Breaking Through the Subjective Limitations of CD25hi Gating with a FoxP3-Centric Treg Identification System
OMIP-006 establishes a FoxP3 nuclear stain-based, CD25-auxiliary Treg recognition logic, fully eliminating the subjective gating dependency on high CD25 expression. This panel provides highly standardized and reproducible Treg subtyping for frozen PBMCs, offering a consistent analytical foundation for multi-center clinical research.
Integrating Functional and Differentiation Markers to Systematically Reveal Treg Subset Heterogeneity
OMIP-006 integrates surface and intracellular markers like CD45RO, CD39, Helios, and CD49d, along with Boolean logic gates and SPICE visualization tools. This solution not only accurately identifies Tregs but also delves into their activation states, differentiation pathways, and functional subpopulations, providing a multi-level phenotypic framework for immune monitoring.
Optimized for Frozen PBMCs, Ensuring Cross-Laboratory Data Comparability
Systematically validated with HIV clinical trial samples, OMIP-006 has been comprehensively optimized for the staining stability, signal background control, and inter-batch consistency of frozen/thawed PBMCs. These optimizations significantly enhance the comparability and reproducibility of data across different experimental environments, supporting long-term sample storage and multi-center research needs
OMIP-006 provides an optimized method for Treg subtyping in frozen PBMCs, effectively avoiding the subjective nature of traditional CD25hi gating. The panel allows not only for accurate Treg identification but also for the assessment of their activation status, differentiation trajectory, and functional heterogeneity, providing a standardized analytical framework for immune monitoring in clinical samples.
1. OMIP-005 Panel
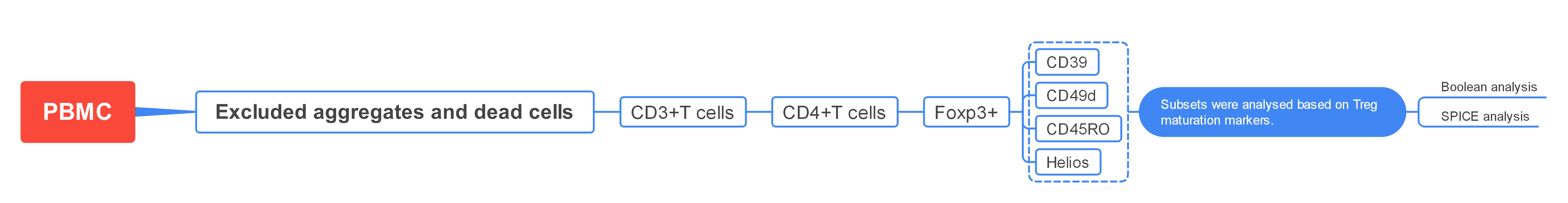
2. Gate Logic
1
Identification of Treg Cells
Exclude aggregates and dead cells. Then, gate on CD3+ T cells using forward scatter (FSC) and side scatter (SSC). Finally, use CD25/FoxP3 gating to identify double-positive Tregs.
2
Analysis of Treg Subsets
Treg subsets are further analysed using markers such as CD45RO, CD39, and CD49d to distinguish different functional states and differentiation pathways.
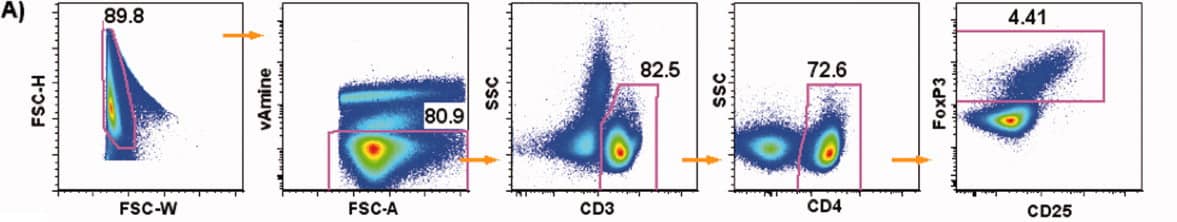
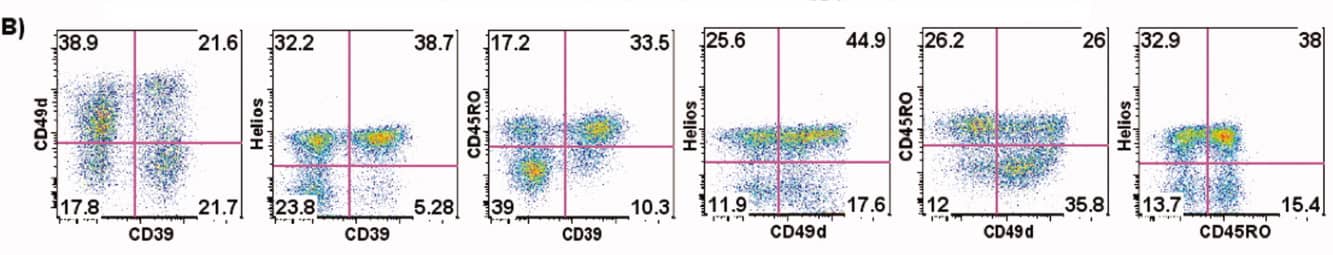
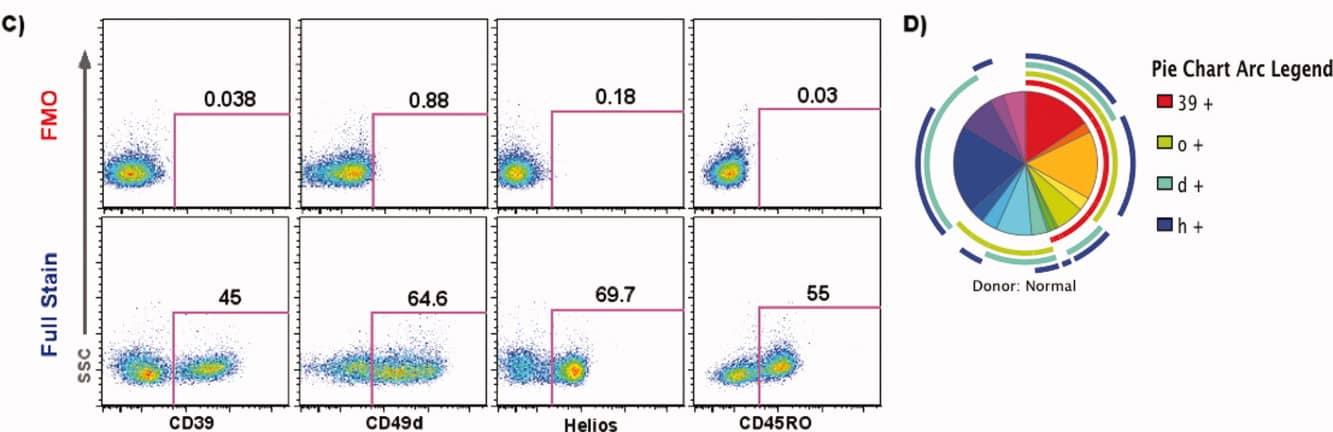
3. Experimental Results
1). Exclude aggregates, then use FSC-A/dead cell discrimination dye to exclude dead cells. Gate on CD3+ lymphocytes using SSC. Finally, identify double-positive Tregs using a CD25/FoxP3 dot plot.
2). Treg subpopulations within CD4+FoxP3+ Tregs are further analysed based on maturation markers such as CD45RO, CD39, and Helios.
3). Boolean analysis is applied to identify Treg subsets (Figure C), and the complex results are visualised using SPICE (Figure D).
4. Panel Interpretation
4.1 Design Optimisation to Avoid Subjective CD25hi Gating
Traditional Treg detection depends on CD25hi expression, which is prone to operator bias. OMIP-006 uses FoxP3 nuclear staining as the primary marker, with CD25 serving as an auxiliary marker, thereby avoiding subjective gating and enhancing the standardisation of analysis.
4.2 FoxP3 Nuclear Staining Optimisation to Reduce Background Signal
OMIP-006 employs eBioscience Fix/Perm and FoxP3-PE (clone PCH101), with an additional blocking step and multiple washes before and after intracellular staining. These optimisations significantly reduce background signal, improving the overall staining quality.
4.3 Optimisation for Frozen Samples
OMIP-006 has been specifically optimised for the analysis of frozen/thawed PBMCs. Extensive validation using HIV clinical trial samples demonstrated that the panel maintains stable staining and consistent data performance in frozen sample conditions, despite the potential impact of cryopreservation. These optimisations are critical for long-term sample storage, transport, and the comparability of results across multi-centre studies.
4.4 Multi-Dimensional Treg Subset Analysis
OMIP-006 provides a comprehensive analysis of Treg subsets by integrating markers such as CD45RO, CD39, Helios, and CD49d within the CD4+FoxP3+ population. Boolean analysis and SPICE visualisation tools enable a clear understanding of Treg heterogeneity, providing an expandable framework for immune monitoring and disease research.
5. Applications
Autoimmune Disease Mechanism Studies Tumour Immune Microenvironment Analysis Infection Immunity and Vaccine Evaluation Immunotherapy Efficacy Assessment Treg Heterogeneity and Functional Analysis Immunosenescence and Chronic Inflammation Research
6. Conclusion
OMIP-006 presents a stable and standardised Treg detection system for use with frozen PBMCs, addressing the challenges of sample variability and inter-laboratory reproducibility. By integrating FoxP3 nuclear staining with CD25 as an auxiliary marker, this solution overcomes the limitations of traditional CD25hi gating methods.
The multi-dimensional analysis of Treg subsets, using markers like CD45RO, CD39, and Helios, along with Boolean analysis and SPICE visualisation, offers a reliable framework for immune monitoring, paving the way for advanced research in immunology and disease.
Get OMIP-006 Compatible Flow Cytometry Antibodies
abinScience provides validated Flow Cytometry Antibodies covering key targets in this panel, supporting your research on Treg cells
References
[1] Murdoch, D.M., Staats, J.S. and Weinhold, K.J. (2012), OMIP-006: Phenotypic subset analysis of human T regulatory cells via polychromatic flow cytometry. Cytometry, 81A: 281-283.

About Us
As a strategic venture of AtaGenix (established 2011), abinScience was founded in 2023 to deliver premium life science reagents that accelerate discovery. Our flow cytometry antibody products cover commonly used detection markers, with a wide variety to meet the research needs of multiple species (Human/Mouse/Rat/Dog/Hamster/Monkey, etc.). We provide stable and reliable support for scientific research.
Explore abinScience Flow Cytometry Antibodies

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский