Systemic lupus erythematosus (SLE) is a highly heterogeneous chronic autoimmune disease characterized by breakdown of B-cell tolerance, massive production of autoantibodies against nuclear antigens, and subsequent systemic inflammation and organ damage. During autoantibody production, the extrafollicular (EF) B-cell differentiation pathway is the primary source of antibody-secreting cells (ASCs) and is closely associated with disease activity. Among these, a B-cell subset with low CD21 expression (CD21lo) and high CD11c expression accumulates in aging, infection, and autoimmune diseases and has been identified as a key precursor of extrafollicular ASCs; however, the developmental trajectory and receptor regulatory mechanisms of this subset remain unclear.
Toll-like receptor 7 (TLR7), an innate immune receptor that recognizes single-stranded RNA, plays a crucial role in SLE pathogenesis by promoting B-cell tolerance escape and autoantibody production. In contrast, complement receptor 2 (CD21), a co-receptor of the B-cell receptor (BCR), normally enhances B-cell activation, yet it is paradoxically downregulated in lupus-associated B-cell subsets. This contradictory phenomenon suggests a more complex role for CD21 in autoimmunity. To address these questions, the research team established a TLR7-driven adoptive transfer lupus model to systematically investigate the regulatory role and molecular mechanisms of CD21 in the extrafollicular differentiation of autoreactive B cells.
The 564Igi Adoptive Transfer Model Is TLR7-Dependent and Supports Extrafollicular Differentiation of Autoreactive B Cells
The research team adoptively transferred splenic B cells from wild-type (WT) and TLR7-knockout (Tlr7-KO) mice at a 1:1 ratio into 564Igi transgenic recipient mice, which provide an autoimmune microenvironment rich in nuclear antigens and T-cell help. Seven days post-transfer, mature B cells, germinal center (GC) B cells, and non-GC B cells derived from Tlr7-KO donors were significantly reduced. Notably, ASCs and DN2-like cells (CD11c+CD21-) decreased by 24-fold and 9.7-fold, respectively, confirming that both B-cell tolerance breakdown and germinal center development depend on TLR7 signaling. Blocking GC formation using Bcl6 conditional knockout mice did not affect ASC numbers. Combined with kinetic analysis showing WT donor B-cell expansion starting on day 4 and ASC accumulation in the splenic red pulp from day 5, the study clearly demonstrated that autoreactive B cells in this model primarily differentiate into ASCs via the extrafollicular pathway, driven by TLR7 signaling.
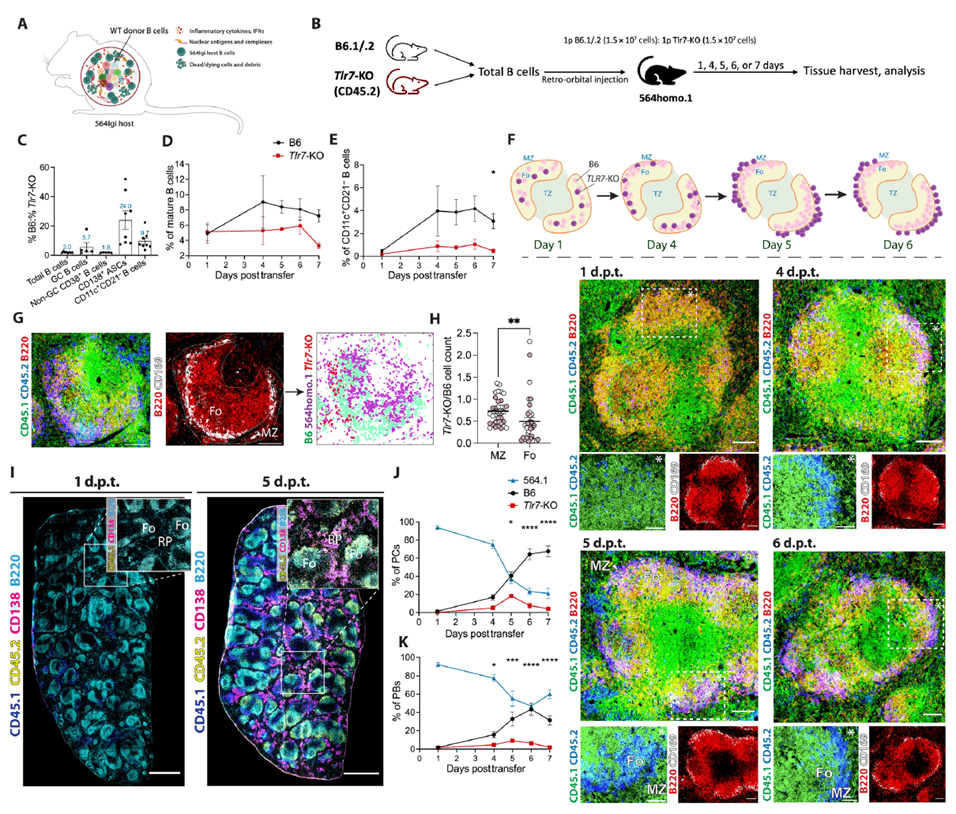
Figure 1. The 564Igi adoptive transfer model is TLR7-dependent and supports extrafollicular (EF) differentiation of autoreactive B cells
Naïve Autoreactive B Cells Downregulate CD21 Through a Fixed Division Program, Enhancing TLR7 Dependency
To dissect the relationship between B-cell proliferation and differentiation, donor B cells were labeled with CellTrace Violet (CTV). The results showed that CD21 expression gradually decreases during donor B-cell proliferation, with most ASCs requiring at least seven divisions. Phenotypic clustering divided donor B cells into marginal zone-like, follicular-like, and DN2-like subsets. The CD21loCD23- subset predominantly consisted of terminally divided cells, whereas in Tlr7-KO mice this subset contained significantly more resting cells and failed to complete terminal divisions. Late-division cells also upregulated the ASC-associated transcription factors BLIMP1 and IRF4, indicating a pre-programmed division-linked differentiation pathway for autoreactive ASCs. CD21 downregulation is tightly linked to TLR7 signaling; TLR7 deficiency impedes continuous cell-cycle progression and differentiation.
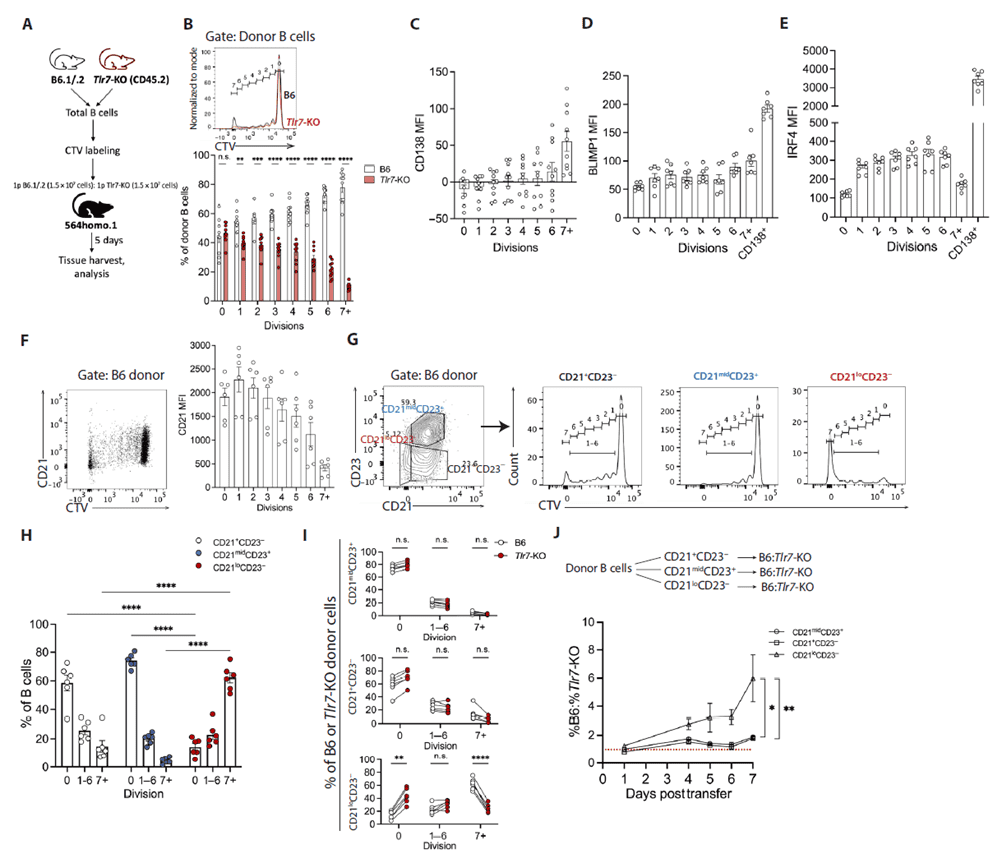
Figure 2. Naïve autoreactive B cells downregulate CD21 and establish TLR7 sensitivity during a fixed division-differentiation program
CD21lo Cells Originate from CD21+ Cells and Serve as the Dominant Precursor of Extrafollicular ASCs
Using CD21-Cre×Rosa26Lox-Stop-Lox-tdTomato fate-mapping mice, nearly all donor-derived CD21loCD23- cells in 564Igi recipients at day 7 were tdTomato+, confirming their origin from initially CD21+ B cells rather than a pre-existing CD21lo pool. Serial adoptive transfer experiments showed that sorted CD21lo cells preferentially differentiated into CD138+ ASCs in secondary recipients, whereas follicular B cells primarily formed GC B cells. In vitro stimulation with TLR7 agonist and cytokines further demonstrated that CD21lo cells efficiently differentiate into plasmablast-like cells and produce large amounts of autoantibodies against cytoplasmic components, establishing CD21lo cells as direct precursors of extrafollicular ASCs with strong autoantibody-secreting potential.
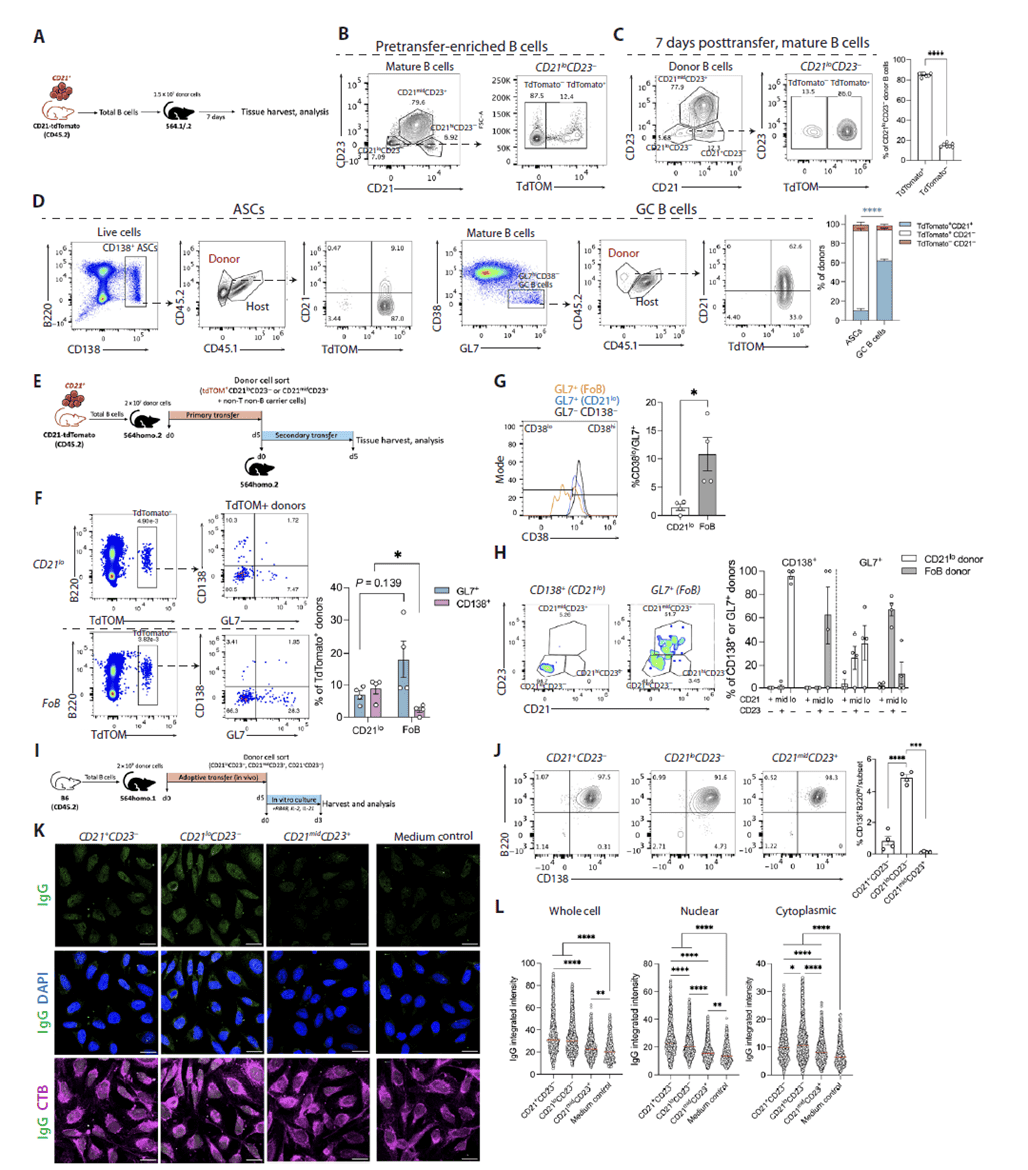
Figure 3. CD21lo cells derive from CD21+ cells and are poised for ASC and autoantibody production
Naïve Follicular B Cells Are the Primary Source of CD21lo Precursors and Extrafollicular ASCs
Purified naïve follicular B cells (CD21midCD23+), marginal zone B cells, and transitional T1 B cells were separately transferred into 564Igi recipients. Only naïve follicular B cells robustly generated CD21loCD23- cells by day 5.5 post-transfer, with most cells undergoing ≥7 divisions expressing CD138. Terminally divided cells were markedly enriched for the CD21loCD23- subset, and a single mouse's starting follicular B cells contributed ~20% of total ASCs. Marginal zone B cells produced few ASCs while largely retaining their original phenotype, and T1 B cells failed to differentiate efficiently into ASCs. These results confirm that naïve follicular B cells are the dominant source of both CD21lo precursors and early autoreactive ASCs, requiring passage through the CD21lo intermediate state for extrafollicular differentiation.
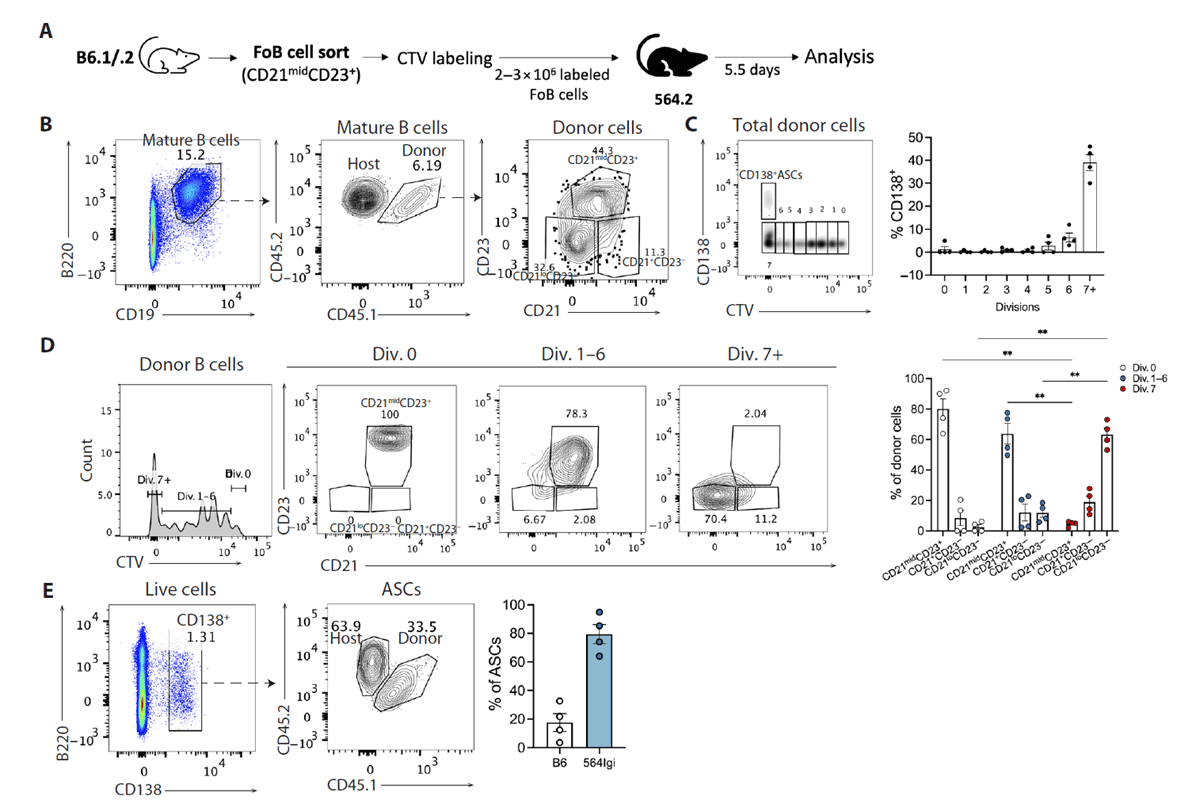
Figure 4. CD21+ ASC precursor B cells originate from naïve follicular B cells (FoB)
BCR Repertoire Sequencing Reveals the Evolutionary Trajectory of Follicular B Cells Toward Autoreactivity
To define the clonal evolution from naïve follicular B cells (FoB) to autoreactive cells, bulk heavy- and light-chain BCR sequencing was performed on donor subsets 6.5 days post-transfer. Hill diversity analysis showed the lowest receptor diversity and most pronounced clonal expansion in CD21lo cells, while ASCs exhibited higher clonal dominance despite greater richness, indicating that CD21lo cells are transitional effector precursors under strict clonal selection in ASCs. κ/λ light-chain ratio analysis and shared heavy-chain gene usage (e.g., Ighv1–82, Ighv1–80) further supported early selection in CD21lo cells. Phylogenetic analysis revealed that the most expanded ASC clones belonged to the same evolutionary branch as the 564Igi autoreactive heavy chain, with transitional BCR sequences in CD21lo cells bridging toward ASCs, confirming that naïve FoB progressively acquire autoreactivity through clonal selection and receptor editing, with CD21lo cells representing a critical intermediate stage.
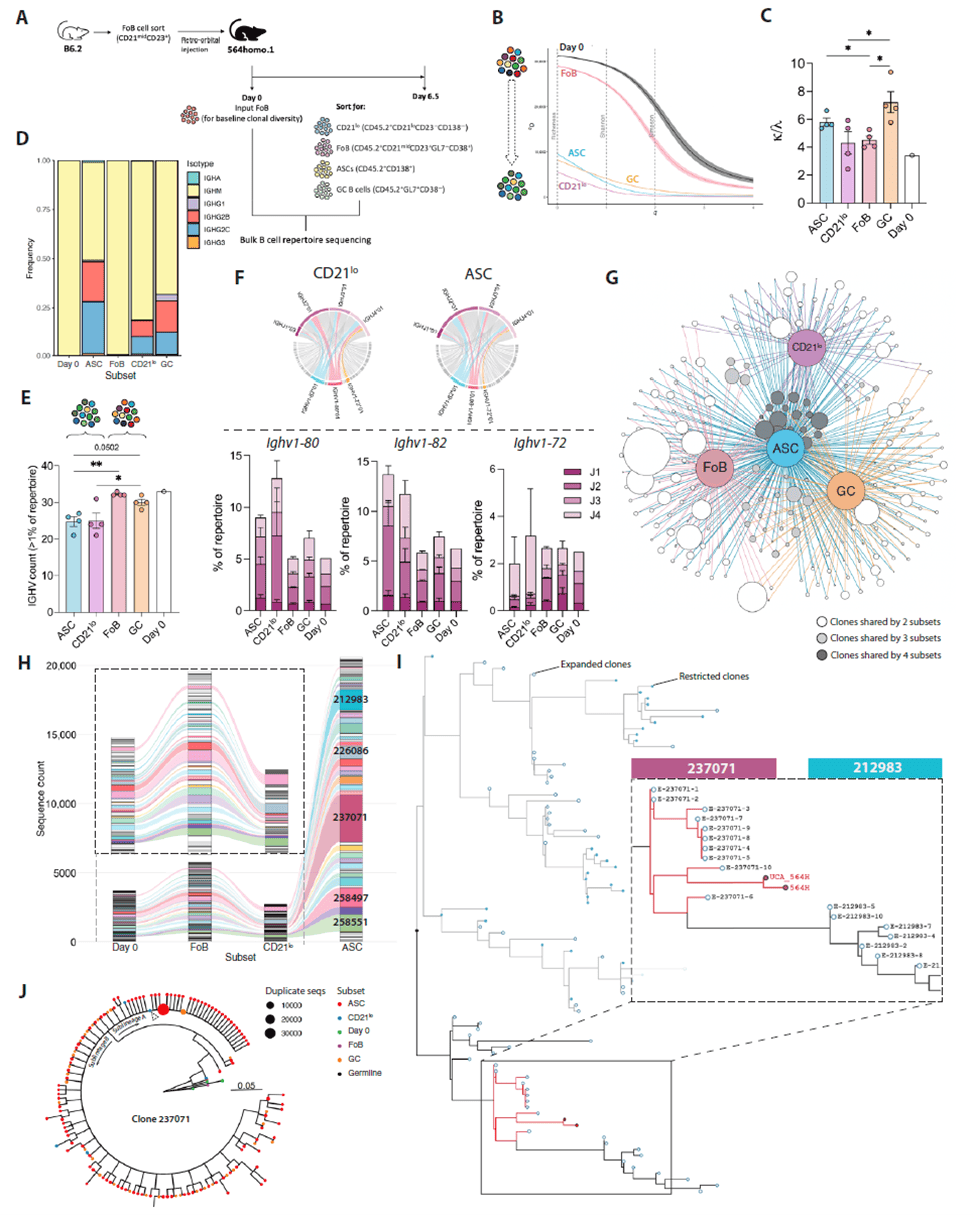
Figure 5. BCR repertoire sequencing reveals the developmental trajectory of follicular B cells toward autoreactivity
Functional Blockade of CD21 Prevents Its Downregulation and Impedes B-Cell Proliferation and ASC Differentiation
Using CD21-knockout and human CD21-transgenic mouse models, mCd21-KO donor B cells showed increased early division but failed to reach terminal divisions, indicating that CD21 loss does not affect initial activation but blocks proliferation progression. Blocking the C3d-binding site on hCD21+ B cells with a monoclonal antibody reduced terminal division, ASC generation, and red pulp ASC accumulation while preserving the CD21+ population and decreasing CD21loCD23- cells. In vitro and in vivo experiments confirmed that ligand (C3d/iC3b) binding triggers rapid CD21 internalization, which is completely prevented by antibody blockade or C3 deficiency. Thus, ligand engagement drives CD21 downregulation, and functional blockade inhibits this process and subsequent proliferation, ultimately reducing ASC formation.
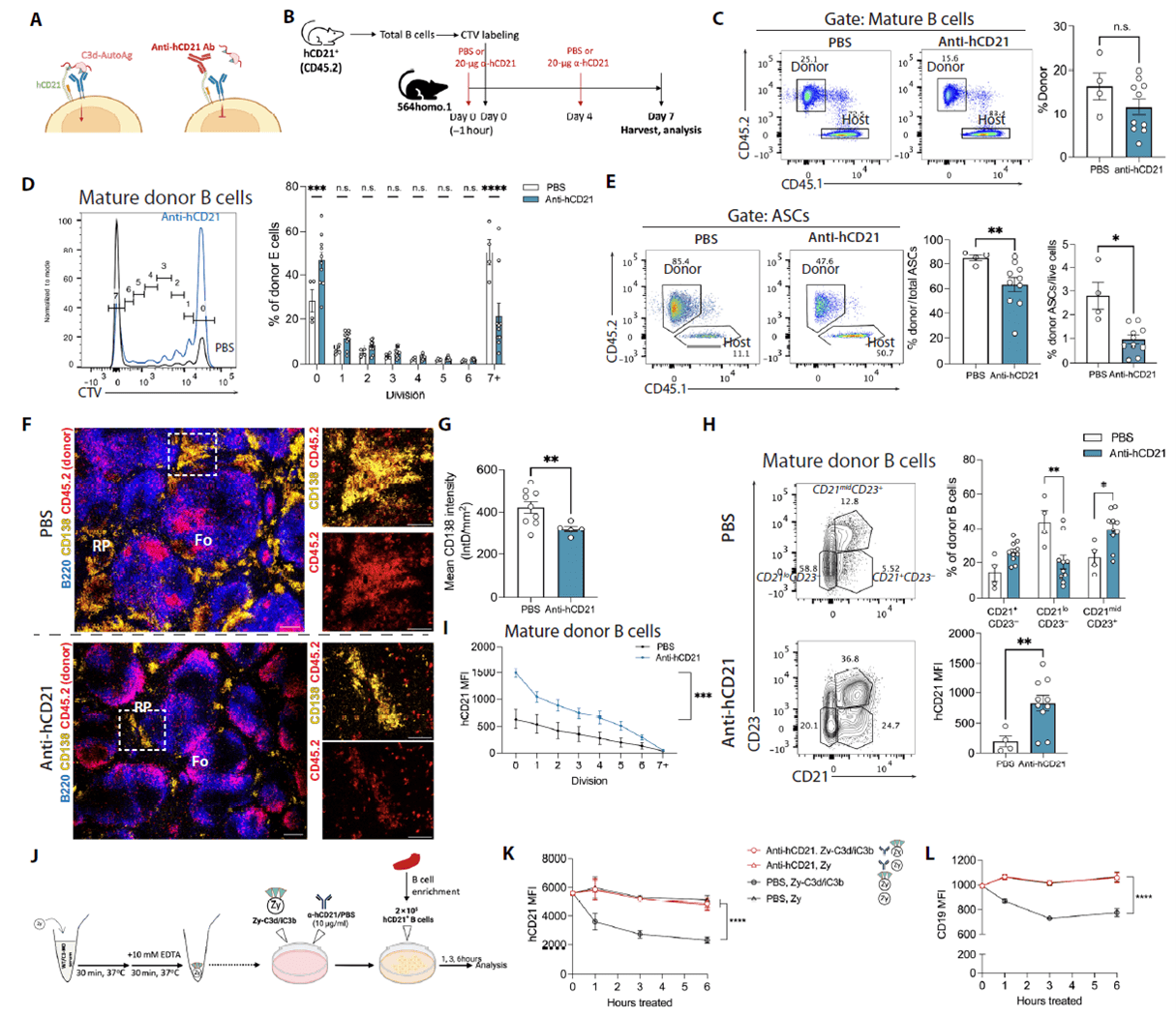
Figure 6. CD21 downregulation during proliferation is suppressed by functional receptor blockade
CD21 Downregulation Requires Sustained Ligand Engagement, Occurs Prior to Proliferation, Is Partially Reversible, and Determines Differentiation Fate
Serial transfer of CD21-tdTomato reporter cells showed that some CD21lo cells formed in primary recipients partially restored CD21 expression in secondary 564Igi hosts, demonstrating that the CD21lo state is not irreversible but depends on continuous ligand presence in the microenvironment. Delayed in vitro blockade experiments revealed that CD21 downregulation stops immediately upon antibody addition and correlates with ligand exposure duration. In vivo delayed blockade starting day 3 post-transfer maintained CD21 levels at the day-3 state and produced bimodal CD21 expression in terminally divided cells. Notably, donor B cells at day 3 were mostly undivided, whereas by day 7 both blocked and unblocked groups reached terminal divisions. These results establish that CD21 downregulation precedes proliferation, represents an early activation event, and full downregulation serves as a critical fate checkpoint for autoreactive B-cell differentiation into ASCs; incomplete downregulation allows cells to revert and alter their differentiation trajectory.
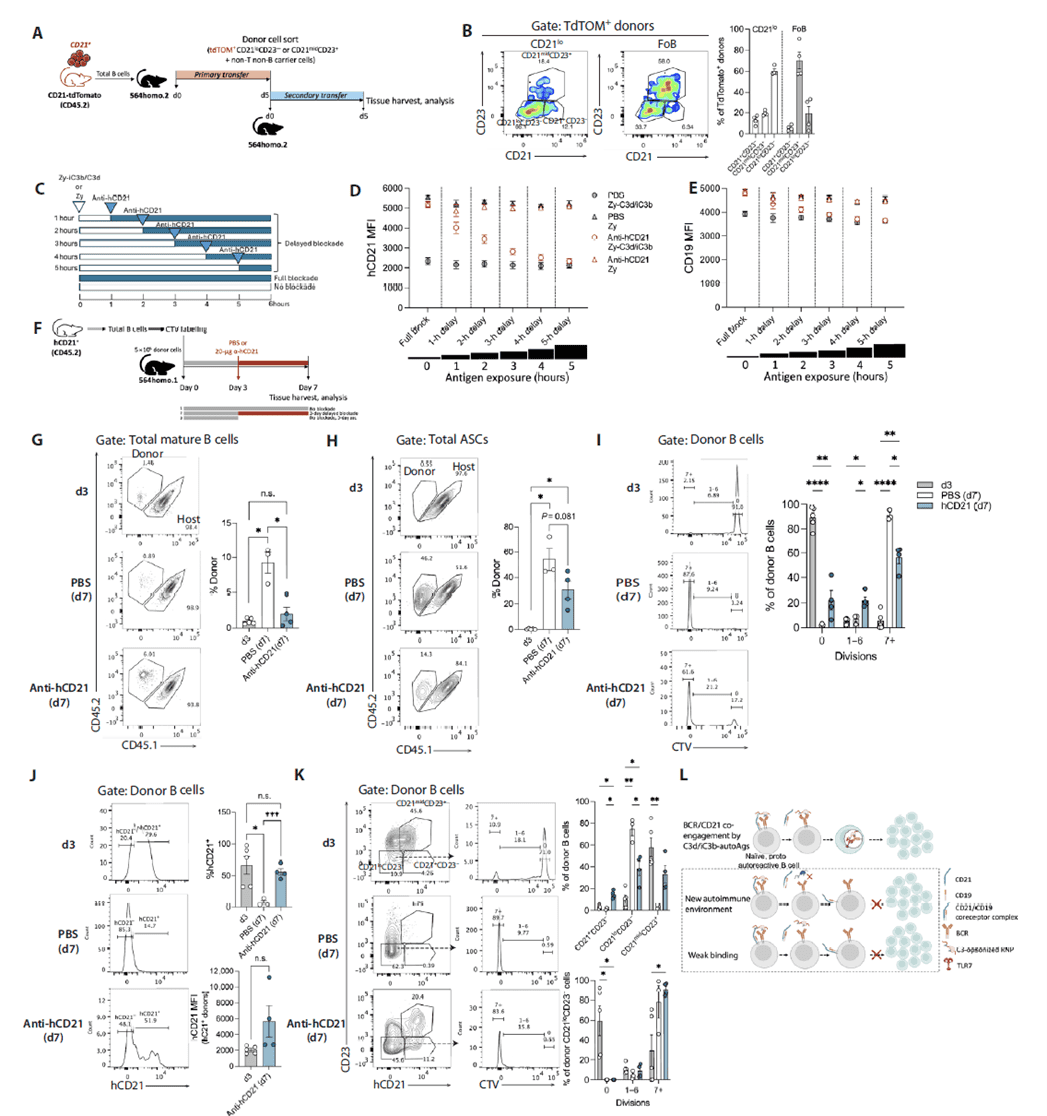
Figure 7. CD21 downregulation requires sustained ligand binding and occurs before B-cell proliferation
Through integrated analyses of cell fate mapping, clonal evolution, and molecular regulation, this study comprehensively elucidates the mechanism of extrafollicular differentiation of autoreactive B cells. It clarifies the origin, differentiation potential, and regulatory mechanisms of CD21lo cells and demonstrates the therapeutic value of targeting CD21 and TLR7—acting at the initiation and maintenance phases of differentiation, respectively. Dual targeting offers a more precise strategy to suppress autoreactive B-cell activation, providing a novel "dual-target" intervention approach for SLE and mechanistic insight into similar B-cell subsets in infection and aging.
abinScience was founded in Strasbourg, France, leveraging the region's outstanding research and innovation ecosystem to focus on the development and production of high-quality life science reagents. Guided by the vision of "Empowering Bioscience Discovery," abinScience is committed to providing efficient and reliable experimental solutions to researchers worldwide, accelerating cutting-edge life science research.
Below is a list of related protein and antibody products offered by abinScience:
| Target |
Type |
Catalog No. |
Product name |
| CD21 |
| Protein |
HB939012 |
Recombinant Human CD21/CR2 Protein, N-GST |
| MB939011 |
Recombinant Mouse CD21/CR2 & CR1L Protein, C-His |
| HB939011 |
Recombinant Human CD21/CR2 Protein, C-His |
| HB939022 |
Recombinant Human CD21/CR2 Protein, N-His |
| Antibody |
HB939107 |
Anti-Human CD21/CR2 Antibody (HB5) |
| HB939207 |
Anti-Human CD21/CR2 Antibody (SAA1459) |
| HB939117 |
Anti-Human CD21/CR2 Antibody (HB5), FITC |
| HB939217 |
Anti-Human CD21/CR2 Antibody (SAA1459), FITC |
| HB939137 |
Anti-Human CD21/CR2 Antibody (HB5), APC |
| HB939237 |
Anti-Human CD21/CR2 Antibody (SAA1459), APC |
| HB939147 |
Anti-Human CD21/CR2 Antibody (HB5), PerCP |
| HB939247 |
Anti-Human CD21/CR2 Antibody (SAA1459), PerCP |
| HB939127 |
Anti-Human CD21/CR2 Antibody (HB5), PE |
| HB939227 |
Anti-Human CD21/CR2 Antibody (SAA1459), PE |
| HB939307 |
Anti-Human CD21/CR2 Antibody (Ab652) |
| HB939317 |
Anti-Human CD21/CR2 Antibody (Ab652), FITC |
| HB939327 |
Anti-Human CD21/CR2 Antibody (Ab652), PE |
| HB939337 |
Anti-Human CD21/CR2 Antibody (Ab652), APC |
| HB939347 |
Anti-Human CD21/CR2 Antibody (Ab652), PerCP |
| TLR7 |
Protein |
HV024012 |
Recombinant Human TLR7 Protein, N-His |
| Antibody |
HV024014 |
Anti-TLR7 Polyclonal Antibody |
| HV024013 |
Anti-Human TLR7 Antibody (SAA1718) |
| BCR |
Protein |
HY023012 |
Recombinant Human BCR Protein, N-His |
| HP839012 |
Recombinant Human BCR/ABL Protein, N-His-SUMO |
| Antibody |
HY023014 |
Anti-BCR Polyclonal Antibody |
| HP839014 |
Anti-Human BCR/ABL Polyclonal Antibody |
| HY023013 |
Anti-Human BCR Antibody (SAA1570) |
| PTX19569 |
Anti-BCR Polyclonal Antibody |
| PTX20541 |
Anti-Human BCR/ABL Polyclonal Antibody |
| CD19 |
Protein |
HB996012 |
Recombinant Human CD19 Protein, N-His |
| HB996011 |
Recombinant Human CD19 Protein, C-His |
| Antibody |
HB996116 |
Research Grade Anti-Human CD19 (ALLO-501) |
| HB996126 |
Research Grade Anti-Human CD19 & CD3E Bispecific Antibody (AMG 562) |
| HB996136 |
Research Grade Anti-Human CD19 (VB119) |
| HB996146 |
Research Grade Anti-Human CD19 (UCART19) |
| HB996417 |
Anti-Human CD19 Antibody (FMC63), FITC |
| HB996737 |
Anti-Human CD19 Antibody (SAA2206), APC |
| HB996747 |
Anti-Human CD19 Antibody (SAA2206), PerCP |
| MB996014 |
Anti-Mouse CD19 Polyclonal Antibody |
| HB996023 |
Anti-Human CD19 Nanobody (SAA1246) |
| HB996033 |
Anti-Human CD19 scFv Antibody (FMC63) |
| HB996043 |
Anti-Human CD19 Nanobody (SAA2078) |
| HB996013 |
Anti-CD19 & CD28 Bispecific Antibody (RG-6333) |

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский









