Macrophages are key immune cells widely distributed throughout the body. Traditionally regarded as mere "scavengers" responsible for phagocytosing pathogens and clearing cellular debris, in-depth research has revealed that macrophages exhibit high plasticity and heterogeneity in their origin, colonization, activation states, functions, and phenotypes. Understanding this diversity is crucial for unraveling immune mechanisms, exploring disease pathogenesis, and developing therapeutic interventions.
1. Developmental Origin of Macrophages: Scientific Evolution from "Single Source" to "Dual-Origin Model"
In early immunological studies, it was generally accepted that macrophages originate from bone marrow hematopoietic stem cells (HSCs). These HSCs differentiate into monocytes in the bloodstream, which then migrate into tissues and mature gradually. However, the advancement of fate-mapping technology has reshaped this traditional view. Researchers have successively found that macrophages in many tissues are barely replenished by circulating monocytes in adulthood; instead, they can maintain their populations through self-proliferation over the long term. This phenomenon prompted a re-examination of macrophage developmental origins.
Growing evidence from embryonic development has clarified that early macrophage precursors emerge during the yolk sac and fetal liver stages. These precursors migrate into various tissues during fetal development to complete colonization, forming multiple major populations of tissue-resident macrophages. For instance, microglia in the central nervous system (CNS), Kupffer cells in the liver, and Langerhans cells in the skin all infiltrate tissues during embryonic development and are maintained via self-proliferation in adulthood.
Thus, modern immunology has formed a more precise understanding of macrophage developmental origins: macrophages do not derive from a single pathway but are composed of both embryonic and adult bone marrow-derived sources. Embryonic-origin macrophages primarily undertake long-term tissue homeostasis functions, while adult monocyte-derived macrophages (formed by monocytes migrating into tissues) are more involved in dynamic processes such as inflammation and tissue repair.
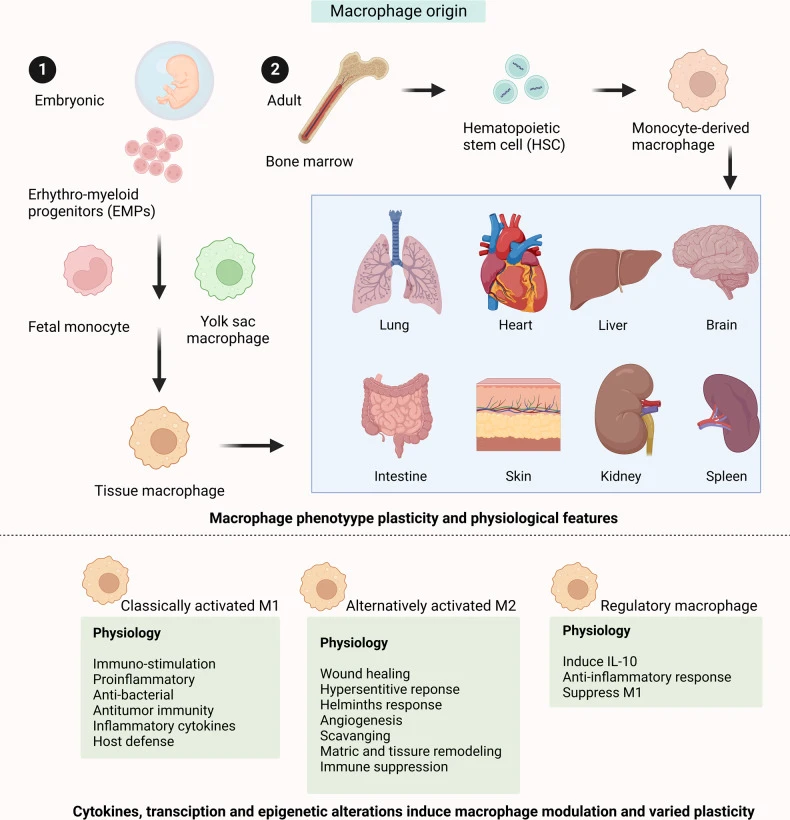
Figure 1. Origin and development of macrophages (DOI: 10.1038/s41392-023-01452-1)
2. Tissue-Resident Macrophages: Tissue Microenvironment Shapes Diverse Phenotypes
Once colonized in specific tissues, the phenotype and function of macrophages undergo profound changes under the influence of the local microenvironment, leading to highly specialized states. These cells are termed tissue-resident macrophages. Their diversity does not stem from distinct developmental pathways but is gradually shaped by signals in the unique tissue microenvironment. For example: Microglia respond to signals from neurons and glial cells in the CNS, developing unique immune homeostasis functions; Kupffer cells, chronically exposed to gut-derived metabolites in the liver, acquire robust tolerance and clearance capabilities; Alveolar macrophages adapt to the surfactant-rich alveolar environment, forming efficient phagocytic functions. Each population of tissue-resident macrophages profoundly embodies the principle of "tissue dictates fate."
