Within the immune system's defensive vanguard, macrophages are core players endowed with both "combat" and "repair" capabilities. They are widely distributed throughout body tissues, capable of vigorously clearing invading pathogens and participating in tissue repair and remodeling after injury. The key to this functional duality lies in their unique ability to undergo polarization—a process of phenotypic switching. Macrophages can adapt their phenotype in response to microenvironmental signals, forming distinct functional subtypes such as pro-inflammatory and anti-inflammatory, thereby regulating immune homeostasis.
1. What is Macrophage Polarization?
The most remarkable biological feature of macrophages is their functional plasticity. Under stimulation by exogenous signals (e.g., cytokines, pathogen-associated molecular patterns), they can differentiate into subtypes with specific phenotypes and functions. This process is termed macrophage polarization.
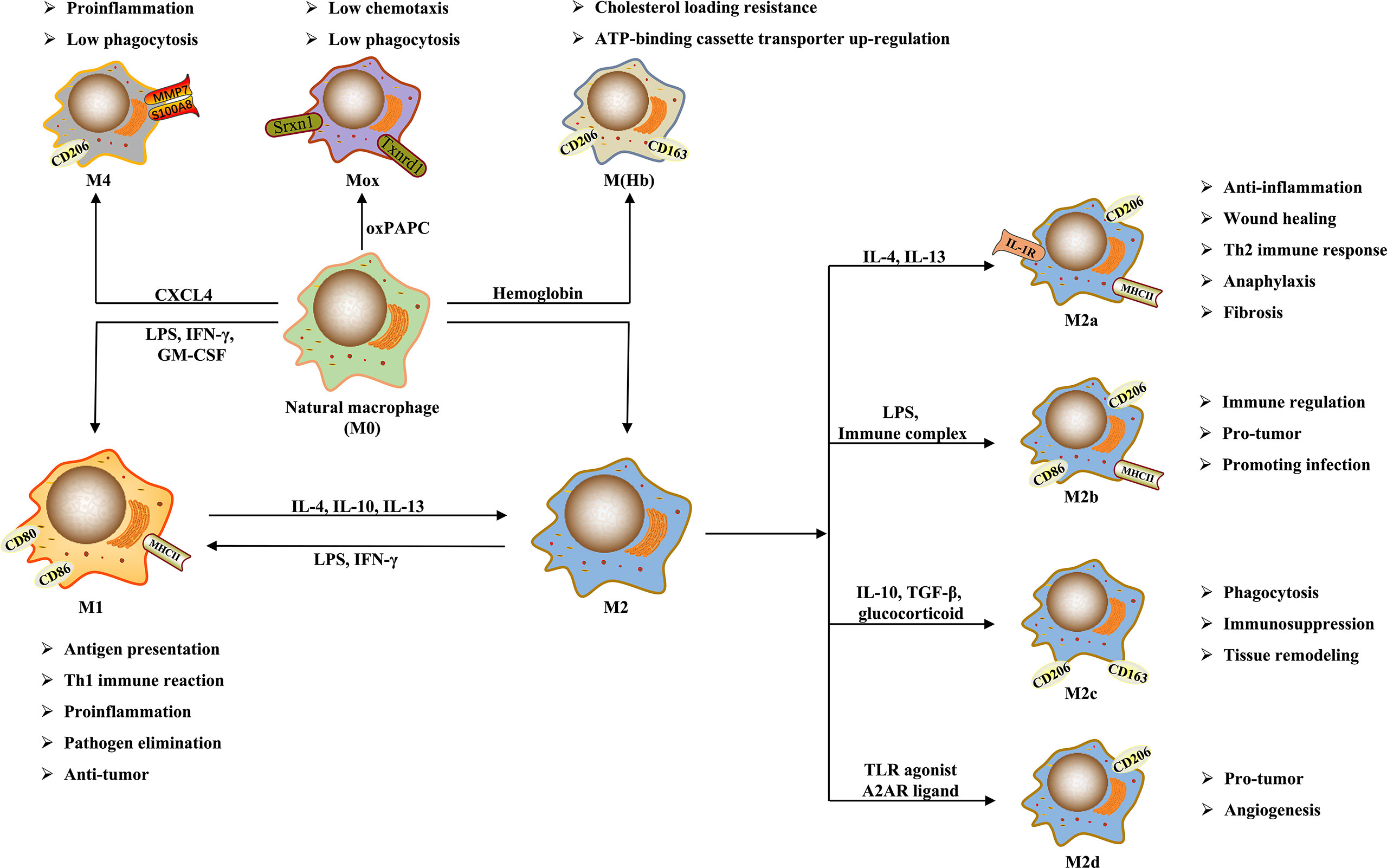
Figure 1. Macrophage Polarization (DOI: 10.3389/fimmu.2021.803037)
Polarization is a key adaptive mechanism for macrophages to respond to microenvironmental changes. In pathological states such as infection and inflammation, macrophages can shift towards a pro-inflammatory phenotype to eliminate pathogens and initiate immune responses. Conversely, during tissue repair and the maintenance of immune homeostasis, they tend towards an anti-inflammatory phenotype, suppressing excessive inflammation and promoting tissue healing. Importantly, macrophage polarization is not an irreversible, one-way process; phenotypes can dynamically switch in response to changing microenvironmental signals, reflecting the flexibility of immune regulation.
2. Classic Classification of Macrophage Polarization
Based on differences in functional phenotypes and inducing signals, macrophage polarization is classically categorized into M1 (classically activated) and M2 (alternatively activated) types. Although this binary classification simplifies the complex phenotypic spectrum in vivo, it provides a clear functional framework for experimental research.
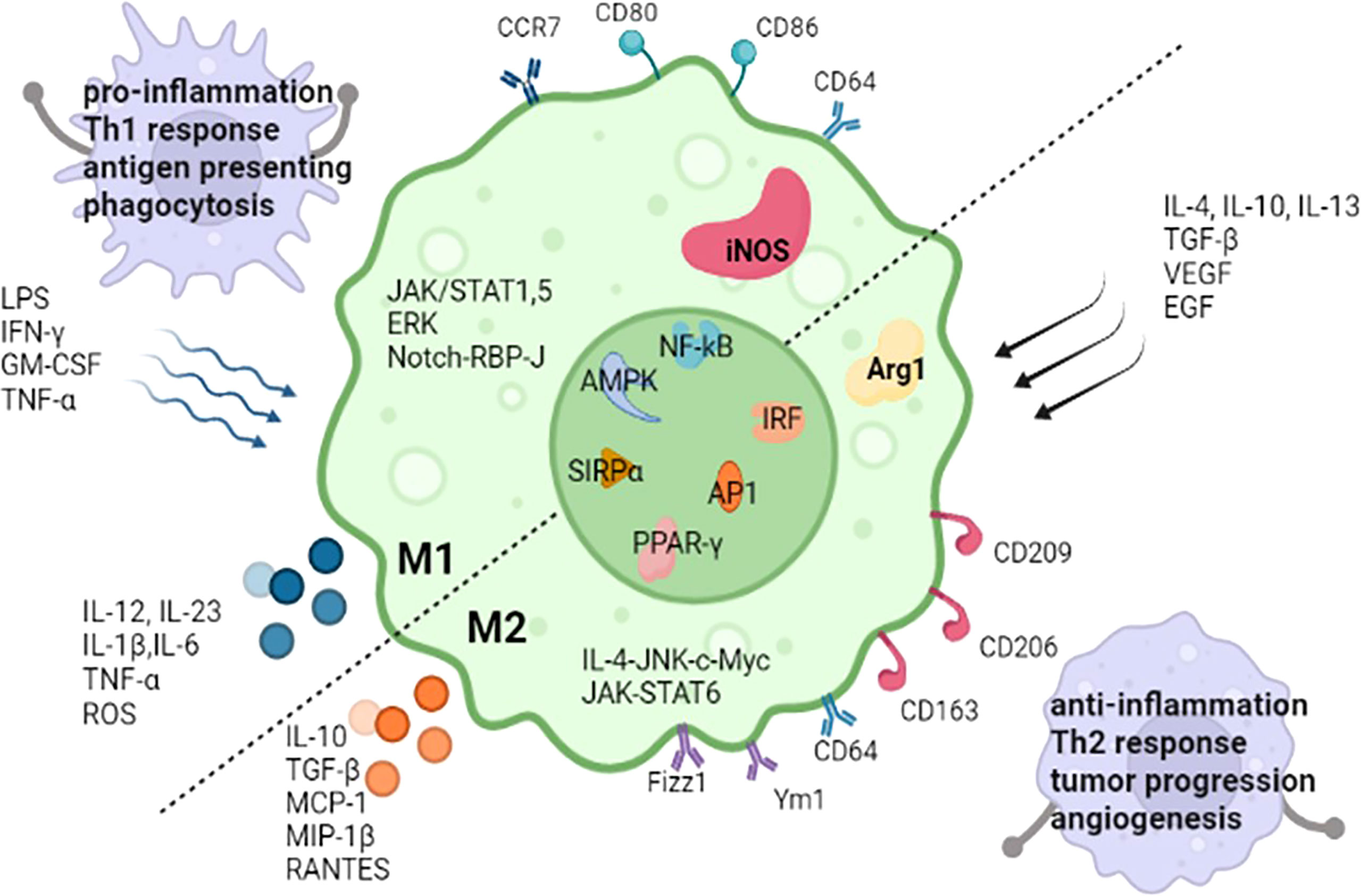
Figure 2. M1 and M2 Macrophages (DOI: 10.3389/fimmu.2022.880286)
2.1 M1 Macrophages (Classically Activated)
M1 macrophages are primarily activated by interferon-gamma (IFNγ) alone or in combination with signals like lipopolysaccharide (LPS) and tumor necrosis factor (TNF). Their core function is to initiate and amplify inflammatory responses, exerting antimicrobial and anti-tumor effects through the secretion of large quantities of pro-inflammatory cytokines and antimicrobial molecules.
Key markers include:
2.2 M2 Macrophages (Alternatively Activated)
M2 macrophages constitute a functionally heterogeneous population. Based on inducing signals and functional differences, they can be further subdivided into M2a, M2b, M2c, and M2d subtypes. Their core functions involve anti-inflammatory activity, immune regulation, tissue repair, and angiogenesis. Key characteristics of each subtype are as follows:
| Subtype | Primary Inducers | Core Markers | Core Functions |
|---|---|---|---|
|
M2a |
IL-4, IL-13 |
CD206 (MRC1), CD200R, TGM2, CCL17, CCL22 |
Wound healing, tissue repair, anti-parasitic infection |
|
M2b |
Immune complexes, TLR agonists |
IL-6, IL-10, TNF |
Immune regulation, inflammatory balance |
|
M2c |
IL-10, glucocorticoids, TGFβ |
CD163, IL-10, TGFβ |
Anti-inflammatory, immune tolerance induction, tissue remodeling |
|
M2d |
TLR agonists, adenosine receptor activation |
IL-10, IL-12, TGFβ |
Angiogenesis, tumor-associated macrophage phenotype |
3. Macrophage Polarization Experiments
Polarization experiments are a "core tool" in macrophage research. Their fundamental value lies in "artificially manipulating phenotypes" to decipher the roles of macrophages in physiology and pathology. These experiments can be broadly categorized into three scenarios:
Defining the experimental objective is crucial for selecting the appropriate cell model and detection methods—e.g., THP-1 cells (human origin) for human disease studies, and BMDMs (primary cells) for investigating murine in vivo mechanisms, as they better reflect physiological conditions.
3.1 Common Macrophage Polarization Protocols
In research, polarization experiments are commonly performed using THP-1 (human cell line), RAW264.7 (mouse cell line), and BMDMs (mouse bone marrow-derived macrophages). Due to differences in origin and characteristics, their polarization protocols vary significantly, as detailed in the table below:
| Cell Type | Source & Characteristics | Pre-treatment Requirements | Polarization Induction Protocol | Key Markers (Species-Specific) |
|---|---|---|---|---|
|
THP-1 |
Human monocytic cell line, easy to culture, fast proliferation, suitable for high-throughput assays |
Requires PMA (100 ng/mL) treatment for 24h to differentiate into adherent macrophages, followed by medium change and 24h rest |
M1: LPS (100 ng/mL) + IFNγ (20 ng/mL); |
M1: CD86, HLA-DR, TNF; |
|
RAW264.7 |
Mouse macrophage cell line, requires no pre-differentiation, good adherence, suitable for beginners |
Plate cells in log-phase growth at appropriate density and incubate overnight |
M1: LPS (100 ng/mL) + IFNγ (20 ng/mL); |
M1: CD86, iNOS, TNF; |
|
BMDM |
Primary macrophages isolated from mouse bone marrow, most closely mimic in vivo function, but require longer culture time |
Differentiate bone marrow cells with M-CSF (20 ng/mL) for 7 days, change medium one day before experiment |
M1: LPS (100 ng/mL) + IFNγ (20 ng/mL); |
Same as RAW264.7. Note strain differences (e.g., C57BL/6 vs. BALB/c polarization efficiency). |
3.2 Key Considerations for Polarization Experiments
Experimental success hinges on meticulous attention to detail. Key points are outlined below from three perspectives—cells, reagents, and operations—covering common pitfalls:
1). Cell-Related: Ensuring Polarization Viability from the Start
2). Reagent-Related: Ensuring Precision of Polarizing Signals
3). Operational Aspects: Minimizing Human Error
These considerations should be adapted flexibly based on cell type. For instance, THP-1 cells require two PBS washes post-PMA differentiation to remove residual PMA, whereas RAW264.7 cells simply require a medium change. Subsequent detection assays must employ markers specific to each cell type to ensure reliable results.
References
[1] Mass E, Nimmerjahn F, Kierdorf K, Schlitzer A. Tissue-specific macrophages: how they develop and choreograph tissue biology. Nat Rev Immunol. 2023 Sep;23(9):563-579.
[2] Chen S, Saeed AFUH, Liu Q, Jiang Q, Xu H, Xiao GG, Rao L, Duo Y. Macrophages in immunoregulation and therapeutics. Signal Transduct Target Ther. 2023 May 22;8(1):207.
[3] Li D, Zhang T, Guo Y, Bi C, Liu M, Wang G. Biological impact and therapeutic implication of tumor-associated macrophages in hepatocellular carcinoma. Cell Death Dis. 2024 Jul 12;15(7):498.
[4] Chen Y, Song Y, Du W, Gong L, Chang H, Zou Z. Tumor-associated macrophages: an accomplice in solid tumor progression. J Biomed Sci. 2019 Oct 20;26(1):78.
[5] Jin R, Neufeld L, McGaha TL. Linking macrophage metabolism to function in the tumor microenvironment. Nat Cancer. 2025 Feb;6(2):239-252.
abinScience Products for Macrophage Polarization
4.1 Recombinant Proteins
4.2 Flow Cytometry Antibodies
4.3 Other Related Antibodies
| Marker | Reactivity | Product Name | Applications | Catalog No. |
|---|---|---|---|---|
| iNOS | Human | Anti-NOS2 Polyclonal Antibody | ELISA, IHC, WB | HW388014 |
| Arg-1 | Human, Mouse, Rat | Anti-ARG1/Arginase-1 Polyclonal Antibody | ELISA, IHC, WB | HY339024 |
| Human, Mouse, Rat, Pig, etc | Anti-ARG1 Polyclonal Antibody | ELISA, IHC, WB | HY339014 | |
| CD11b | Human | Anti-CD11b/ITGAM Polyclonal Antibody | ELISA, IHC, WB | HY474014 |
| Mouse | Anti-Mouse CD11b/ITGAM Polyclonal Antibody | ELISA, IHC, WB | MY474014 | |
| CD86 | Human | Anti-CD86 Polyclonal Antibody | ELISA, IHC, WB | HW776014 |
| Mouse | Anti-Mouse CD86/B7-2 Polyclonal Antibody | ELISA, IHC, WB | MW776014 | |
| CD206 | Human, Mouse, Rat | Anti-CD206/MRC1 Polyclonal Antibody | ELISA, IHC, WB | HB976014 |
| IL-1 | Human, Cercocebus atys, Macaca fascicularis, etc | Anti-IL1B/IL1F2 Polyclonal Antibody | ELISA, IHC, WB | HF943014 |
| IL-6 | Human | Anti-Human IL6 Antibody | ELISA, FCM, WB, IHC, IF | HY328033 |
| Mouse | Anti-Mouse IL6 Monoclonal Antibody | ELISA, IHC, WB | MY328085 | |
| IL-10 | Human | Anti-Human IL10 Antibody | ELISA, WB, IHC, FCM | HB997023 |
| TNF-α | Human, Dog, Cat, Pig, etc | Anti-TNFa/TNF-alpha Polyclonal Antibody | ELISA, IHC, WB | HF879014 |
| Mouse, Rat, Peromyscus leucopus | Anti-TNFa/TNF-alpha Polyclonal Antibody | ELISA, IHC, WB | MF879014 | |
| Danio rerio | Anti-Zebrafish TNFa Polyclonal Antibody | ELISA, IHC, WB | ZA439014 | |
| TGF‐β | Human, Mouse, Dog, Rat, etc | Anti-TGFB1/TGF-beta-1 Polyclonal Antibody | ELISA, IHC, WB | HF977014 |
| Mouse | Anti-Mouse TGFB1/TGF-beta-1 Polyclonal Antibody | ELISA, IHC, WB | MF977014 |
abinScience offers a comprehensive range of antibodies and recombinant proteins for macrophage research, widely applicable in experiments such as ELISA, WB, IHC, FCM, etc. These products support researchers in in-depth studies of macrophage polarization, activation, protein expression, functions, and phenotypes. For more macrophage-related products, visit the abinScience official website: www.abinscience.com.
+86-027-65523339
중국 우한시 심둔사로 666번지 C동, 우한, 430206

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский