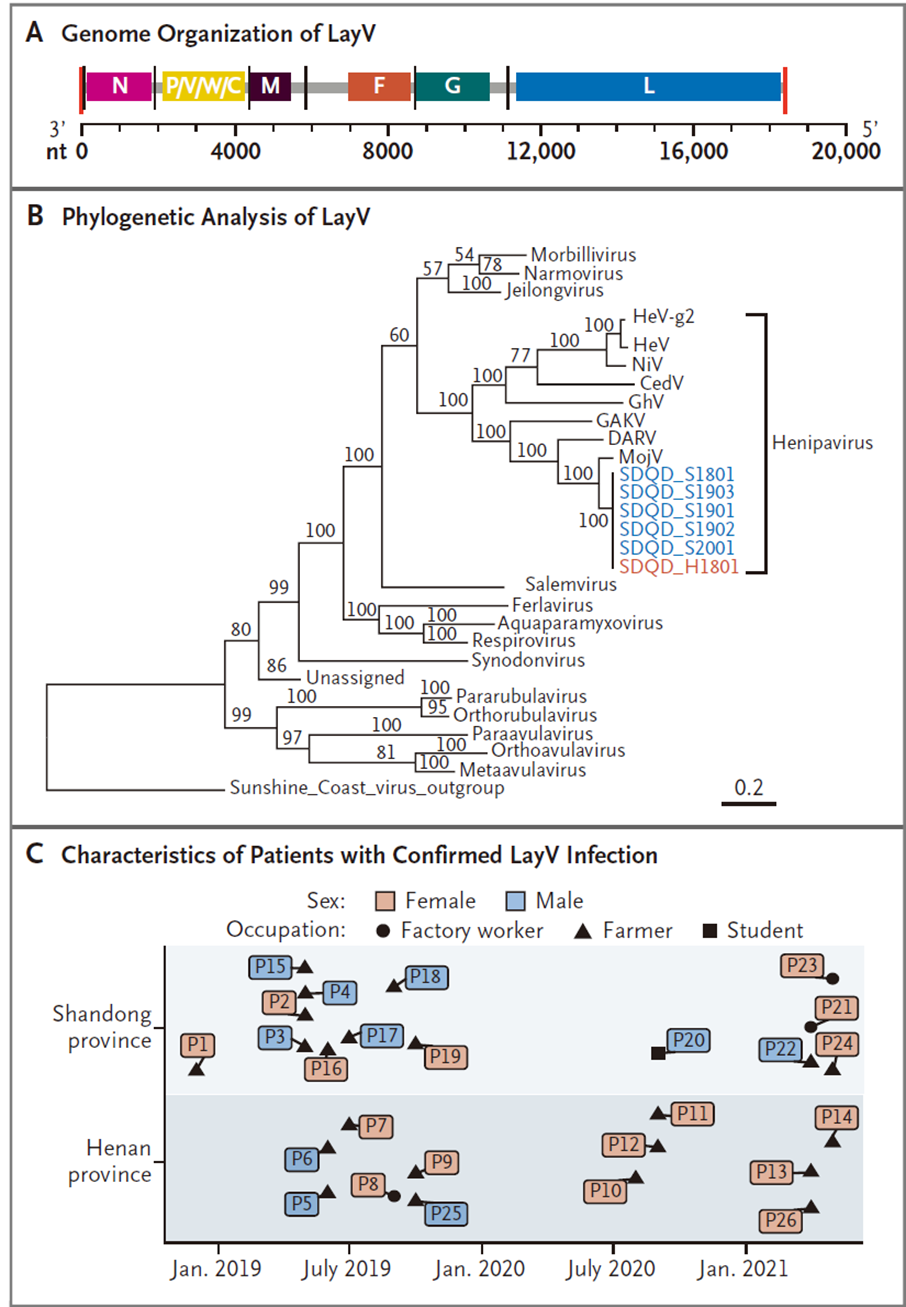
Figure 6. Genetic and epidemiological features of LayV
Key Prevention Measures: Nipah virus has a high fatality rate (40%–75%), long incubation period (up to 45 days), and human-to-human transmission risk. With no specific vaccine or treatment available, core measures include strengthened border screening, avoiding contact with wildlife and contaminated food, and strict healthcare worker protection.
Prevention and Research Outlook
The emergence of new henipaviruses and recurrent Nipah outbreaks highlight ongoing zoonotic risks from bats and shrews. Current prevention focuses on border screening, avoiding wildlife contact and contaminated food, and proper healthcare worker protection.
In research, the G protein-receptor interface and F protein fusion peptide remain key targets for vaccines and therapeutics. Advances in clinical trials, new diagnostic tools, and studies on viruses like Langya are paving the way for a comprehensive prevention system against the henipavirus family. abinScience has developed recombinant G and F proteins as well as monoclonal antibodies to support fundamental research.
Below are the latest abinScience NiV-related recombinant proteins and antibodies. Catalog numbers link directly to product pages.
abinScience Latest Product Recommendations for Nipah Virus
Protein (Recombinant Proteins)
| Catalog No. |
Product Name |
| VK631011 |
Recombinant Nipah virus G protein/Glycoprotein G Protein, C-His |
| VK521011 |
Recombinant Nipah virus/HeV F/Fusion glycoprotein F0 Protein, C-His |
| VK631012 |
Recombinant Nipah virus/NiV G protein/Glycoprotein G Protein, N-His |
| VK521012 |
Recombinant Nipah virus/NiV F/Fusion glycoprotein F0 Protein, N-His-SUMO & C-Strep |
| VK521022 |
Recombinant Nipah virus/NiV F/Fusion glycoprotein F0 Protein, N-His |
| VK432012 |
Recombinant Nipah virus/NiV Protein N/Nucleoprotein Protein, N-His |
| VK432022 |
Recombinant Nipah virus/NiV Protein N/Nucleoprotein Protein, N-His-SUMO & C-Strep |
| VK499012 |
Recombinant Nipah henipavirus M/Matrix Protein, N-His |
| VK408012 |
Recombinant Nipah virus/NiV M/Matrix Protein, N-His |
| VK078012 |
Recombinant Nipah virus Phosphoprotein, N-His |
| VK631021 |
Recombinant Nipah virus Glycoprotein, C-His |
| VK521042 |
Recombinant Nipah virus Fusion Protein, N-GST&C-His |
Antibody (Antibodies)
| Catalog No. |
Product Name |
| VK631010 |
InVivoMAb Anti-Nipah virus/NiV G protein/Glycoprotein G Antibody (nAH1.3) |
| VK521010 |
InVivoMAb Anti-Nipah virus/NiV Prefusion Antibody (4H3) |
| VK521020 |
InVivoMAb Anti-Nipah virus/NiV Prefusion Antibody (1H8) |
| VK521030 |
InVivoMAb Anti-Nipah virus/NiV Prefusion Antibody (1A9) |
| VK521040 |
InVivoMAb Anti-Nipah virus/NiV F/Fusion glycoprotein F0 Antibody (2D3) |
| VK521050 |
InVivoMAb Anti-Nipah virus/NiV Prefusion Protein Antibody (2B12) |
| VK521060 |
InVivoMAb Anti-Nipah virus/NiV Prefusion Protein Antibody (1H1) |
| VK521070 |
InVivoMAb Anti-Nipah virus/NiV Fusion glycoprotein/F Trimer Antibody (Fab92) |
| VK631020 |
InVivoMAb Anti-Nipah virus/NiV Glycoprotein G Antibody (SAA2190) |
| VK631030 |
InVivoMAb Anti-Nipah virus/NiV Glycoprotein G Antibody (SAA2191) |
| VK631040 |
InVivoMAb Anti-Nipah virus/NiV Glycoprotein G Antibody (SAA2192) |
| VK521053 |
Anti-Nipah virus/NiV Fusion glycoprotein Nanobody (DS90) |
| VK631014 |
Anti-Nipah virus/HeV G protein/Glycoprotein G Polyclonal Antibody |
| VK521014 |
Anti-Nipah virus/HeV F/Fusion glycoprotein F0 Polyclonal Antibody |
| VK432014 |
Anti-Nipah virus/HeV Protein N/Nucleoprotein Polyclonal Antibody |
| VK408014 |
Anti-Nipah virus M/Protein M Polyclonal Antibody |
| VK499014 |
Anti-Nipah henipavirus M/Matrix protein Polyclonal Antibody |
| VK521023 |
Anti-Nipah virus/NiV Fusion glycoprotein F2 Antibody (11F10) |
| VK521033 |
Anti-Nipah virus/NiV Fusion glycoprotein F1 Antibody (5G7) |
| VK521043 |
Anti-Nipah virus/NiV Fusion glycoprotein F2 Antibody (6D3) |
References
- Zhou D, Cheng R, Yao Y, et al. An attachment glycoprotein nanoparticle elicits broadly neutralizing antibodies and protects against lethal Nipah virus infection. NPJ Vaccines. 2024;9(1):158. Published 2024 Aug 31. doi:10.1038/s41541-024-00954-5
- Isaacs A, Nieto GV, Zhang X, et al. A nanobody-based therapeutic targeting Nipah virus limits viral escape. Nat Struct Mol Biol. 2025;32(10):1920-1931. doi:10.1038/s41594-025-01598-2
- Lo MK, Feldmann F, Gary JM, et al. Remdesivir (GS-5734) protects African green monkeys from Nipah virus challenge. Sci Transl Med. 2019;11(494):eaau9242. doi:10.1126/scitranslmed.aau9242
- Jin K, Huang P, Li B, et al. A Single-Copy Sensitive and Field-Deployable One-Pot RT-RPA CRISPR/Cas12a Assay for the Specific Visual Detection of the Nipah Virus. Transbound Emerg Dis. 2024;2024:4118007. Published 2024 Nov 20. doi:10.1155/2024/4118007
- Luby SP, Gurley ES, Hossain MJ. Transmission of human infection with Nipah virus. Clin Infect Dis. 2009;49(11):1743-1748. doi:10.1086/647951
- Madhukalya R, Yadav U, Parray HA, et al. Nipah virus: pathogenesis, genome, diagnosis, and treatment. Appl Microbiol Biotechnol. 2025;109(1):158. Published 2025 Jul 1. doi:10.1007/s00253-025-13474-6
- Chakraborty S, Chandran D, Mohapatra RK, et al. Langya virus, a newly identified Henipavirus in China - Zoonotic pathogen causing febrile illness in humans, and its health concerns: Current knowledge and counteracting strategies - Correspondence. Int J Surg. 2022;105:106882. doi:10.1016/j.ijsu.2022.106882
- Zhang XA, Li H, Jiang FC, et al. A Zoonotic Henipavirus in Febrile Patients in China. N Engl J Med. 2022;387(5):470-472. doi:10.1056/NEJMc2202705

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский