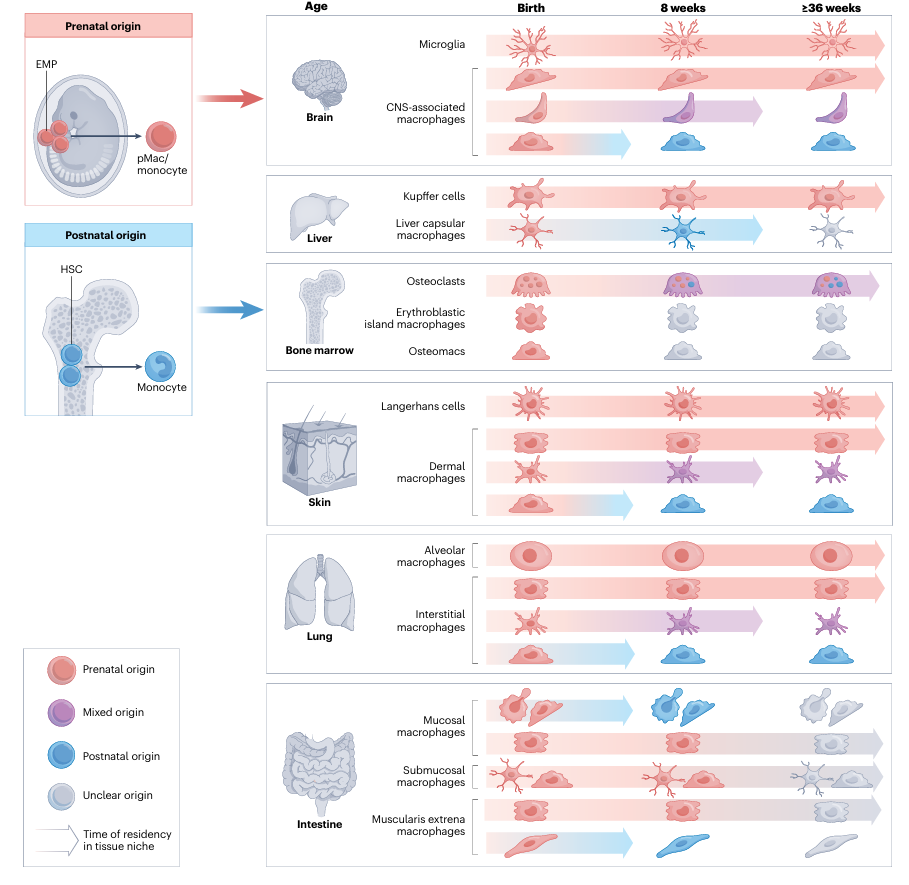
Figure 2. Types of tissue-resident macrophages (DOI: 10.1038/s41577-023-00848-y)
3. M1/M2 Macrophages: The Classical Classification System
Macrophage classification remains a highly controversial topic. Multiple factors can induce distinct phenotypes and activation states, including signaling molecules, growth factors, transcription factors, epigenetic and post-transcriptional mechanisms, as well as niche signals such as cytokines, cell-cell contacts, and metabolites. Generally, macrophages are classified into two subtypes based on their functions and activation patterns: classically activated M1 macrophages and alternatively activated M2 macrophages.
3.1 Classically Activated M1 Macrophages (Characterized by Proinflammatory and Immune Effector Functions)
The M1 phenotype represents a population of macrophages primarily specialized in proinflammatory responses and immune effector functions. Experimentally and histologically, common M1-associated characteristics include high expression or potent functional activity of proinflammatory mediators and molecules (e.g., TNF-α, IL-1β, IL-12) as well as molecular products involved in bactericidal/lytic processes (e.g., ROS, NO). At the flow cytometry or transcriptomic level, M1-related markers are typically accompanied by elevated expression of MHC II and co-stimulatory molecules (e.g., CD80/CD86), which is consistent with their strong capacity for antigen presentation and activation of adaptive immunity.
In clinical or pathological contexts, M1 macrophages are commonly observed in acute infections, certain inflammatory lesions, and immunocompetent regions of some tumors. Their core functions can be summarized as eliminating pathogens, limiting lesion spread, and initiating or sustaining robust immune responses.
3.2 Alternatively Activated M2 Macrophages (Characterized by Immunoregulation and Tissue Repair)
The M2 phenotype denotes a population of macrophages focused on immune suppression, tissue repair, and remodeling. At the molecular and functional levels, M2-related macrophages tend to secrete anti-inflammatory or reparative factors (e.g., IL-10, TGF-β) and play key roles in processes such as efferocytosis (clearance of apoptotic cells), promotion of stromal reconstruction, and angiogenesis. Common surface or metabolism-related markers include CD206 (mannose receptor) and certain arginine metabolism-related enzymes (e.g., Arg1).
M2-like phenotypes are prevalent during tissue repair phases, late-stage parasitic infections, and the microenvironment of many chronic diseases or tumors. Their biological roles are geared toward limiting excessive inflammation, restoring tissue homeostasis, and supporting repair processes.
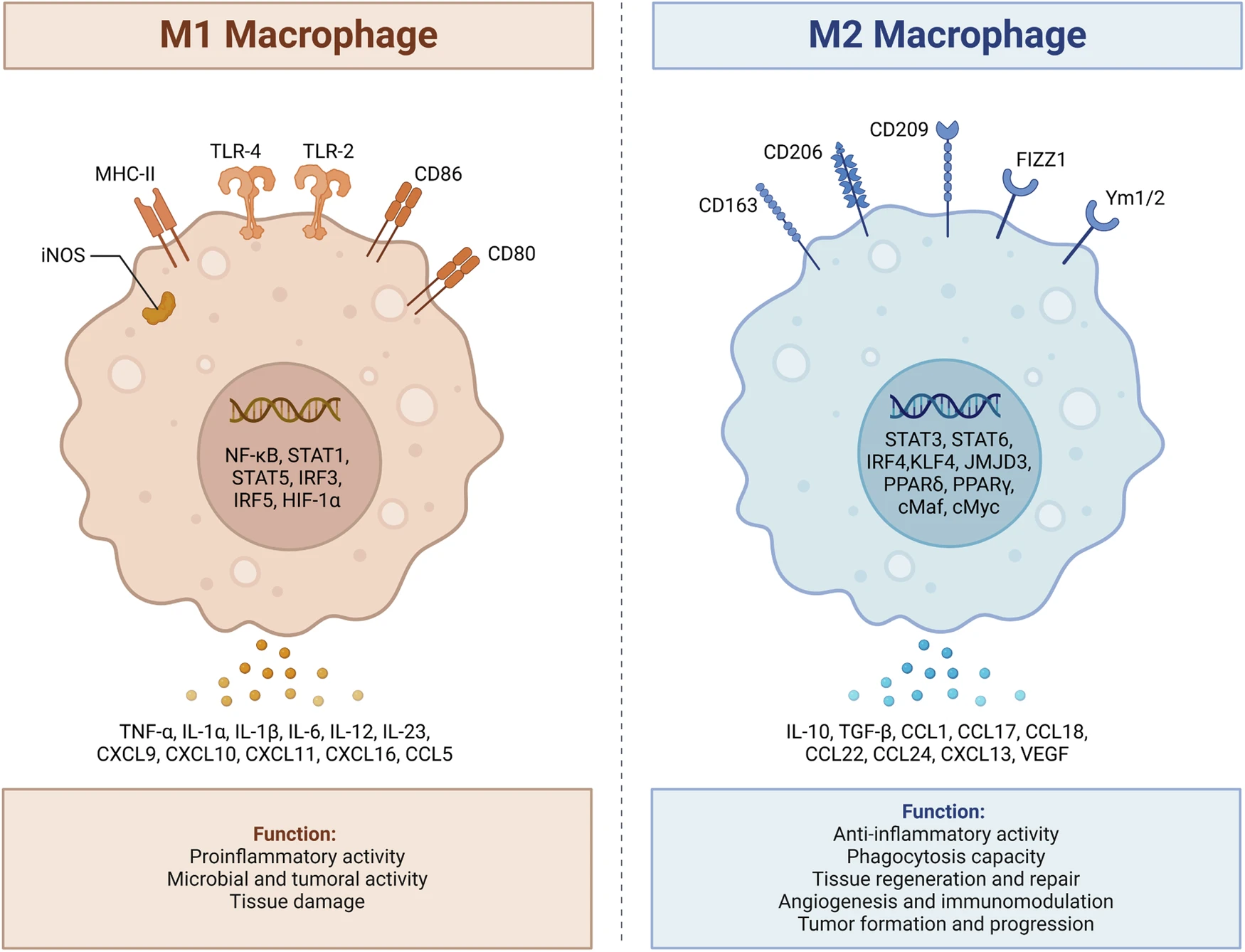
Figure 3. Two phenotypes of macrophages (DOI: 10.1038/s41419-024-06888-z)
4. Specialized Macrophage Subsets: New Perspectives on Tumor Immunity and Disease Regulation
In recent years, advancements in single-cell omics, spatial transcriptomics, and fate-mapping technologies have led to the recognition of a series of highly specialized macrophage subtypes beyond resident and non-resident populations in specific disease contexts or tissue microenvironments. These subsets accumulate significantly under certain pathological conditions, possess unique functions and markers, and exert important impacts on disease progression.
4.1 Tumor-Associated Macrophages (TAMs)
In tumor tissues, TAMs are among the most abundant and functionally complex immune components. Chronic exposure to tumor-secreted cytokines, lactic acid accumulation, and hypoxic environments endows TAMs with highly adaptive immune-suppressive and tissue-remodeling characteristics. TAMs produce large quantities of immunosuppressive factors that impair the ability of effector T cells to recognize tumors; simultaneously, they secrete MMPs and VEGF to promote tumor angiogenesis and stromal remodeling, creating a more favorable niche for tumor growth and metastasis.
Notably, TAMs are not a homogeneous population but consist of multiple subsets with distinct functional orientations—such as regional TAMs enriched in antigen-presentation genes, perivascular angiogenic TAMs, and inflammatory TAMs under long-term metabolic stress. Collectively, they form a critical component of the tumor ecosystem and represent a key target for cancer immunotherapy in recent years.
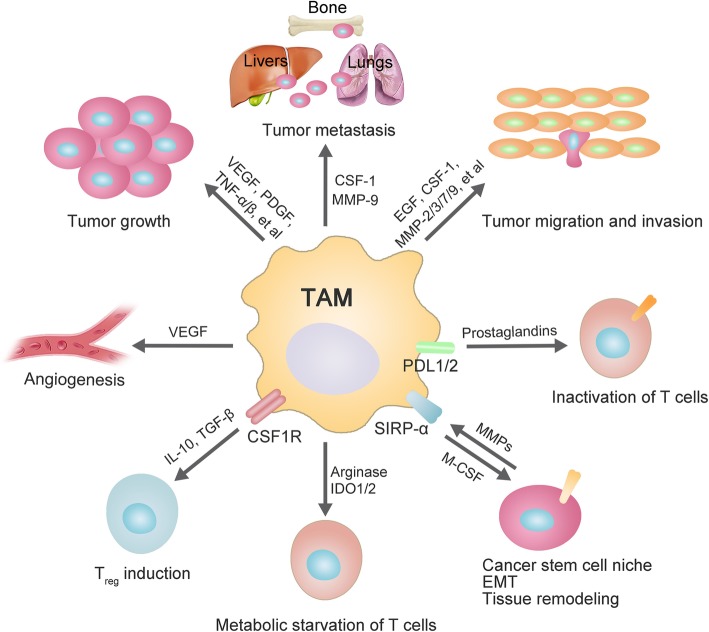
Figure 4. Roles of TAMs in tumorigenesis (DOI: 10.1186/s12929-019-0568-z)
4.2 CD169+ Macrophages
Unlike TAMs in the tumor microenvironment, CD169+ macrophages are primarily distributed in anatomical structures responsible for antigen drainage, such as the subcapsular sinuses of lymph nodes and the marginal zone of the spleen. They serve as important sentinels for recognizing pathogens invading through the bloodstream or lymphatic system. These macrophages efficiently capture antigens reaching immune organs via humoral circulation and collaborate with dendritic cells to activate naive CD8+ T cells, thus playing a pivotal role in antiviral immunity and vaccine-induced responses.
Recent studies on acute viral infections and vaccine responses have demonstrated that CD169+ macrophages exert a far greater impact on early immune responses than previously recognized—their functional state may even determine the speed and quality of immune responses, acting as a crucial hub connecting innate and adaptive immunity.
4.3 TCR+ Macrophages
Compared to the aforementioned two subsets, TCR+ macrophages are a more "novel" discovery, repeatedly observed in multiple chronic disease models, tumor tissues, and long-term inflammatory environments. Their most notable feature is the expression of the TCR/CD3 complex, a molecule traditionally associated with T cells. This observation suggests that these macrophages may possess unique signal recognition and regulatory capabilities, enabling communication with surrounding cells through non-canonical pathways.
While their specific functions remain under investigation, existing evidence indicates their involvement in maintaining local immune balance, regulating inflammation, and remodeling the tissue microenvironment. With the continuous advancement of spatial omics and ligand-receptor interaction analysis tools, TCR+ macrophages are likely to emerge as a key focus for uncovering novel immune regulatory mechanisms in future research.
5. Conclusion
Macrophages are far more than simple "scavengers"—they are highly plastic, multifunctional cells in the body's immune system. From tissue-resident macrophages colonizing tissues during embryonic development to inflammation-associated macrophages replenished by monocytes in adulthood, and specialized subsets such as TAMs, CD169+ macrophages, or TCR+ macrophages emerging in specific disease contexts, each type of macrophage fulfills unique functions in its respective microenvironment, regulating immune responses, tissue homeostasis, and repair processes.
References
[1] Mass E, Nimmerjahn F, Kierdorf K, Schlitzer A. Tissue-specific macrophages: how they develop and choreograph tissue biology. Nat Rev Immunol. 2023 Sep;23(9):563-579.
[2] Chen S, Saeed AFUH, Liu Q, Jiang Q, Xu H, Xiao GG, Rao L, Duo Y. Macrophages in immunoregulation and therapeutics. Signal Transduct Target Ther. 2023 May 22;8(1):207.
[3] Li D, Zhang T, Guo Y, Bi C, Liu M, Wang G. Biological impact and therapeutic implication of tumor-associated macrophages in hepatocellular carcinoma. Cell Death Dis. 2024 Jul 12;15(7):498.
[4] Chen Y, Song Y, Du W, Gong L, Chang H, Zou Z. Tumor-associated macrophages: an accomplice in solid tumor progression. J Biomed Sci. 2019 Oct 20;26(1):78.
[5] Jin R, Neufeld L, McGaha TL. Linking macrophage metabolism to function in the tumor microenvironment. Nat Cancer. 2025 Feb;6(2):239-252.
abinScience offers a comprehensive range of antibodies and recombinant proteins for macrophage research, widely applicable in experiments such as ELISA, WB, IHC, and FCM. These products empower researchers to conduct in-depth studies on macrophage polarization, activation, protein expression, functions, and phenotypes. For more macrophage-related products, visit the abinScience official website: www.abinscience.com
+86-027-65523339
중국 우한시 심둔사로 666번지 C동, 우한, 430206

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский