I. Introduction to Macrophages
As a core component of the innate immune system, macrophages originate from bone marrow hematopoietic stem cells. After progressing through the monocyte stage, they colonize various tissues and organs, differentiating into tissue-resident macrophages with specialized functions—such as Kupffer cells in the liver, microglia in the brain, and osteoclasts in bone tissue. Their crucial roles in host defense, inflammatory regulation, tissue homeostasis maintenance, and disease progression have been widely documented.
In recent years, macrophage research has emerged as a hotspot in immunology and biomedicine. From basic immunology to clinical studies, growing attention has been paid to the roles of macrophages in infection, tumorigenesis, immune regulation, and chronic diseases. According to literature data from PubMed, the number of macrophage-related studies has increased annually, exceeding 22,000 publications in 2025. With deeper insights into macrophage functions, an increasing number of studies are focusing on their roles under different pathological conditions and their potential therapeutic applications.
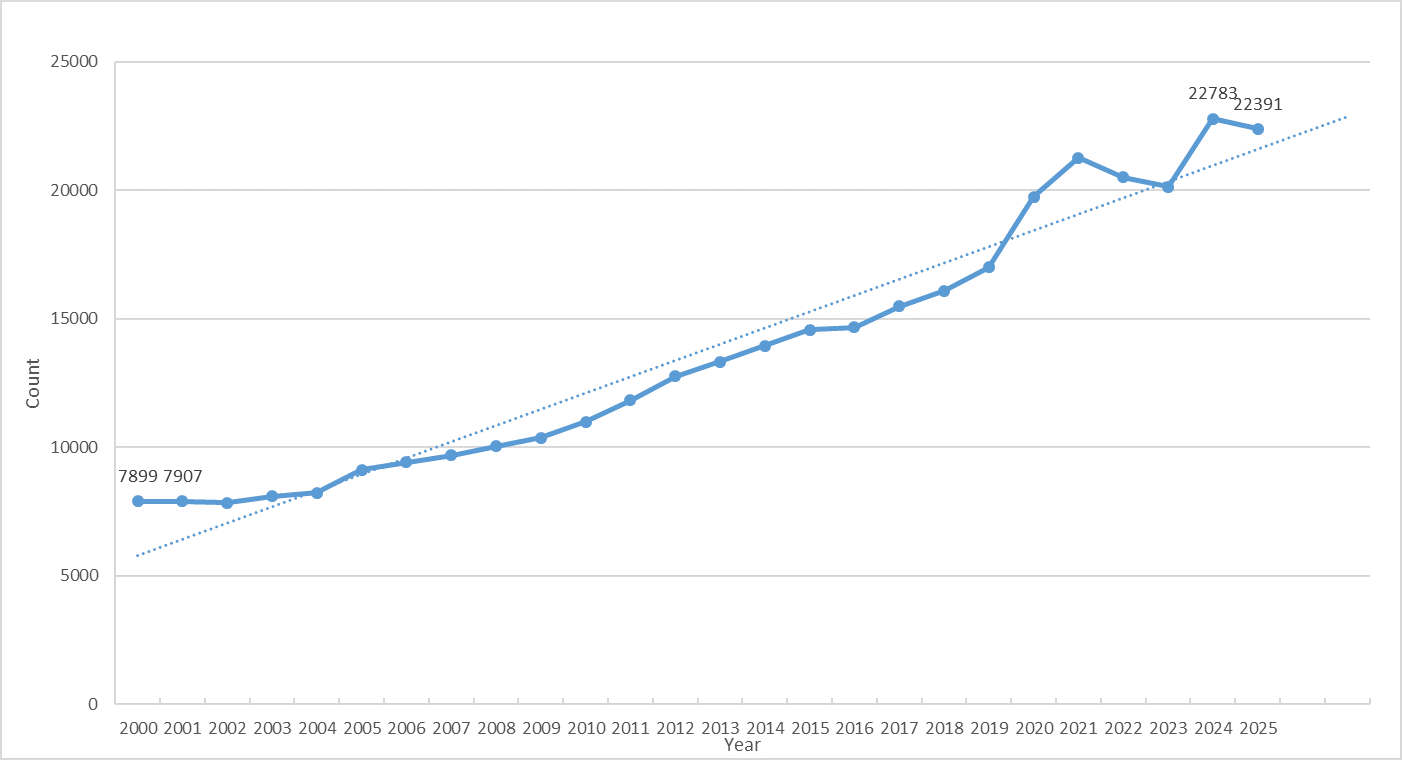
Figure 1. Number of macrophage-related publications (Data source: PubMed)
II. Classification of Macrophages
The classification of macrophages remains a highly controversial topic in immunology. Their phenotypes and functions are regulated by multiple factors, including cytokines, chemokines, transcription factors, epigenetic modifications, metabolic reprogramming, and microenvironmental signals.
Currently, the most widely accepted classification system categorizes macrophages into two major subsets based on their activation status: classically activated macrophages (M1 type) and alternatively activated macrophages (M2 type).
M1 macrophages are primarily induced by interferon-gamma (IFN-γ), lipopolysaccharide (LPS), and granulocyte-macrophage colony-stimulating factor (GM-CSF). They highly express pro-inflammatory cytokines such as TNF-α, IL-6, and IL-12, and produce inducible nitric oxide synthase (iNOS). Possessing potent microbicidal and anti-tumor capabilities, M1 macrophages may also cause tissue damage under certain circumstances.
In contrast, M2 macrophages tend to exert immunosuppressive effects and are involved in processes such as tissue repair and tumor progression. M2 macrophages can be further subdivided into multiple subtypes (M2a, M2b, M2c, M2d), induced by IL-4/IL-13, immune complexes, IL-10/glucocorticoids, and other factors, respectively. They are characterized by high expression of molecules including CD206, CD163, Arg-1, and IL-10.
Notably, while the M1 and M2 phenotypes are widely used in experimental settings, they are not mutually exclusive. These subsets can interconvert under different microenvironments, forming a continuous functional spectrum that reflects the high plasticity of macrophages.
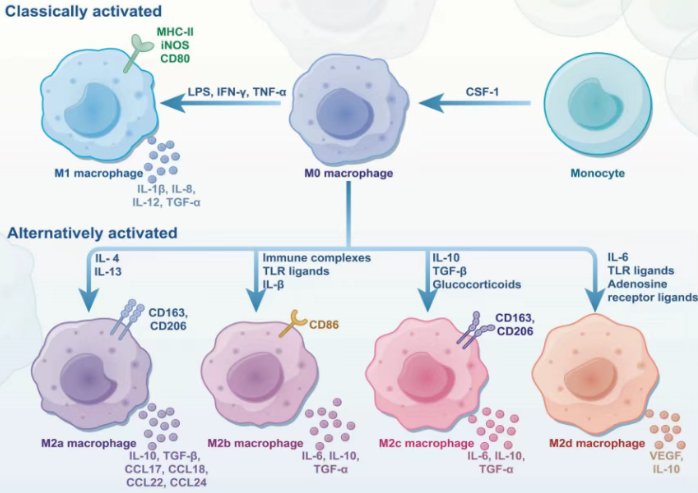
Figure 2. CSF-1-induced differentiation of monocytes into macrophages(DOI: 10.1038/s41392-025-02124-y)
Table 1. Phenotypes of Different Macrophages (DOI: 10.1002/JLB.3RU1018-378RR)
| Phenotypes |
Stimuli |
Markers |
Functions |
| Human monocytes/macrophages |
| M1 |
IFN‐γ, LPS, GM‐CSF, TNF‐α |
CXCL9, IL‐12high/IL‐10low, iNOS, IL‐6, CD80, CD86, TNF‐α |
Pro‐inflammation, microbicidal effect, tumor resistance |
| M2a |
IL‐4, IL‐13 |
CCL17, IL‐1R, CD206, Dectin‐1, IL‐10, DC‐SIGN |
Anti‐inflammatory, wound healing |
| M2b |
LPS+IC, IL‐1β+IC |
CCL1, IL‐10high/IL‐12low, TNF‐α, CD86, IL‐6 |
Immunoregulation, promoting infection, tumor progression |
| M2c |
IL‐10, Glucocorticoids |
CXCL13, CD206, CD163, IL‐10, TGF‐β, MerTK |
Immunosuppression, phagocytosis, tissue remodeling |
| M2d |
LPS+A2R ligands, IL‐6 |
VEGF, IL‐10, TGF‐β |
Tumor progression, angiogenesis |
| Mouse monocytes/macrophages |
| M1 |
IFN‐γ, LPS, GM‐CSF, TNF‐α |
CXCL9, IL‐12high/IL‐10low, iNOS, IL‐6, CD80, CD86, TNF‐α |
Pro‐inflammation, microbicidal effect, tumor resistance |
| M2a |
IL‐4, IL‐13 |
CCL17, IL‐1R, Dectin‐1, IL‐10, Arg‐1, Chil3, FIZZ1 |
Anti‐inflammatory, wound healing |
| M2b |
LPS+IC, IL‐1β+IC |
CCL1, IL‐10high/IL‐12low, TNF‐α, CD86, IL‐6, LIGHT |
Immunoregulation, promoting infection, tumor progression |
| M2c |
IL‐10, Glucocorticoids |
CXCL13, CD206, CD163, IL‐10, TGF‐β, MerTK, Arg‐1 |
Immunosuppression, phagocytosis, tissue remodeling |
| M2d |
LPS+A2R ligands, IL‐6 |
VEGF, IL‐10, TGF‐β, iNOS |
Tumor progression, angiogenesis |
III. Functions of Macrophages
Macrophages exhibit diverse functions in the immune system, participating not only in immune defense but also in tissue repair, chronic inflammation, and tumor immunity.
Immune Defense: Macrophages effectively eliminate harmful substances from the body by phagocytosing pathogens, dead cells, and other exogenous materials, protecting the host from infections.
Immune Regulation: Macrophages can secrete immunosuppressive cytokines (e.g., IL-10, TGF-β) to inhibit excessive immune responses, thereby preventing the development of autoimmune diseases.
Tissue Repair: During tissue injury or inflammation, macrophages contribute to tissue repair and regeneration by secreting pro-reparative factors—with M2 macrophages playing a particularly critical role in this process.
Tumor Immunity: Macrophages display complex and diverse functions in the tumor microenvironment (TME). Some tumor cells induce the conversion of macrophages to the M2 phenotype, which suppresses immune responses and promotes tumor growth and metastasis.
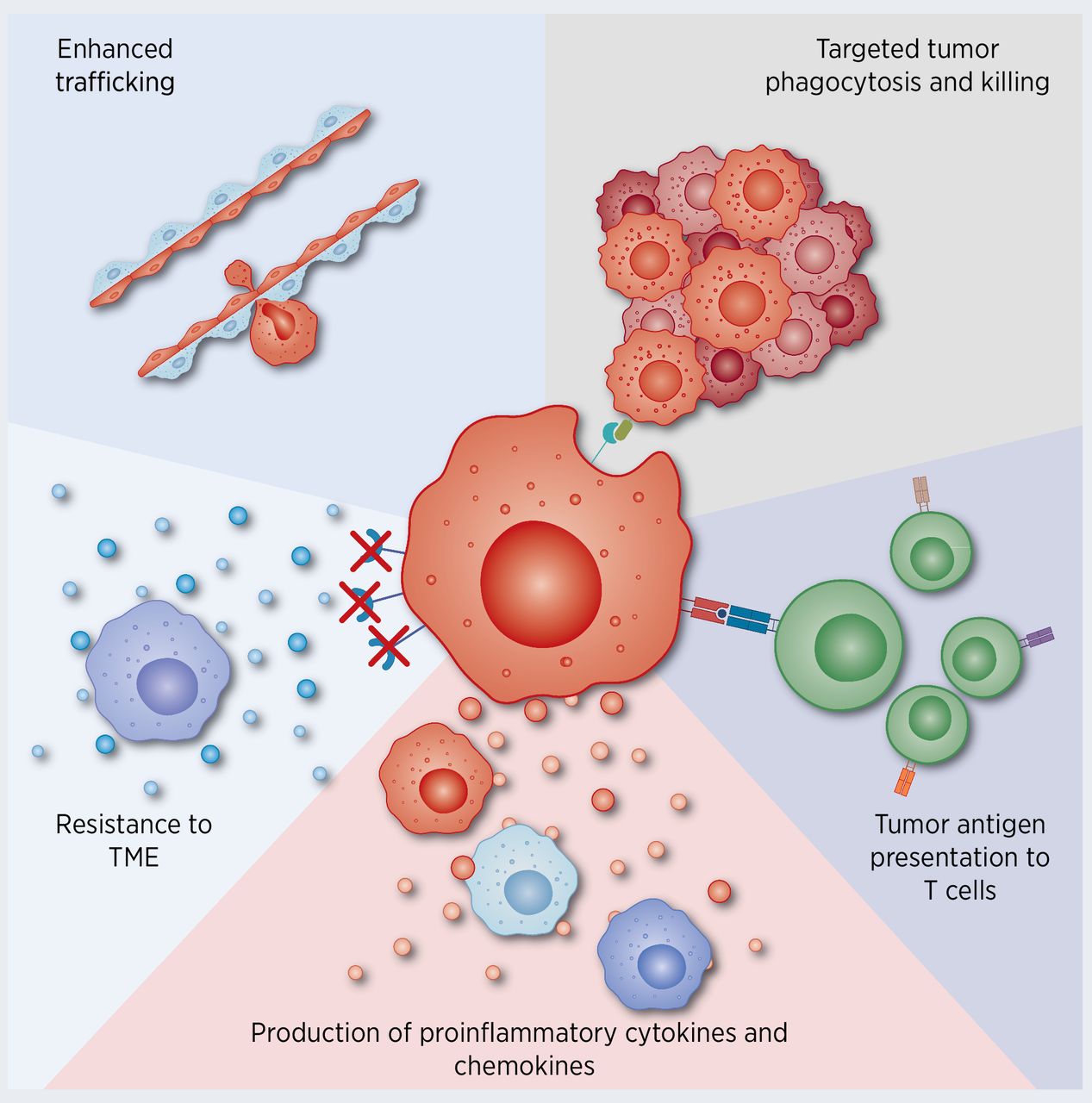
Figure 3. Macrophages and tumor immunity (DOI: 10.1158/0008-5472.CAN-20-2990)
IV. Detection Methods for Macrophages
The detection of macrophages is fundamental to studying their functions, phenotypes, and roles in various immune responses. Commonly used detection methods include:
1. Gene Expression Analysis: Quantitative real-time PCR (qPCR) technology enables the detection of expression levels of M1- and M2-related genes, such as TNF-α, IL-10, Arg-1, and iNOS.
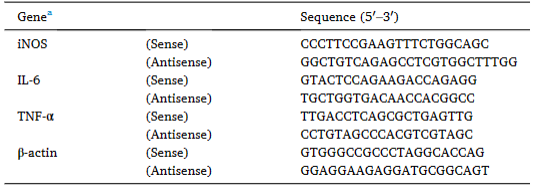
Figure 4. Primer sequences for quantitative real-time PCR
2. Protein Level Detection: Western Blot (WB) can be used to detect the expression of specific proteins, such as iNOS, Arg-1, and CD206. Enzyme-linked immunosorbent assay (ELISA) and liquid chip technology allow for the quantitative measurement of cytokine levels (e.g., IL-6, IL-10, TNF-α) in cell supernatants or serum.
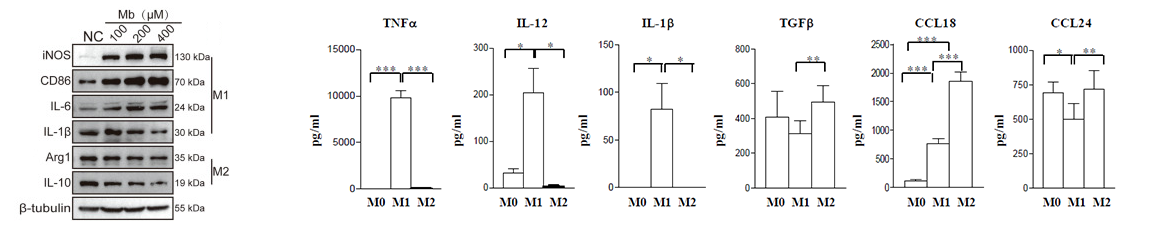
Figure 5. WB/ELISA detection of macrophage-related markers (DOI: 10.3389/fimmu.2017.01097)
3. IHC/IF: These techniques are used for the localization and phenotypic identification of macrophages in tissues.
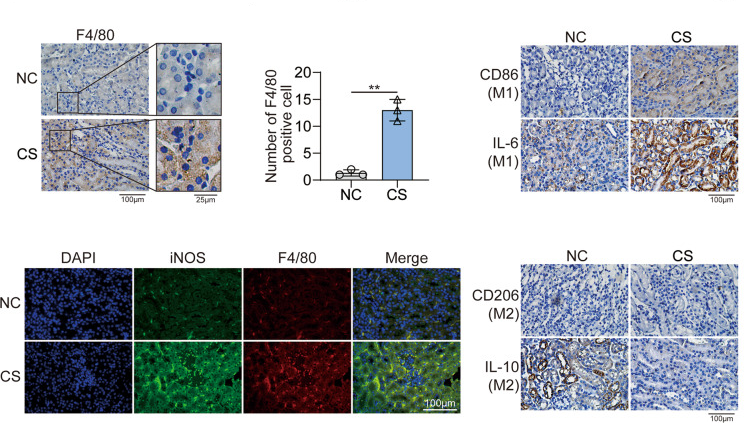
Figure 6. IHC/IF detection of macrophages (DOI: 10.1038/s41420-022-00894-w)
4. Flow Cytometry: Combined with multiple surface markers and intracellular staining, flow cytometry enables multiparametric phenotypic analysis and subtyping of macrophages. It is widely applied in macrophage function research and disease diagnosis.
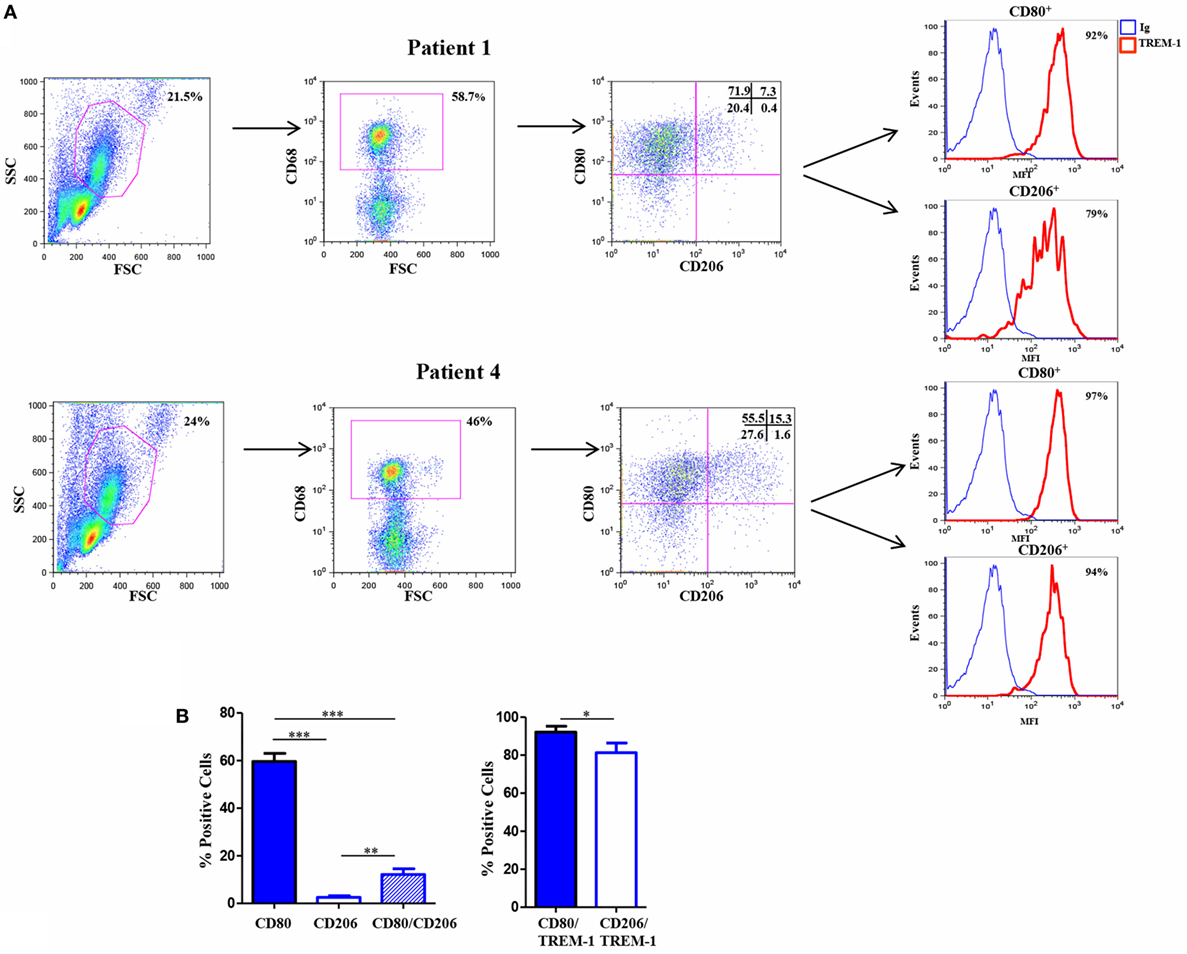
Figure 7. Flow cytometry results of mouse macrophages (DOI: 10.3389/fimmu.2017.01097)
abinScience Products for Macrophage Detection
1. Flow Cytometry Antibodies
2. Recombinant Proteins
3. Other Antibodies
| Target |
Reactivity |
Product Name |
Application |
Catalog No. |
| iNOS |
Human |
Anti-NOS2 Polyclonal Antibody |
ELISA, IHC, WB |
HW388014 |
| Arg-1 |
Human, Mouse, Rat |
Anti-ARG1/Arginase-1 Polyclonal Antibody |
ELISA, IHC, WB |
HY339024 |
| Human, Mouse, Rat, Pig, etc |
Anti-ARG1 Polyclonal Antibody |
ELISA, IHC, WB |
HY339014 |
| CD11b |
Human |
Anti-CD11b/ITGAM Polyclonal Antibody |
ELISA, IHC, WB |
HY474014 |
| Mouse |
Anti-Mouse CD11b/ITGAM Polyclonal Antibody |
ELISA, IHC, WB |
MY474014 |
| CD86 |
Human |
Anti-CD86 Polyclonal Antibody |
ELISA, IHC, WB |
HW776014 |
| Mouse |
Anti-Mouse CD86/B7-2 Polyclonal Antibody |
ELISA, IHC, WB |
MW776014 |
| CD206 |
Human, Mouse, Rat |
Anti-CD206/MRC1 Polyclonal Antibody |
ELISA, IHC, WB |
HB976014 |
| IL-1 |
Human, Cercocebus atys, Macaca fascicularis, etc |
Anti-IL1B/IL1F2 Polyclonal Antibody |
ELISA, IHC, WB |
HF943014 |
| IL-6 |
Human |
Anti-Human IL6 Antibody |
ELISA, FCM, WB, IHC, IF |
HY328033 |
| Mouse |
Anti-Mouse IL6 Monoclonal Antibody |
ELISA, IHC, WB |
MY328085 |
| IL-10 |
Human |
Anti-Human IL10 Antibody |
ELISA, WB, IHC, FCM |
HB997023 |
| TNF-α |
Human, Dog, Cat, Pig, etc |
Anti-TNFa/TNF-alpha Polyclonal Antibody |
ELISA, IHC, WB |
HF879014 |
| Mouse, Rat, Peromyscus leucopus |
Anti-TNFa/TNF-alpha Polyclonal Antibody |
ELISA, IHC, WB |
MF879014 |
| Danio rerio |
Anti-Zebrafish TNFa Polyclonal Antibody |
ELISA, IHC, WB |
ZA439014 |
| TGF‐β |
Human, Mouse, Dog, Rat, etc |
Anti-TGFB1/TGF-beta-1 Polyclonal Antibody |
ELISA, IHC, WB |
HF977014 |
| Mouse |
Anti-Mouse TGFB1/TGF-beta-1 Polyclonal Antibody |
ELISA, IHC, WB |
MF977014 |
References
[1] Guan F, Wang R, Yi Z, Luo P, Liu W, Xie Y, Liu Z, Xia Z, Zhang H, Cheng Q. Tissue macrophages: origin, heterogenity, biological functions, diseases and therapeutic targets. Signal Transduct Target Ther. 2025 Mar 7;10(1):93.
[2] Anderson NR, Minutolo NG, Gill S, Klichinsky M. Macrophage-Based Approaches for Cancer Immunotherapy. Cancer Res. 2021 Mar 1;81(5):1201-1208.
[3] Raggi F, Pelassa S, Pierobon D, Penco F, Gattorno M, Novelli F, Eva A, Varesio L, Giovarelli M, Bosco MC. Regulation of Human Macrophage M1-M2 Polarization Balance by Hypoxia and the Triggering Receptor Expressed on Myeloid Cells-1. Front Immunol. 2017 Sep 7;8:1097.
[4] Li N, Chen J, Geng C, Wang X, Wang Y, Sun N, Wang P, Han L, Li Z, Fan H, Hou S, Gong Y. Myoglobin promotes macrophage polarization to M1 type and pyroptosis via the RIG-I/Caspase1/GSDMD signaling pathway in CS-AKI. Cell Death Discov. 2022 Feb 28;8(1):90.
[5] Raggi F, Pelassa S, Pierobon D, Penco F, Gattorno M, Novelli F, Eva A, Varesio L, Giovarelli M, Bosco MC. Regulation of Human Macrophage M1-M2 Polarization Balance by Hypoxia and the Triggering Receptor Expressed on Myeloid Cells-1. Front Immunol. 2017 Sep 7;8:1097.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский









