2025 Latest Review of Systemic Lupus Erythematosus (SLE) Research: Core Targets, Clinical Progress, and Research Tools
Systemic Lupus Erythematosus (SLE) is a classic systemic autoimmune disease characterized by the production of antinuclear antibodies, immune complex deposition, and chronic inflammation in multiple organs. It predominantly affects women of childbearing age (female-to-male ratio approximately 9:1), with a relatively high incidence in the Chinese population and increasing diagnosis rates in recent years. Typical symptoms include the malar (butterfly) rash, arthritis, nephritis (lupus nephritis), hematologic abnormalities, and neuropsychiatric manifestations. Onset usually occurs between ages 20 and 40, with a relapsing-remitting course that can lead to organ failure, cardiovascular events, or infections.
The fundamental pathogenesis of SLE involves genetic susceptibility, environmental triggers (such as UV light, infections, and smoking), and breakdown of immune tolerance, resulting in defective clearance of apoptotic debris, nucleic acid exposure, excessive activation of the type I interferon (IFN-I) pathway, B-cell hyperactivation producing autoantibodies, T-cell dysregulation, and enhanced neutrophil NETosis. These processes ultimately create an inflammatory cascade that amplifies tissue damage.
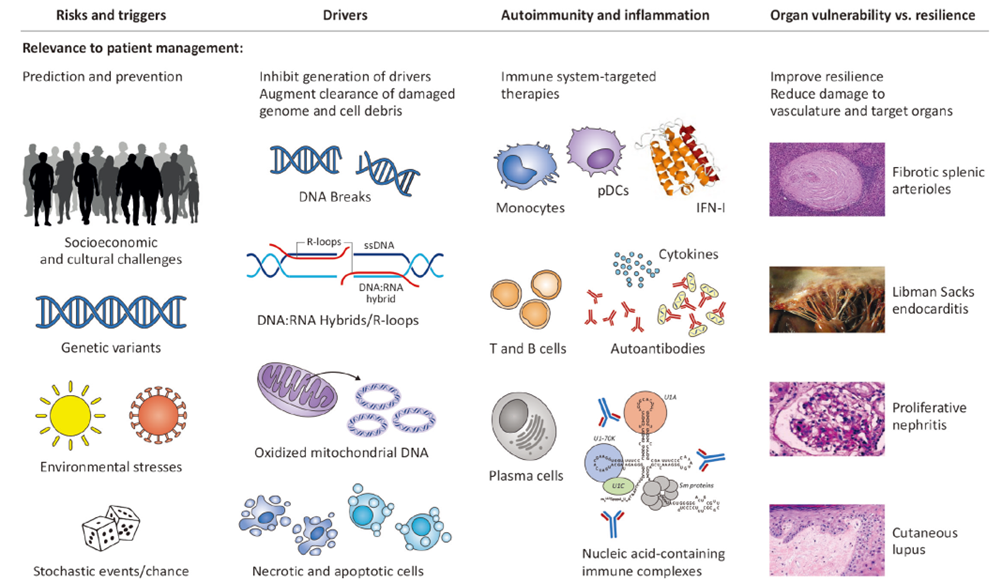
Fig 1. A broad view of the pathogenesis of SLE.
Key Targets and Antibodies in SLE Research
Although SLE is driven by multiple factors, its downstream pathological network is highly complex, involving multiple pathways such as IFN-I signaling, B-cell survival, T-cell costimulation, and complement activation.
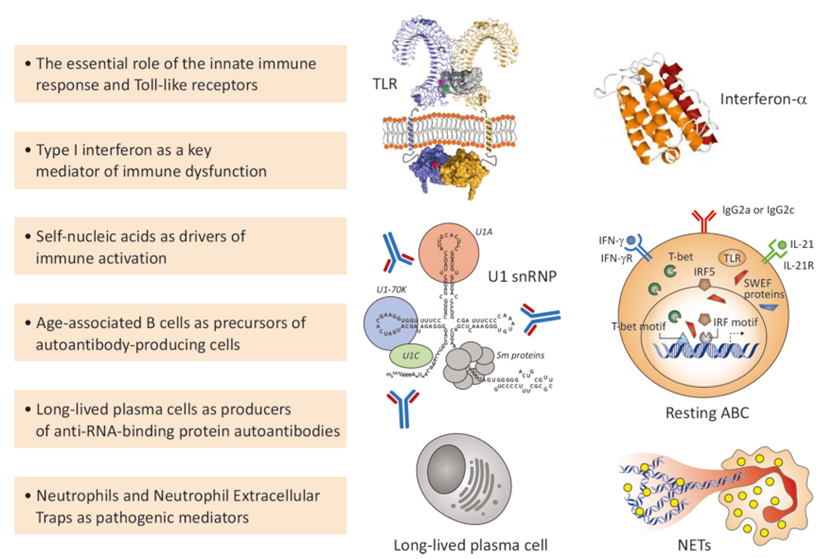
Fig 2. Selected key scientific advances relevant to SLE pathogenesis
The following table systematically summarizes currently recognized core targets and their roles in SLE, providing a theoretical foundation for targeted interventions.
| Target |
Normal Biological Function |
Pathological Role in SLE |
Research Applications |
| IFNAR (Type I IFN receptor) |
Mediates antiviral and immunoregulatory effects of IFN-I |
Overactivation leads to IFN signature, amplified inflammation, and autoantibody production |
IFN signature detection, validation of Anifrolumab efficacy |
| BAFF / APRIL |
B-cell survival and maturation factors |
Overexpression promotes survival of autoreactive B cells and plasma cell differentiation |
BAFF level ELISA, screening for Belimumab/Telitacicept |
| CD20 |
B-cell surface marker |
B-cell hyperactivation producing autoantibodies |
B-cell depletion experiments, validation of Rituximab/Obinutuzumab |
| CD19 / BCMA |
B-cell and plasma cell markers |
Sustained autoantibody production and immune complex formation |
CAR-T targeting depletion, evaluation of deep B-cell clearance |
| BDCA2 (on pDCs) |
pDC surface receptor that inhibits IFN production |
Functional defects lead to excessive IFN-α production by pDCs |
pDC activation detection, screening for Litifilimab inhibitors |
In-Depth Interpretation of Core SLE Pathogenic Mechanisms (2025 Latest Consensus)
Research from 2024–2025 has further clarified the multi-pathway synergistic pathogenesis of SLE. The core mechanisms can be summarized into three key stages:
- Upstream Trigger: Nucleic Acid Exposure and IFN-I Storm — Defective clearance of apoptotic/necrotic cells leads to release of nucleic acids (DNA:RNA hybrids, R-loops, oxidized mitochondrial DNA), which activate pDCs via TLRs to produce large amounts of IFN-α, establishing the “IFN signature” and initiating systemic inflammation.
- Midstream Amplification: Abnormal B-Cell Activation and Autoantibody Production — Excessive BAFF/APRIL expression breaks B-cell tolerance, promoting differentiation of autoreactive B cells into long-lived plasma cells that continuously produce antinuclear antibodies, anti-dsDNA antibodies, etc., forming immune complexes deposited in target organs.
- Downstream Damage: Immune Complex Deposition and Tissue Inflammation — Immune complexes deposit in kidneys, skin, blood vessels, and other tissues, activating the complement system, recruiting neutrophils to release NETs, and inducing local inflammation, vascular damage, and fibrosis, ultimately causing multi-organ dysfunction.
Recent research highlights: Age-associated B cells (ABCs) have been confirmed as key precursor cells for autoantibody production; enhanced NETosis forms a positive feedback loop with the IFN-I pathway; gut microbiome dysbiosis contributes to disease initiation through mucosal immune abnormalities, opening new therapeutic targets.
Therapeutic Implications: Blocking the IFN-I pathway (Anifrolumab), inhibiting BAFF (Belimumab/Telitacicept), B-cell depletion (Rituximab), and CD19 CAR-T–mediated deep immune reset are currently the most promising disease-modifying strategies.
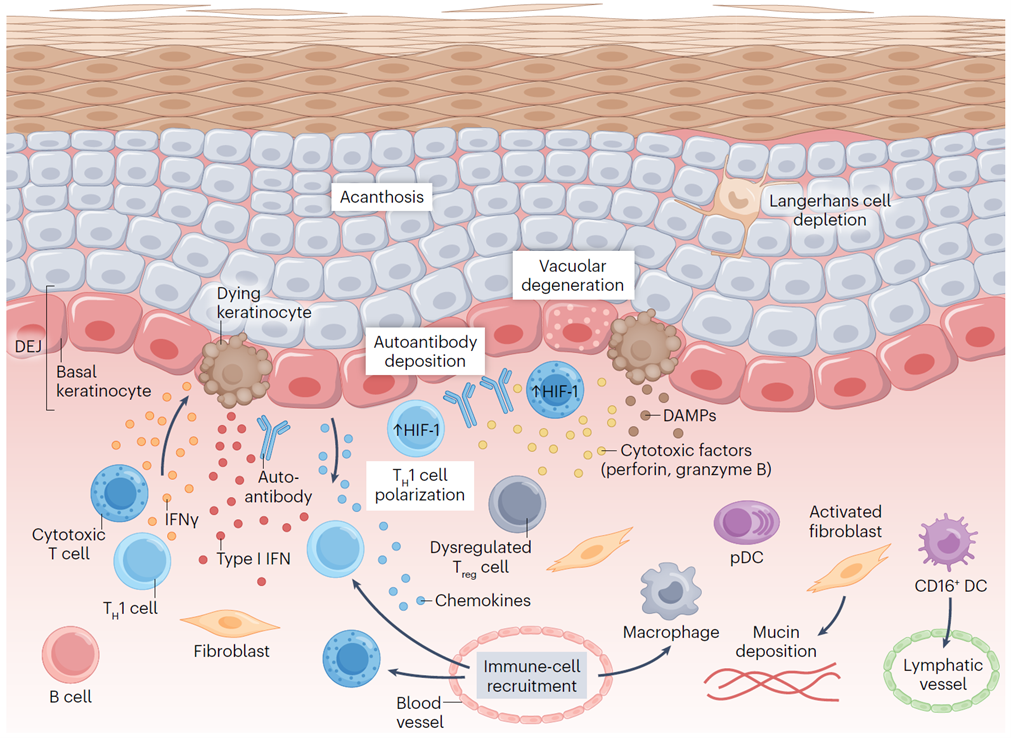
Fig 3. Inflammatory circuits in lesional lupus skin.
Latest Advances in SLE Research
Clarification of Core Pathways and Triggers: The type I interferon (IFN-I) pathway has been confirmed as the central driver of immune dysregulation, closely linked to abnormal B-cell activation and autoantibody production. Real-world data for Anifrolumab (anti-IFNAR mAb) confirm significant efficacy in skin and joint manifestations with good long-term safety.
Pathway Blockade and Innovative Drugs: The Chinese-developed dual-target biologic Telitacicept, which blocks both BLyS and APRIL, significantly improves response rates in moderate-to-severe patients. Drugs targeting the IFN-I receptor (Anifrolumab), TYK2 inhibitors (Deucravacitinib), and new targets such as TLR7 and IRAK4 are entering clinical use, offering new options for patients refractory to conventional therapy.
Cell Therapy Innovations Lowering Barriers: In vivo CAR-T therapy uses LNP vectors to generate CAR-T cells directly in patients without chemotherapy preconditioning, rapidly depleting pathogenic B cells with improved safety. Multiple trials show drug-free remission after deep B-cell depletion, with some patients remaining relapse-free for over two years.
Optimized Clinical Assessment: Revised SRI-4 criteria combined with dynamic monitoring of complement and anti-dsDNA antibodies more accurately reflect treatment response. For target organ involvement such as lupus nephritis, individualized regimens combining biologics and immunosuppressants are emerging, alongside comprehensive management emphasizing sun protection and microbiome modulation to reduce relapse risk.
Current Research Challenges in SLE
Numerous studies highlight that the greatest obstacles in SLE are strong patient heterogeneity and the lack of reliable early biomarkers, making it difficult to rapidly validate and personalize novel therapies such as CAR-T. Concurrent activation of IFN-I, excessive B-cell survival, T-cell dysregulation, and NETosis collectively drive the disease; as recent studies emphasize, single-target interventions rarely alter the disease trajectory completely. Increasingly, research is shifting toward multi-target combinations or cellular therapies to achieve more robust breakthroughs. These challenges also underscore the importance of multi-pathway systematic research—abinScience’s tools targeting IFNAR, BAFF, CD20, and CD19 are designed precisely for this purpose.
Why Choose abinScience SLE Research Tools?
- Coverage of core SLE mechanisms including the IFN-I pathway, B-cell survival (BAFF/APRIL), and B-cell depletion (CD20/CD19);
- InVivoMAb-grade antibodies (low endotoxin) suitable for long-term administration in mouse, rat, and non-human primate SLE models;
- Multicolor flow cytometry antibodies (APC/FITC/PE/PerCP) that fully support subset sorting and functional analysis of peripheral blood B cells/plasma cells;
- All products undergo rigorous QC to ensure batch-to-batch consistency and high activity, enabling rapid and reliable construction of in vitro and in vivo SLE models.
Below are the latest abinScience recombinant proteins and antibodies targeting core SLE pathways. Catalog numbers link directly to product pages.
1. Recombinant Proteins
| Catalog No. |
Product Name |
| HC260012 |
Recombinant Human BTK Protein, N-His |
| HB199012 |
Recombinant Human CD154/CD40LG/TNFSF5 Protein, C-His |
| HB199011 |
Recombinant Human CD154/CD40LG/TNFSF5 Protein, C-His |
| HB996012 |
Recombinant Human CD19 Protein, N-His |
| HV212021 |
Recombinant Human CD257/BAFF/TNFSF13B Protein, C-His |
| HV212011 |
Recombinant Human CD257/BAFF/TNFSF13B Protein, N-Fc |
| HV212012 |
Recombinant Human CD257/TNFSF13B Protein, N-His |
| HT248011 |
Recombinant Human CD267/TNFRSF13B Protein, C-Fc |
| HX959011 |
Recombinant Human CD269/TNFRSF17 Protein, C-Fc |
| HX959021 |
Recombinant Human CD269/TNFRSF17/BCMA Protein, C-Fc |
| HV560012 |
Recombinant Human CD303/CLEC4C Protein, N-His |
| HB103011 |
Recombinant Human CD32b/FCGR2B Protein, C-His |
| HB103012 |
Recombinant Human CD32b/FCGR2B Protein, N-His |
| HB823011 |
Recombinant Human IFNAR1 Protein, C-His |
| HB823012 |
Recombinant Human IFNAR1 Protein, N-His |
| HB769011 |
Recombinant Human IL12B/IL-12 p40/NKSF2 Protein, C-Flag |
| HB769021 |
Recombinant Human IL12B/IL-12 p40/NKSF2 Protein, C-His |
| HB829022 |
Recombinant Human JAK1 Protein, C-His |
| HB829012 |
Recombinant Human JAK1 Protein, N-His |
| HC519012 |
Recombinant Human PPP3CA Protein, N-His |
| HB039012 |
Recombinant Human TYK2 Protein, N-His |
Click to show remaining recombinant protein products (21 total)
2. Antibodies
| Catalog No. |
Product Name |
| HX959016 |
Research Grade Belantamab |
| HV212016 |
Research Grade Belimumab |
| HB769026 |
Research Grade Briakinumab |
| HB996326 |
Research Grade Budoprutug |
| HB996016 |
Research Grade Coltuximab |
| HB199026 |
Research Grade Dapirolizumab |
| HB996056 |
Research Grade Denintuzumab |
| HB769036 |
Research Grade Ebdarokimab |
| HF565026 |
Research Grade Faralimomab |
| HB199056 |
Research Grade Frexalimab |
| HY257036 |
Research Grade Ibritumomab |
| HX959076 |
Research Grade Ispectamab |
| HB199016 |
Research Grade Letolizumab |
| HV560016 |
Research Grade Litifilimab |
| HB996106 |
Research Grade MDX-1342 |
| HB996076 |
Research Grade Obexelimab |
| HY257466 |
Research Grade Ofatumumab |
| HX959286 |
Research Grade pamlectabart |
| HY257076 |
Research Grade Ripertamab |
| HB199046 |
Research Grade Toralizumab |
| HY257066 |
Research Grade Ublituximab |
| HB769016 |
Research Grade Ustekinumab |
| HF941046 |
Research Grade Zigakibart |
| HB199086 |
Research Grade Anti-Human CD154/CD40LG/TNFSF5 (ABI793) |
| HB996126 |
Research Grade Anti-Human CD19 & CD3E Bispecific Antibody (AMG 562) |
| HY257456 |
Research Grade Anti-Human CD20/MS4A1 Antibody (KM3575) |
| HF941026 |
Research Grade Anti-Human CD256/TNFSF13/APRIL (BION 1301) |
| HX959116 |
Research Grade Anti-Human CD269/TNFRSF17/BCMA (ALLO-605) |
| HB103026 |
Research Grade Anti-Human CD32b/FCGR2B (BI-1607) |
| HB823036 |
Research Grade Anti-Human IFNAR1 (QX 006N) |
| HB769046 |
Research Grade Anti-Human IL12B/IL-12 p40/NKSF2 (CEP-37248) |
| AB996023 |
Anti-4G7 scFv Monoclonal Antibody (SAA1997) |
| HB996013 |
Anti-CD19 & CD28 Bispecific Antibody (RG-6333) |
| HY257014 |
Anti-CD20/MS4A1 Polyclonal Antibody |
| HB103014 |
Anti-CD32b/FCGR2B Polyclonal Antibody |
| AB996013 |
Anti-FMC63 scFv Monoclonal Antibody (SAA1996) |
| AB996033 |
Anti-FMC63 scFv Monoclonal Antibody (SAA2162) |
| HB199207 |
Anti-Human CD154/CD40LG/TNFSF5 Antibody (5C8) |
| HB199107 |
Anti-Human CD154/CD40LG/TNFSF5 Antibody (SAA0549) |
| HB996307 |
Anti-Human CD19 Antibody (4G7) |
| HB996043 |
Anti-Human CD19 Nanobody (SAA2078) |
| HY257107 |
Anti-Human CD20/MS4A1 Antibody (SAA0006) |
| HY257013 |
Anti-Human CD20/MS4A1 Nanobody (SAA1332) |
| HF941107 |
Anti-Human CD256/TNFSF13 Antibody (VIS-649) |
| HT248013 |
Anti-Human CD267/TNFRSF13B Antibody (SAA1832) |
| HX959307 |
Anti-Human CD269/TNFRSF17/BCMA Antibody (J22.9-xi) |
| HV974093 |
Anti-Human CD274/PD-L1/B7-H1 Antibody (405.9A11) |
| HB103207 |
Anti-Human CD32b/FCGR2B Antibody (6G08) |
| HB823013 |
Anti-Human IFNAR1 Antibody (64G12) |
| HB769107 |
Anti-Human IL12B/IL-12 p40/NKSF2 Antibody (SAA0381) |
| HB823010 |
InVivoMAb Anti-Human IFNAR1 Antibody (H3K1) |
| HB769010 |
InVivoMAb Anti-Human IL12B/IL-12 p40/NKSF2 (Iv0026) |
Click to show remaining antibody products (42 total)
3. Kits
Click to show remaining kit products (12 total)
abinScience — Empowering SLE Research with High-Purity, High-Activity Recombinant Proteins and Ultra-High-Specificity Antibodies (Including Nanobodies)
abinScience continues to support mechanistic studies, drug screening, and biomarker development in SLE, helping researchers unravel autoimmune diseases faster while building critical target tool matrices for rheumatoid arthritis, Sjögren’s syndrome, ankylosing spondylitis, and other autoimmune conditions.
References
- Elia A, Zucchi D, Silvagni E, et al. Systemic lupus erythematosus: one year in review 2025. Clin Exp Rheumatol. 2025;43(3):397-406. doi:10.55563/clinexprheumatol/m0pi1k
- Shumnalieva R, Padjen I, Monov S, Velikova T (2025) CAR T-cell therapy in systemic lupus erythematosus: a scoping review. Pharmacia 72: 1-13. https://doi.org/10.3897/pharmacia.72.e167410
- Saegusa K, Tsuchida Y, Komai T, Tsuchiya H, Fujio K. Advances in Targeted Therapy for Systemic Lupus Erythematosus: Current Treatments and Novel Approaches. Int J Mol Sci. 2025;26(3):929. Published 2025 Jan 23. doi:10.3390/ijms26030929
- Scherlinger, M., Kolios, A.G.A., Kyttaris, V.C. et al. Advances in the treatment of systemic lupus erythematosus. Nat Rev Drug Discov 24, 926–944 (2025). https://doi.org/10.1038/s41573-025-01242-0
- Zhou J, Lei B, Shi F, et al. CAR T-cell therapy for systemic lupus erythematosus: current status and future perspectives. Front Immunol. 2024;15:1476859. Published 2024 Dec 19. doi:10.3389/fimmu.2024.1476859
We detected that your visit originates from China.
Would you prefer to continue in Chinese?
This will switch to the Chinese-language model.
Your data will be processed according to local laws.

 中文
中文 English
English 한국어
한국어 日本語
日本語 Español
Español Français
Français Русский
Русский

