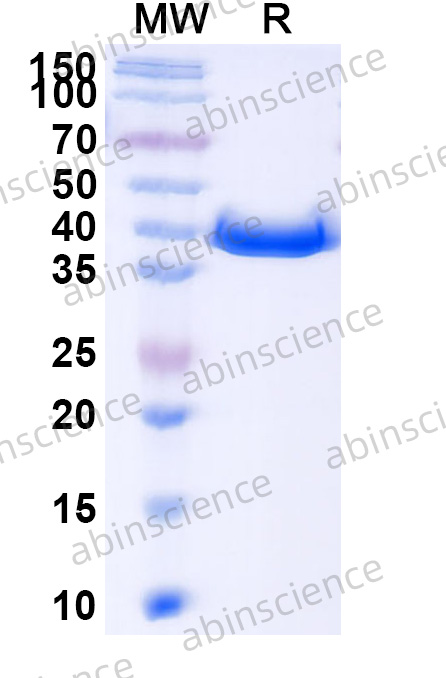
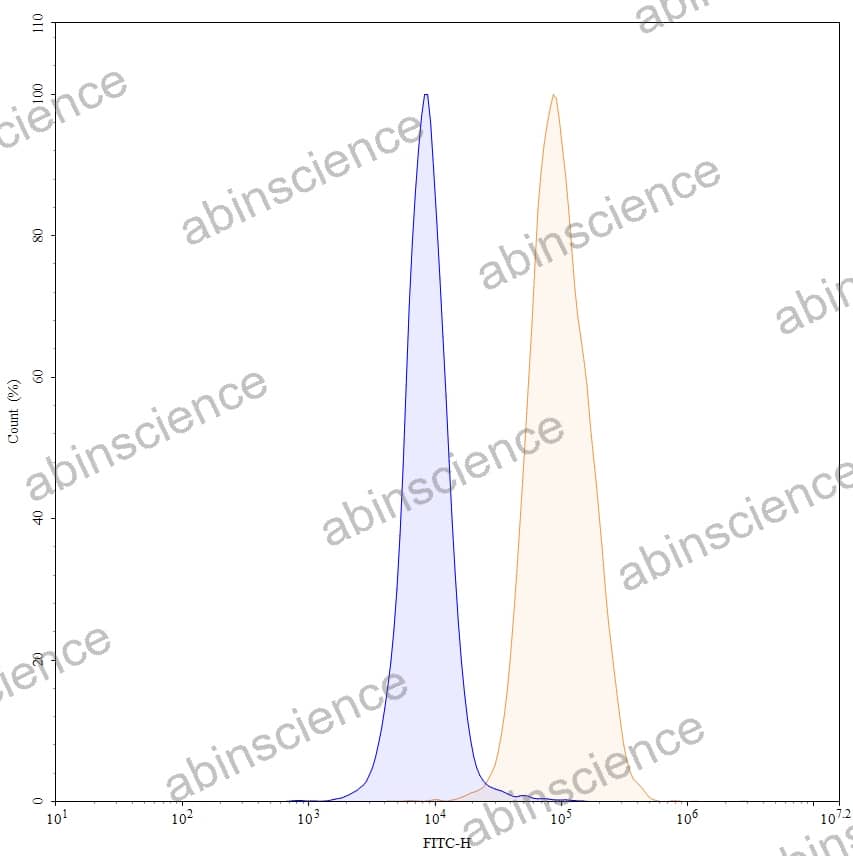
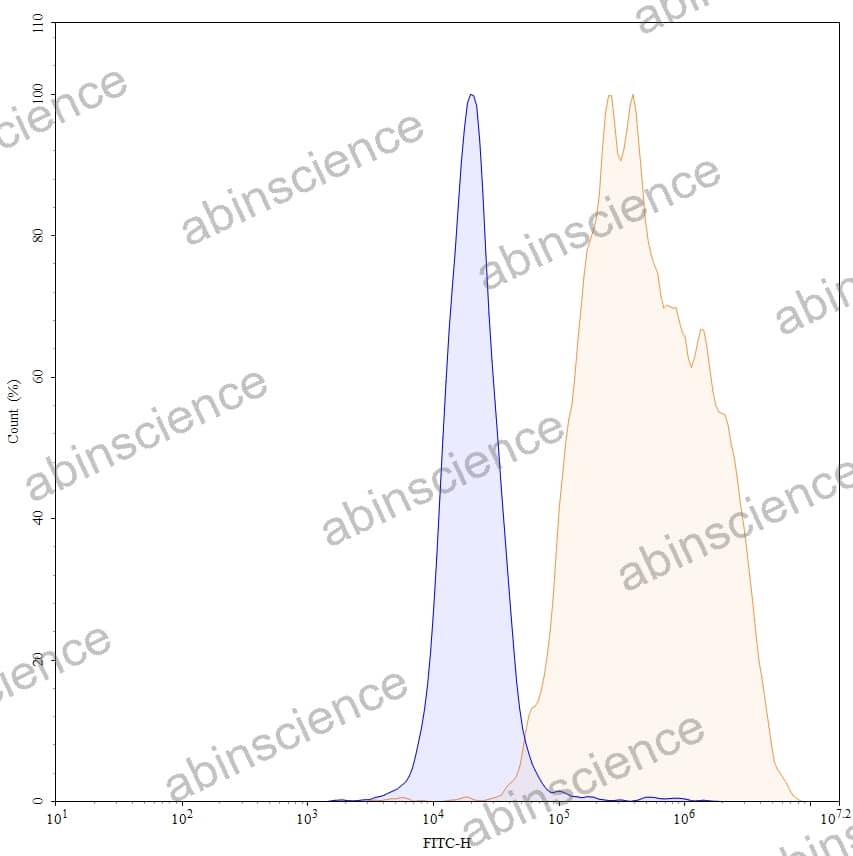
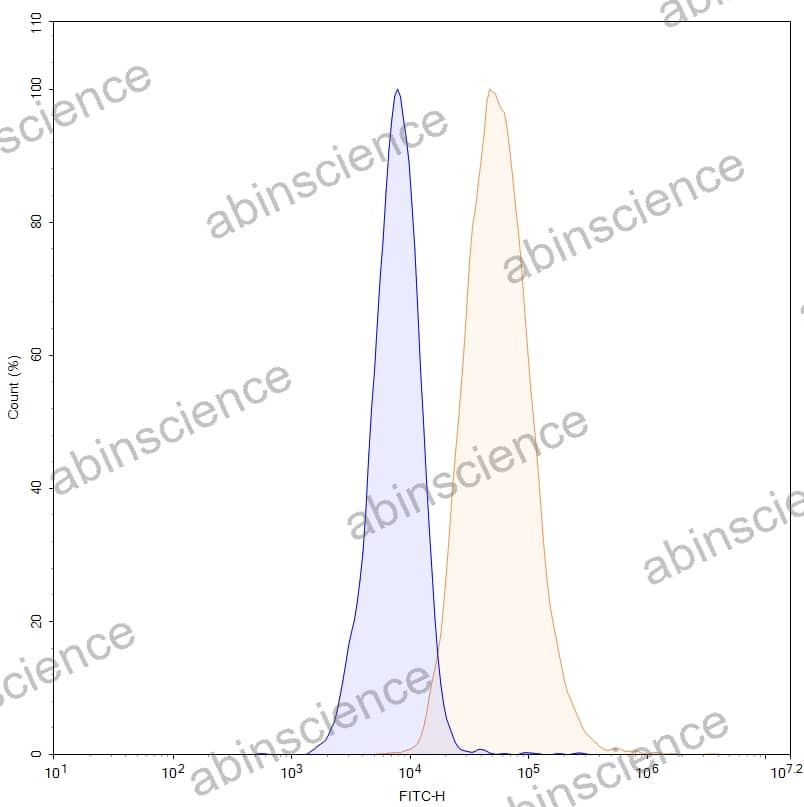
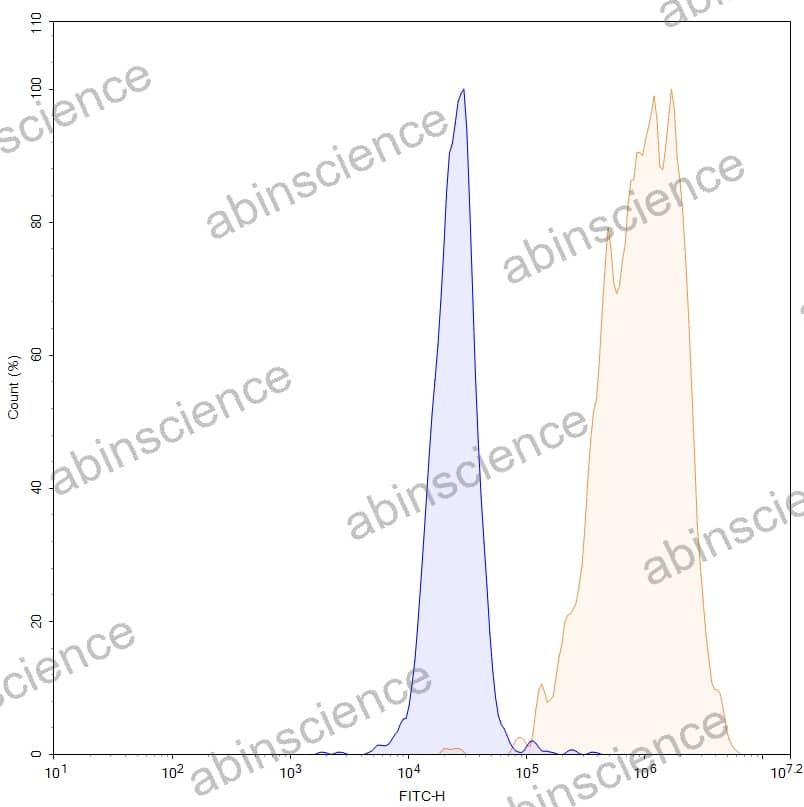
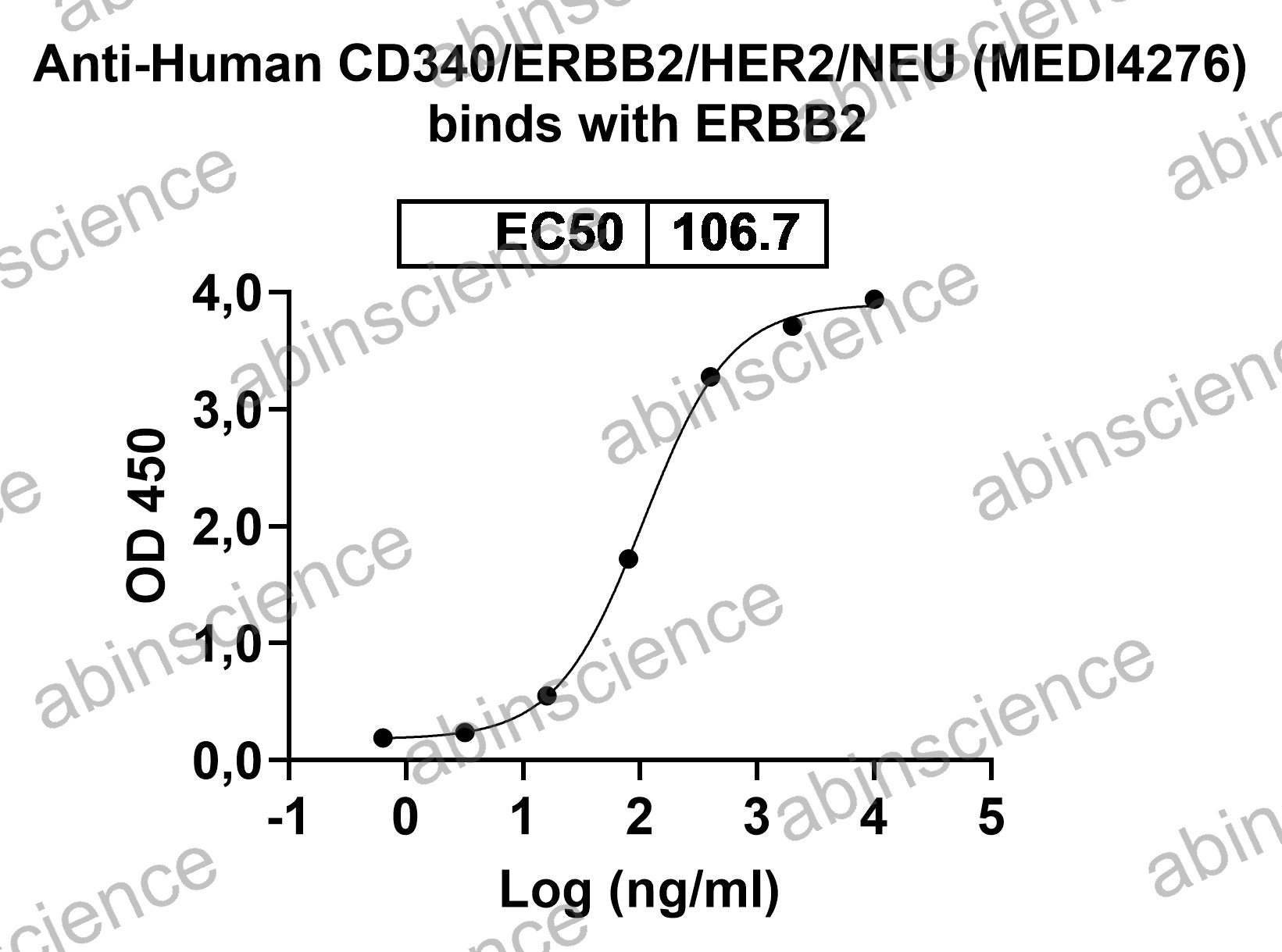
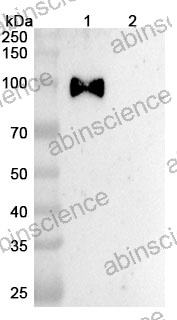
Lung Cancer — Disease Background
Lung cancer remains the most lethal malignancy globally, causing an estimated 1.8 million deaths annually. Non-small cell lung cancer (NSCLC) accounts for approximately 85% of cases and is classified into adenocarcinoma, squamous cell carcinoma, and large cell carcinoma subtypes, each with distinct molecular drivers and therapeutic vulnerabilities.
The therapeutic landscape has been revolutionized by targeted kinase inhibitors (EGFR, ALK, ROS1, RET, MET, KRAS G12C) and immune checkpoint blockade (anti-PD-1/PD-L1). Comprehensive genomic profiling and PD-L1 tumor proportion score (TPS) testing now guide first-line treatment decisions, making biomarker-validated research reagents essential for translational oncology.
Research Use Only (RUO)
Not intended for diagnostic or therapeutic procedures.

Fig. 1 Major pathogenic pathways in Lung Cancer
Key Research Targets
EGFR (Epidermal Growth Factor Receptor) — EGFR activating mutations (exon 19 deletions, L858R) drive oncogenic signaling through RAS-MAPK and PI3K-AKT pathways in approximately 15-30% of NSCLC adenocarcinomas. Anti-EGFR antibodies are essential for IHC-based expression profiling, mutation-specific detection, and monitoring therapeutic resistance mechanisms including T790M and C797S mutations.
ALK (Anaplastic Lymphoma Kinase) — EML4-ALK fusion rearrangements occur in 3-7% of NSCLC cases and define a distinct molecular subtype responsive to ALK tyrosine kinase inhibitors (crizotinib, alectinib, lorlatinib). Anti-ALK antibodies enable IHC screening as a companion diagnostic surrogate and support research into resistance mechanisms and bypass signaling pathways.
PD-L1 (Programmed Death-Ligand 1, CD274) — PD-L1 expression on tumor cells and tumor-infiltrating immune cells determines eligibility for immune checkpoint inhibitor therapy. Anti-PD-L1 antibodies and recombinant PD-L1 proteins support TPS scoring validation, PD-1/PD-L1 binding assays, and functional immune checkpoint blockade studies critical for immunotherapy development.
KRAS G12C — KRAS G12C mutations occur in approximately 13% of NSCLC adenocarcinomas and represent the most recently druggable RAS pathway alteration. Anti-KRAS antibodies (including mutation-specific clones) enable immunohistochemical detection of mutant KRAS and downstream MAPK pathway activation monitoring in preclinical and translational studies.
References
1. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553(7689):446-454. DOI
2. Reck M, Rodríguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N Engl J Med. 2016;375(19):1823-1833. DOI
3. The Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature. 2014;511(7511):543-550. DOI
4. Skoulidis F, Li BT, Dy GK, et al. Sotorasib for lung cancers with KRAS p.G12C mutation. N Engl J Med. 2021;384(25):2371-2381. DOI
5. Mok TS, Wu Y-L, Thongprasert S, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361(10):947-957. DOI

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский