Idiopathic Inflammatory Myopathies (IIM) constitutes a heterogeneous group of autoimmune disorders characterized primarily by chronic skeletal muscle inflammation and progressive muscle weakness. It frequently involves multiple systems including the skin, lungs, and joints. With complex etiologies and distinct subtype differentiation, the disease carries a significantly elevated mortality rate when complicated by Interstitial Lung Disease (ILD), making it a key research focus in rheumatology and neurology.
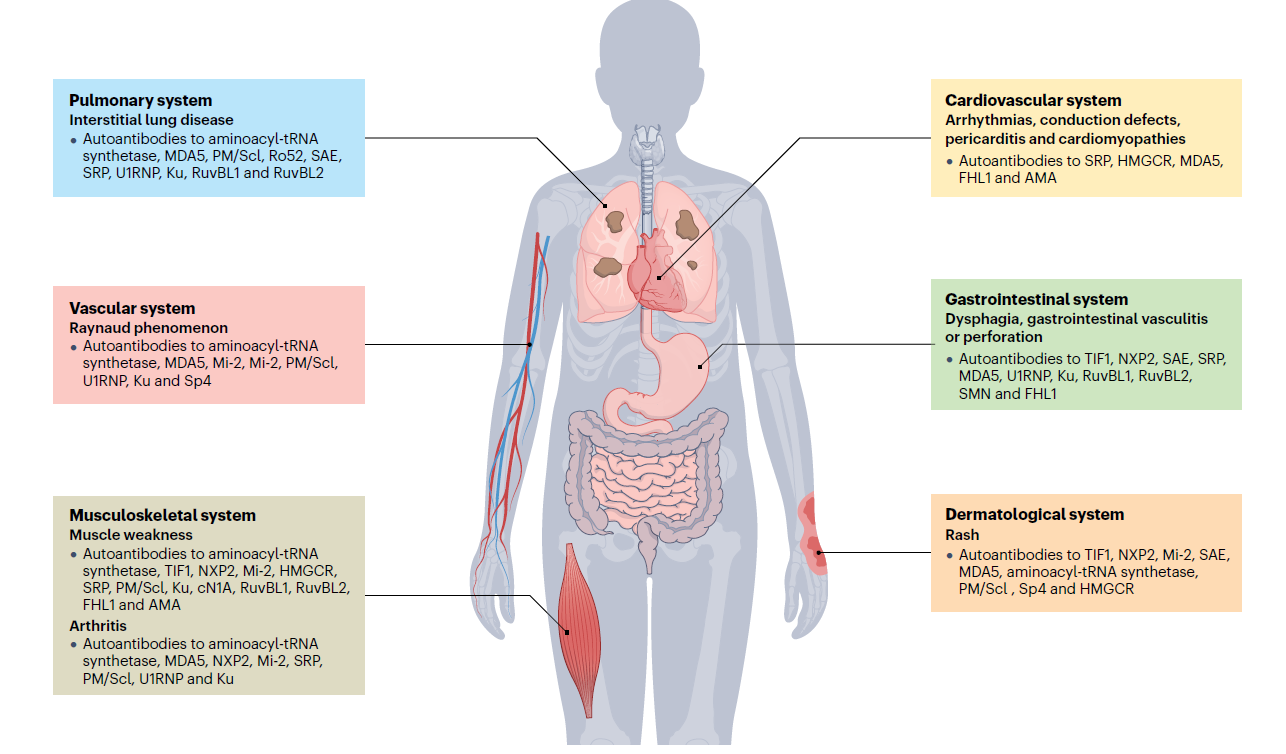
Figure 1. Manifestations of idiopathic inflammatory myopathies (IIM)
Traditionally, IIM was mainly classified into three subtypes: Polymyositis (PM), Dermatomyositis (DM), and Inclusion Body Myositis (IBM). However, with deeper understanding of the disease and the discovery of myositis-specific autoantibodies, the 2017 European League Against Rheumatism/American College of Rheumatology (EULAR/ACR) classification criteria further refined it into six major subtypes: Dermatomyositis (DM), Antisynthetase Syndrome (ASyS), Immune-Mediated Necrotizing Myopathy (IMNM), Inclusion Body Myositis (IBM), Polymyositis (PM), and Overlap Myositis (OM).
I. Molecular Mechanisms of the IIM Immune Regulatory Network: Analysis of Subtype-Specific Core Pathways
The pathogenesis of IIM stems from immune network dysregulation co-driven by genetic susceptibility (e.g., variants in IFIH1 and HLA-DRB1 genes) and environmental triggers (e.g., viral infections, tumor antigen exposure). Each subtype features distinct core regulatory pathways, and the crosstalk between these pathways serves as the key molecular basis for disease heterogeneity.
1. Humoral Immunity-Dominant Subtypes (DM/ASyS): Type I Interferon-Complement-Cytokine Network
- Core regulatory axis of DM: Involves IFIH1 gene variants → MDA5 recognition of self-antigens → activation of the Type I interferon-JAK-STAT1 pathway → upregulated expression of interferon-stimulated genes → B cell activation to produce specific autoantibodies (e.g., anti-MDA5/TIF1-γ) → activation of the classical complement pathway → perifascicular myofiber damage. Studies have confirmed that specific immune cells in the tissues of DM patients are the main source of interferons, and serum IFN-α levels are positively correlated with ILD progression.
- Molecular crosstalk mechanism of ASyS: The binding of antisynthetase antibodies to cell surface antigens can activate pathways such as NF-κB, releasing cytokines including IL-6 and CXCL8 to recruit immune cell infiltration and form a pro-inflammatory positive feedback loop. Recent research has revealed that anti-Jo-1 antibodies may induce ferroptosis in lung epithelial cells by upregulating ACSL4, providing a novel mechanistic perspective for ASyS-associated ILD.
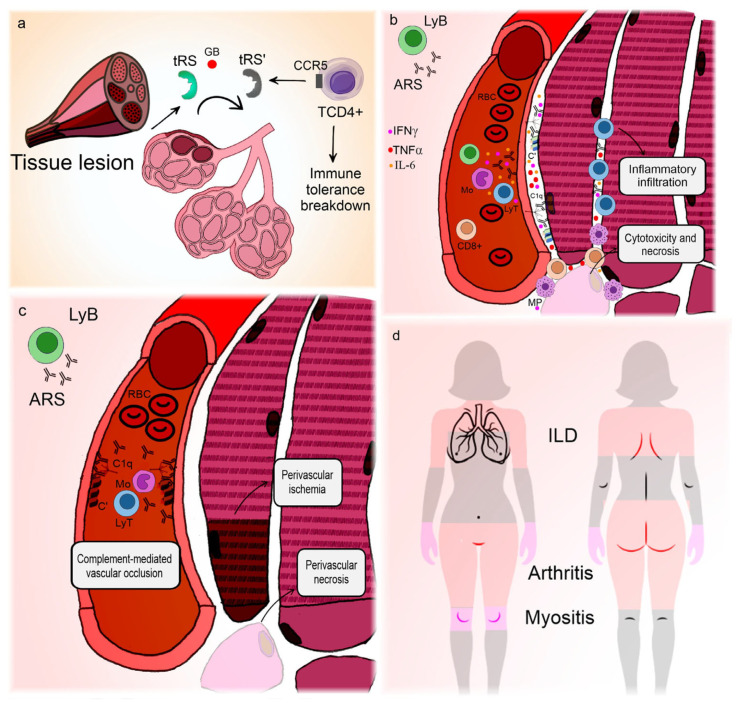
Figure 2. Pathogenesis and clinical manifestations of anti-synthetase syndrome
2. Cellular Immunity-Dominant Subtype (PM): Clonal Activation and Cytotoxic Mechanism of CD8+ T Cells
The core pathogenesis of PM lies in antigen-specific CD8+ T cell-mediated targeted killing of myofibers: abnormal overexpression of MHC-I molecules on the myofiber surface; restricted TCR clonal expansion of intramuscular CD8+ T cells; high expression of cytotoxic mediators such as perforin and granzyme B by effector T cells; and concurrent dysfunction of Treg cells leading to insufficient immune suppression and amplified tissue damage. Current research focuses on identifying specific self-antigens and deciphering the structure of the TCR-antigen-MHC complex.
3. Subtype with Intrinsic Myocellular Abnormalities (IBM): Dysregulation of the Autophagy-Protein Homeostasis-Mitochondrial Function Network
IBM research centers on "cell-autonomous damage": excessive activation of the mTOR pathway inhibits autophagy, resulting in the accumulation of abnormal proteins such as β-amyloid and TDP-43; mitochondrial DNA mutations and reactive oxygen species accumulation exacerbate protein misfolding; mild CD8+ T cell infiltration further aggravates autophagic impairment through IFN-γ secretion, forming synergistic damage. Currently, there is a lack of ideal animal models, and monotherapy with mTOR inhibitors yields limited efficacy, necessitating combination strategies targeting other pathways.
4. Necroptosis-Dominant Subtype (IMNM): Specific Activation of the RIPK3-MLKL Signaling Axis
The core of IMNM involves an "antibody-complement-necroptosis" cascade: anti-SRP/HMGCR antibodies activate the alternative complement pathway, forming membrane attack complexes that damage the myolemma, which in turn activates the RIPK3-MLKL pathway via DAMPs. A key characteristic is significantly elevated MLKL phosphorylation levels with no substantial change in caspase-3 activity. Small-molecule inhibitors targeting RIPK3-MLKL are currently a major research focus.
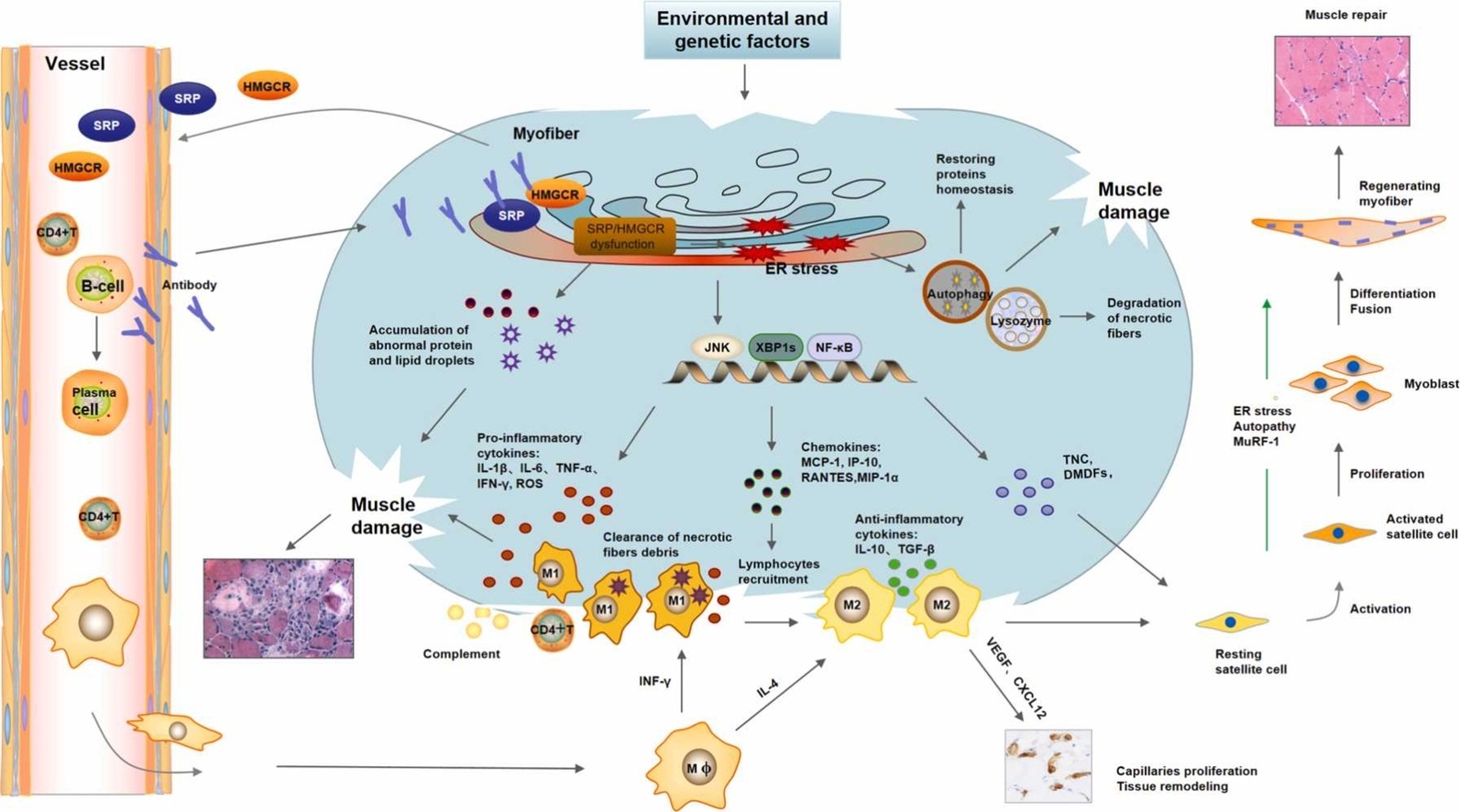
Figure 3: IMNM muscle damage and repair
5. Molecular Basis of Pathway Crosstalk
Core pathways of different subtypes interact through key nodes: for example, the Type I interferon pathway in DM can enhance NF-κB activity; IFN-γ induced by T cell activation in PM can feedback-upregulate MHC-I expression; complement components in IMNM can promote RIPK3 phosphorylation. Such network crosstalk provides the molecular rationale for disease heterogeneity and combination therapeutic strategies.
II. Key Immunological Features and Autoantibody Profiles
Autoantibodies are not only important diagnostic biomarkers for IIM but also critical effector molecules driving disease pathogenesis, determining clinical phenotypes, and influencing prognosis. Based on their specificity and clinical significance, they are mainly categorized into myositis-specific autoantibodies and myositis-associated autoantibodies.
Table 1: Major Autoantibodies in IIM and Their Clinical Significance
| Autoantibodies |
Subtypes |
Pathogenic Mechanisms |
| Anti-aminoacyl-tRNA synthetase antibodies |
ASyS |
These antibodies may directly induce tissue damage and are closely associated with activation of the Type I interferon pathway. |
| Anti-Mi-2 antibodies |
DM |
As components of the nucleosome remodeling complex, their specific pathogenic mechanisms remain under investigation. |
| Anti-TIF1-γ antibodies |
DM |
Linked to tumor immune surveillance, involving abnormal activation of CD8+ T cells. |
| Anti-MDA5 antibodies |
DM |
Closely associated with excessive activation of the Type I interferon signaling pathway and dysregulated formation of neutrophil extracellular traps (NETs), carrying a high mortality rate. |
| Anti-SRP antibodies / Anti-HMGCR antibodies |
IMNM |
These antibodies act directly on antigens on the myofiber surface, activating complement (e.g., C5b-9) and leading to myocyte necrosis. |
| Anti-cN1A antibodies |
IBM |
May interfere with protein homeostasis in muscle cells and contribute to myofiber degeneration. |
Additionally, the role of B cells in IIM has gained increasing attention. Studies have found that the proportions of B cell subsets (e.g., transitional B cells, naive B cells) in the peripheral blood of patients with active IIM are altered, with increased plasma cells directly correlated with autoantibody production.
III. Core Signaling Pathway Networks and Key Therapeutic Targets
The pathogenesis of IIM can be summarized as the abnormal activation and crosstalk of several core signaling pathways, which form the framework of potential targets for therapeutic intervention:
Type I interferon pathway: Initiates core inflammation in DM/ASyS, driving autoantibody production and tissue damage; key targets include IFNAR and the downstream JAK-STAT pathway.
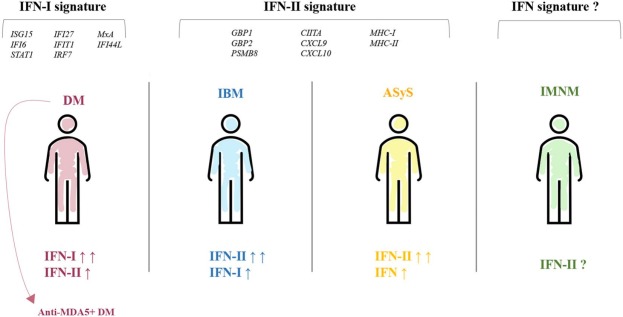
Figure 4: Different IFN signatures in IIM subtypes
JAK-STAT pathway: Serves as a common downstream hub for cytokine signal transduction, mediating signals from Type I/II interferons, IL-6, and other pro-inflammatory cytokines.
T/B cell activation and co-stimulation pathways: Mediate specific immune responses; targets include CD20, CD19, CD38, and CD6.
Complement activation pathway: Acts as a direct effector arm causing tissue damage in DM/IMNM; components such as C5 are potential targets.
Cell death/homeostasis pathways: Include the necroptosis pathway (RIPK3/MLKL) in IMNM and the autophagy/protein homeostasis pathway (mTOR, TDP-43) in IBM.
Inflammatory hub pathway: Centered on NF-κB, it extensively integrates upstream signals to amplify and sustain the inflammatory microenvironment.
Table 2: Core Pathways Involved in IIM Pathogenesis and Therapeutic Targets
| Core Pathways/Mechanisms |
Associated Subtypes |
Representative Research/Therapeutic Targets |
| Type I interferon signaling pathway |
DM, ASyS |
JAK1/2/3, TYK2, interferon receptors |
| Complement activation pathway |
DM, IMNM |
C5, C3 |
| T/B cell activation and co-stimulation pathways |
PM, ASyS, Multiple subtypes |
CD20, CD19, CTLA-4-Ig |
| Necroptosis pathway |
IMNM |
RIPK3, MLKL |
| Autophagy/protein homeostasis pathway |
IBM |
mTOR, TDP-43, p62 |
| Cytokine network |
Multiple subtypes |
IL-6, IL-21, TNF-α |
IV. Cutting-Edge Research Advances: Mechanistic Breakthroughs and Therapeutic Innovations
Recent research has moved beyond traditional pathological descriptions, advancing toward in-depth directions such as molecular subtyping, signaling pathways, and novel intervention strategies.
1. In-Depth Exploration of Molecular Mechanisms
2. Evolution of Therapeutic Strategies
- Expansion of targeted therapy applications: JAK inhibitors, B cell depletion therapies, and other agents are transitioning from preclinical exploration to partial clinical application, providing new options for refractory patients.
- Personalized and combination therapy: Precision stratification based on immune characteristics such as autoantibody profiles and pathway activity to guide combination regimens (e.g., JAK inhibitors combined with calcineurin inhibitors) is a key direction to improve treatment efficacy.
- Exploration of novel intervention models: Small-molecule inhibitors targeting specific pathways (e.g., RIPK3-MLKL) and degraders promoting the clearance of abnormal proteins are currently in preclinical or early clinical research.
V. Conclusion
Idiopathic Inflammatory Myopathies (IIM) are rapidly advancing autoimmune diseases. The shift from clinical to autoantibody/molecular-based subtyping reflects deeper pathogenic understanding, with core pathways like Type I interferon and JAK-STAT enabling targeted therapy. Key challenges include IBM’s treatment dilemma, unclear subtype mechanisms, and personalized medicine. Future progress relies on multi-omics research to develop novel biomarkers and precision drugs, supported by advanced research tools.
abinScience IIM Research Solutions
1. Antibodies
| Product Name |
Catalog No. |
| InVivoMAb Anti-Human TNFa/TNF-alpha (Iv0050) |
HF879010 |
| Anti-Human MET/c-Met/HGFR Antibody (SAA0765) |
HY196013 |
| Anti-Human GARS1/Glycyl-tRNA synthetase Antibody (SAA1935) |
HW710013 |
| Anti-Human EN2 Antibody (SAA1595) |
HB086013 |
| Anti-Human EPRS1 Polyclonal Antibody |
HY479014 |
| Anti-JAK1 Polyclonal Antibody |
HB829014 |
| Anti-TNFa/TNF-alpha Polyclonal Antibody |
HF879014 |
View more IIM Antibodies
2. Assay Kits
View more IIM Assay Kits
3. Research Biosimilars
| Target |
Product Name |
Catalog No. |
| TNF-α |
Research Grade Adalimumab |
HF879026 |
| Research Grade Anti-Human TNFa/TNF-alpha (BOW050) |
HF879136 |
| Research Grade Golimumab |
HF879046 |
| Research Grade Etanercept |
HF879296 |
| Research Grade Onercept |
HF879386 |
| MET |
Research Grade Davutamig |
HY196186 |
| Research Grade Emibetuzumab |
HY196016 |
| Research Grade Onartuzumab |
HY196026 |
| Research Grade Anti-Human MET/c-Met/HGFR (ABBV-400) |
HY196046 |
View more IIM Research Biosimilars
abinScience offers high-quality research tools covering core mechanisms and cutting-edge targets in Idiopathic Inflammatory Myopathies (IIM) research. We provide highly specific antibodies and recombinant proteins targeting the Type I interferon pathway (e.g., IFNAR, JAK/STAT family), B cell and plasma cell markers (e.g., CD19, CD20, CD38), complement components, key cell death proteins (e.g., RIPK3, MLKL, ACSL4), and molecules involved in autophagy and protein homeostasis (e.g., mTOR, TDP-43). Additionally, our Anti-Drug Antibody (ADA) detection kits and Pharmacokinetic (PK) kits deliver accurate, reliable data support for immunogenicity assessment and pharmacokinetic research of targeted biologics (e.g., JAK inhibitors, monoclonal antibodies, and Research Biosimilars). We empower researchers to delve into IIM molecular subtyping, signaling networks, and therapeutic responses, accelerating the innovation process from basic research to clinical translation.
References
[1] Lundberg IE, Fujimoto M, Vencovsky J, Aggarwal R, Holmqvist M, Christopher-Stine L, Mammen AL, Miller FW. Idiopathic inflammatory myopathies. Nat Rev Dis Primers. 2021 Dec 2;7(1):87. doi: 10.1038/s41572-021-00325-7. PMID: 34857780; PMCID: PMC10425161.
[2] Raaphorst J, van der Kooi AJ, Mecoli CA, Weihl CC, Tas SW, Schmidt J, de Visser M. Advances in the classification and management of idiopathic inflammatory myopathies. Lancet Neurol. 2025 Sep;24(9):776-788. doi: 10.1016/S1474-4422(25)00233-9. Erratum in: Lancet Neurol. 2025 Oct;24(10):e12. doi: 10.1016/S1474-4422(25)00320-5. PMID: 40818476.
[3] Connolly CM, Gupta L, Fujimoto M, Machado PM, Paik JJ. Idiopathic inflammatory myopathies: current insights and future frontiers. Lancet Rheumatol. 2024 Feb;6(2):e115-e127. doi: 10.1016/S2665-9913(23)00322-3. PMID: 38267098.
[4] Lundberg IE, Miller FW, Tjärnlund A, Bottai M. Diagnosis and classification of idiopathic inflammatory myopathies. J Intern Med. 2016 Jul;280(1):39-51. doi: 10.1111/joim.12524. PMID: 27320359; PMCID: PMC5021058.
[5] Allameen NA, Ramos-Lisbona AI, Wedderburn LR, Lundberg IE, Isenberg DA. An update on autoantibodies in the idiopathic inflammatory myopathies. Nat Rev Rheumatol. 2025 Jan;21(1):46-62. doi: 10.1038/s41584-024-01188-4. Epub 2024 Nov 28. PMID: 39609638.
[6] Gasparotto M, Franco C, Zanatta E, Ghirardello A, Zen M, Iaccarino L, Fabris B, Doria A, Gatto M. The interferon in idiopathic inflammatory myopathies: Different signatures and new therapeutic perspectives. A literature review. Autoimmun Rev. 2023 Jun;22(6):103334. doi: 10.1016/j.autrev.2023.103334. Epub 2023 Apr 15. PMID: 37068699.
[7] Wu H, Li X, Xu H, Li Z, Feng F, Zhang J, Xu Z, Ni H, Guo Y, Li Y. Malignancy in Idiopathic Inflammatory Myopathies: Recent Insights. Clin Rev Allergy Immunol. 2025 Aug 18;68(1):83. doi: 10.1007/s12016-025-09080-z. PMID: 40824431; PMCID: PMC12361316.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский






