Marburg Virus Research in 2026: Outbreaks, Virulence Mechanisms & Vaccine Pipeline
Global Epidemiology with 2025 Ethiopia Outbreak Update
Marburg virus disease (MVD) is a severe hemorrhagic fever caused by Marburg marburgvirus (MARV), with case fatality rates historically ranging from 24–88% depending on outbreak management.[1] The virus is zoonotic, with Egyptian rousette bats (Rousettus aegyptiacus) as the established reservoir.
UPDATE (January 2026): Ethiopia’s first recorded Marburg outbreak, declared in November 2025, has shown strong containment progress. As of early January 2026, 14 laboratory-confirmed cases (including 9 deaths) have been reported, with no new confirmed cases detected for over 21 days.[2][3] Authorities indicate the outbreak may be declared over after a full 42-day period without new cases.
Contact tracing and surveillance continue, supported by international partners. No cases related to this outbreak have been reported outside Ethiopia.

Fig. 1. Map of Ethiopia Highlighting Affected Regions in 2025 Marburg Outbreak (Al Jazeera)
Pathogen Structure and Key Virulence Factors
Marburg virus is a filamentous, negative-sense single-stranded RNA virus (~19 kb genome) encoding seven structural proteins. Virions are enveloped with surface glycoprotein (GP) spikes.
Major virulence factors enable immune evasion, cell entry, and systemic effects:
| Target | Function and Immunological Features | Application Scenarios |
|---|---|---|
| Glycoprotein (GP) | Mediates attachment and entry (via NPC1 receptor); primary target for neutralizing antibodies; induces immune shielding | Neutralization assays; vaccine antigen design; pseudovirus studies |
| VP40 | Matrix protein; drives virion assembly and budding; forms virus-like particles (VLPs) | VLP platforms; structural biology; budding mechanism research |
| VP35 | Interferon antagonist; blocks RIG-I/MDA5 and PKR pathways | Innate immunity evasion studies; antiviral screening |
| VP24 | Secondary interferon antagonist; inhibits STAT1 signaling | Interferon pathway research; therapeutic target validation |
| Nucleoprotein (NP) | Core ribonucleoprotein component; protects viral RNA; immunogenic | Diagnostic antigen; replication complex studies |
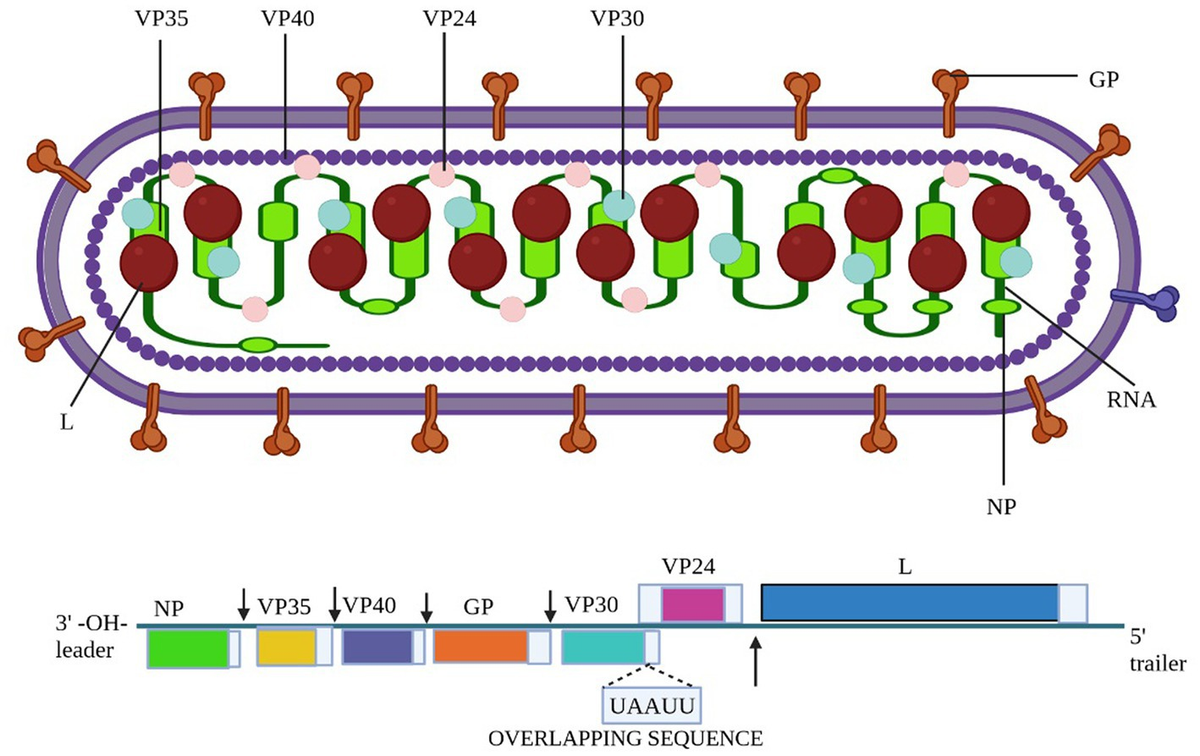
Fig. 2. Schematic of Marburg Virus Virion Structure and Genome Organization (Frontiers in Microbiology, Open Access)
Prevention Challenges and Vaccine Pipeline
- Diagnosis: Early symptoms overlap with other VHFs; RT-PCR on blood is gold standard early in illness; IgM/IgG serology useful in later phases.[1]
- Treatment: Supportive care (rehydration, symptom management) is the mainstay and improves outcomes; no approved antivirals or monoclonal therapies currently licensed, though candidates (e.g., remdesivir, mAb cocktails) have been evaluated in past outbreaks.
- Vaccines: No licensed vaccines as of 2026. Several candidates (chimpanzee adenovirus vectors, VSV platforms, VLPs) have shown promise in preclinical and Phase I/II trials; ring vaccination strategies are prepared for future outbreaks.[4]
Fig. 3. Marburg Virus Transmission Cycle from Bat Reservoir to Human Spread
Research Application Scenarios
Current priorities in Marburg research include:
- Neutralizing Antibody Studies: GP-based assays and pseudovirus neutralization.
- Immune Evasion: VP35 and VP24 interferon antagonist models.
- VLP Platforms: VP40 + GP for vaccine immunogenicity testing.
- Diagnostics: NP and GP antigens for rapid serological tests.
Fig. 4. Comprehensive Insights into Marburg Virus Proteins and Immunology (Springer/Open Access)
Marburg Virus Product Catalog
High-purity recombinant proteins and antibodies for Marburg virus research:
| Type | Catalog No. | Product Name |
|---|---|---|
| Protein | VK735012 | Recombinant MARV VP40/Marburg VP40 Protein, N-His |
| VK504012 | Recombinant MARV VP35/Marburg VP35 Protein, N-His | |
| VK538012 | Recombinant MARV VP30/Minor nucleoprotein VP30 Protein, N-His | |
| VK668012 | Recombinant MARV VP24/Marburg VP24 Protein, N-His | |
| VK660012 | Recombinant MARV NP/Nucleoprotein Protein, N-His | |
| VK669022 | Recombinant MARV GP2/Envelope glycoprotein 2 Protein, N-His | |
| VK669031 | Recombinant MARV GP2/Envelope glycoprotein 2 Protein, C-Strep | |
| VK669012 | Recombinant MARV GP1/Envelope glycoprotein 1 Protein, N-His | |
| VK669021 | Recombinant MARV GP1/Envelope glycoprotein 1 Protein, C-His | |
| VK669011 | Recombinant MARV GP/Envelope glycoprotein Protein, C-His | |
| Antibody | VK735014 | Anti-MARV VP40/Marburg VP40 Polyclonal Antibody |
| VK504013 | Anti-MARV VP35/Marburg VP35 Antibody (SAA1444) | |
| VK660013 | Anti-MARV NP/Nucleoprotein Antibody (SAA1443) | |
| VK669014 | Anti-MARV GP1/Envelope glycoprotein 1 Polyclonal Antibody | |
| VK669023 | Anti-MARV GP/Envelope glycoprotein Antibody (MR78) | |
| VK669013 | Anti-MARV GP/Envelope glycoprotein Antibody (MR191) |
About abinScience
abinScience is a biotechnology company headquartered in France, dedicated to infectious disease research and public health tools. We provide high-quality research-grade reagents to support global efforts against emerging filoviruses like Marburg—from surveillance to mechanism studies and candidate evaluation.
Empowering Bioscience Discovery
References
- World Health Organization. Marburg virus disease fact sheet. Link.
- CDC. Marburg Outbreak in Ethiopia: Current Situation (updated December 2025). Link.
- Outbreak News Today. Ethiopia Marburg virus outbreak update (January 2026). Link.
- Multiple sources including WHO and Sabin Vaccine Institute on investigational Marburg vaccine candidates.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский


